Carbohydrates 1 Photosynthesis The process by which green

Carbohydrates 1
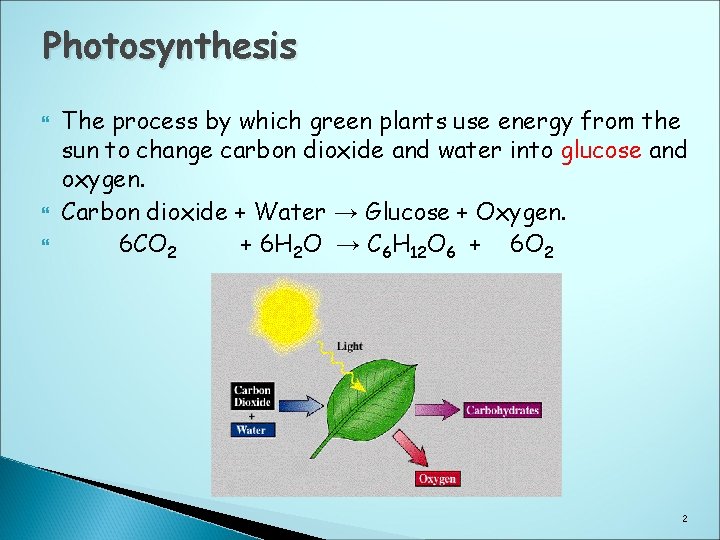
Photosynthesis The process by which green plants use energy from the sun to change carbon dioxide and water into glucose and oxygen. Carbon dioxide + Water → Glucose + Oxygen. 6 CO 2 + 6 H 2 O → C 6 H 12 O 6 + 6 O 2 2

Classification & Sources Class Chemical Formula Example Source Monosaccharides C 6 H 12 O 6 Glucose Fructose Galactose Fruit Honey Digested milk Disaccharides C 12 H 22 O 11 Maltose=Glucose+Glucose Sucrose=Glucose+Fructose Lactose=Glucose+Galactose Barley Table sugar Milk Polysaccharides (Complex Carbs) (C 6 H 10 O 5)n Starch Cellulose Pectin Glycogen non-starch polysaccharides Bread, pasta Whole cereals Fruit cell wall Liver and muscle cells 3

Energy Value & Dietary Recommendations Energy Value- 1 g carbohydrate= 4 kcal/17 kj of energy Over consumption of sugar & refined starches & lack of cellulose leads to; ◦ Obesity ◦ Dental Caries ◦ Bowel Disorders Increase- wholemeal bread, pasta & rice, skins of fruit & vegetables, high fibre cereals Decrease- white bread, pasta & rice, fruit juices, high sugar breakfast cereals, biscuits & cakes 4

Biological Functions of Carbohydrates Used for heat & energy They spare protein so it can be used for growth and repair. Excess carbohydrate is changed to glycogen and stored in liver and muscle as an energy reserve or it is changed to body fat (adipose tissue) which insulates the body. Cellulose moves food through intestine preventing constipation. 5

Classification of Carbohydrates There are 3 types of carbohydrates 1. 2. 3. Monosaccharides Disaccharides Polysaccharides 6
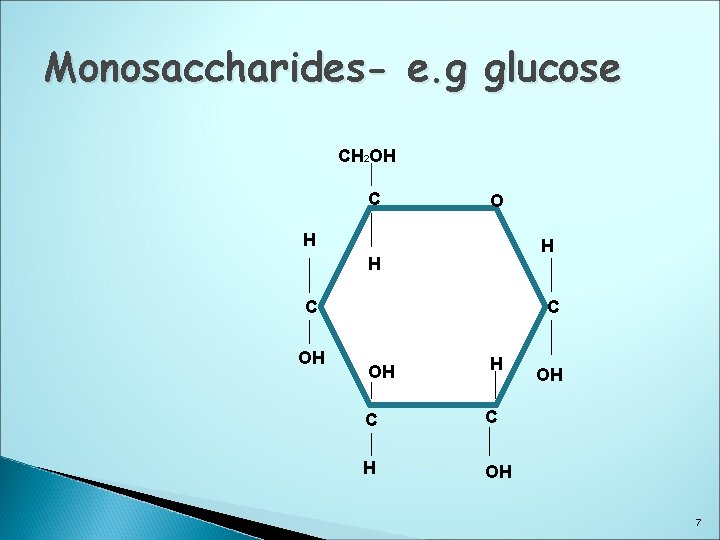
Monosaccharides- e. g glucose CH 2 OH C O H H H C OH H C C H OH OH 7

Monosaccharides A Monosaccharide contains one sugar unit C 6 H 12 O 6 is the chemical formula of a monosaccharide Glucose, fructose and galactose are the 3 monosaccharides 8

Disaccharides Are formed when two mono saccharides join together with the elimination of water (condensation) There are three disaccharides: maltose, sucrose & lactose The chemical formula is C 12 H 22 O 11 C 6 H 12 O 6 +C 6 H 12 O 6 C 12 H 24 O 12 - H 2 O C 12 H 22 O 11 9
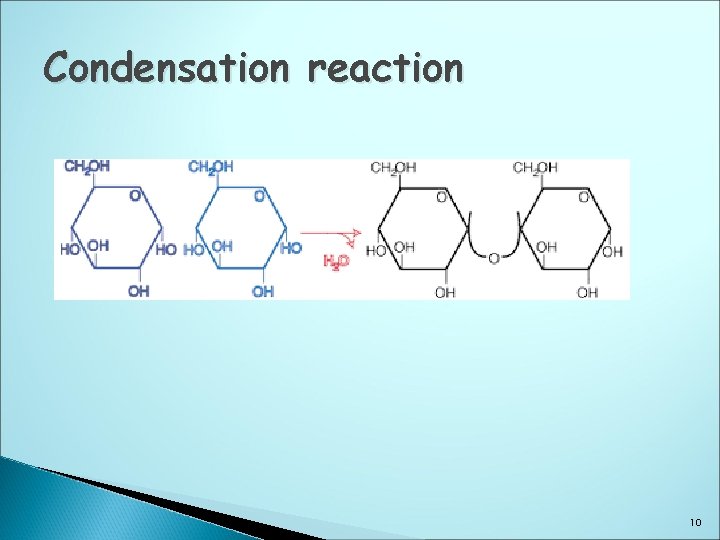
Condensation reaction 10

Polysaccharides These are formed when three or more monosaccharides join together with a loss of a water molecule each time. They may be straight or branched Examples: Starch, pectin, cellulose, gums & glycogen Pectin, cellulose & gums are also known as Non-Starch Polysaccharides Starch is made up of glucose units arranged as follows: 1. Straight chains are known as amylose or 2. Branched chains are known as amylopectin 11
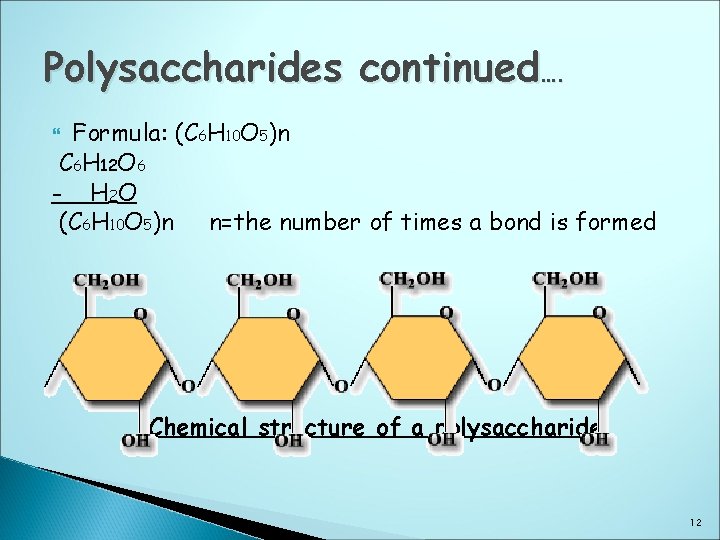
Polysaccharides continued…. Formula: (C 6 H 10 O 5)n C 6 H 12 O 6 - H 2 O (C 6 H 10 O 5)n n=the number of times a bond is formed Chemical structure of a polysaccharide 12

Non-Starch Polysaccharides These are also known as NSPs, dietary fibre and roughage NSPs cannot be digested in the body and absorb large amounts of water They aid the removal of waste from the body by a process known as peristalsis Peristalsis is the muscular movement of food along the gut Sources of NSPs include wholemeal bread, brown rice & wholemeal pasta Refined foods contain few if any NSPs 13

Properties of Carbohydrates 1. 2. 3. Sugar Starch Non-Starch Polysaccharides 14

1. Properties of Sugar 1. Solubility v Sugars are white crystalline compounds that are soluble in water v Solubility is increased by heating the water v A syrup is formed when sugar is heated 2. Assists Aeration v Sugar denatures egg protein, enabling aeration to occur, e. g. in the making of sponge cakes – the egg when whisked with sugar becomes aerated 15

3. Crystallisation v This occurs if more sugar is added than can be absorbed by a liquid v Crystal particles are formed when the mixture cools v Crystallisation is used in the confectionery and sweet industry 16

1. Properties of Sugar cont…. 4. Caramelisation v When sugars are heated, they produce a range of brown substances know as a caramel v There are ten gradual changes in sugar between melting and caramelisation v These stages occur between 104°C & 177°C v Eventually, the heat will cause carbonisation (burning) 17

1. Properties of sugar cont…. 5. Maillard Reaction v Sugar (Carbohydrate) + Amino Acid + Dry Heat = Browning of foods, e. g. roast potatoes 6. Sweetness v Sugar has varying degrees of sweetness based on a point scale using the tasting method v Sucrose has a relative sweetness of 100 v Fructose has a relative sweetness of 170 v Lactose has a relative sweetness of 15 18

1. Properties of sugar cont…. 7. Hydrolysis v Hydrolysis is the chemical breakdown of a molecule by adding water to produce smaller molecules v This occurs when water is added to a disaccharide to produce two monosaccharides v Hydrolysis is the reverse of the condensation reaction 19

1. Properties of sugar cont…. 8. Inversion v The hydrolysis of sucrose is also known as the inversion of sucrose (mixture of glucose & fructose), known as ‘invert sugar’ v Inversion may be brought about by either: (a) heating sucrose with an acid; or (b) adding the enzyme invertase, or sucrase v Invert sugar is used in production of jam 20

2. Properties of Starch 1. v Flavour Starch (a white powder) is not sweet in flavour 2. Solubility v Starch is insoluble in cold water 3. v Hygroscopic This property relates to how starch absorbs moisture from the air e. g. biscuits soften if they are not kept air tight 21

2. Properties of Starch 4. Dextrinisation v Dextrins are shorter chains of starch v On heating, dextrins form longer chains & become brown-coloured substances called pyrodextrins v An example of dextrinisation is toasting bread 22

2. Properties of Starch cont…. . 5. Gelatinisation is based on the principal that when starch is heated in the presence of water, starch grains swell, burst & absorb the liquid, resulting in the thickening of the liquid v As the temperature rises, this mixture becomes even more viscous, forming a sol (A sol contains particles that do not fully dissolve but are evenly dispersed throughout the liquid) v On cooling, this becomes a gel v An example of this is using flour to thicken soups and sauces 23

2. Properties of Starch 6. Hydrolysis v Hydrolysis is a chemical breakdown of a molecule by adding water to produce smaller molecules v Disaccharides become monosaccharides partly due to hydrolysis 24

Properties of Non-Starch Polysaccharides 1. v v Cellulose Can absorb large amounts of water Cannot be digested, however adds bulk to the diet (gives a feeling of fullness) Aids the removal of waste from the body Is insoluble in water 25

3. Properties of Non-Starch Polysaccharides Pectin v Pectin is a polysaccharide found in fruit and vegetables v It is involved in setting jams & jellies v The following shows the pectin change in the ripening of fruit: 2. Under-Ripe to ripe to Over-Ripe Protopectin to Pectic Acid (pectose) v For pectin extraction : 1. Use fruit rich in pectin, e. g. Blackcurrants & Apples 2. Heat needs to be applied to the fruit 3. Add an acid, e. g. Lemon juice changes protopectin to pectin 26

3. Properties of Non-Starch Polysaccharides 3. Gel Formation When pectin is heated in the presence of acid and sugar, water becomes trapped v The long chains of polysaccharides cool to form a gel v An example of this is in making jam v 27

Effects of Heat on Carbohydrates Dry Heat § § § Carbohydrate foods browns due to the presence of dextrins, e. g. Toast Sugar caramelises, e. g. Caramel slices Maillard reaction occurs because of the interaction between sugar & amino acids, e. g. roast potatoes Moist Heat § § Cellulose softens, e. g. cooked vegetables Starch grains swell, burst & absorb liquid, e. g. flour used to thicken sauces Pectin is extracted by heating fruit in water with sugar & acid, e. g. jam making Sugar dissolves in warm liquid, e. g. making syrups 28

Culinary uses of sugar Sweetener - desserts Preservative - jam Caramelisation – caramel custard Fermentation – yeast bread Gel formation – sugar combines with pectin to form gel – jam making. Colour – a sugar solution prevents discolouration of cut fruit. 29

Culinary uses of starch Thickener – sauces, soups, stews. Hygroscopic – absorbs moisture to increase shelf life of cakes, keeps baking powder dry. Dextrinisation – browning e. g. toast. 30

Culinary uses of non -starch polysaccharides Gel formation – jam pectin forms gel with acid and sugar. Cellulose absorbs moisture and gives feeling of fullness. Cellulose adds texture e. g. breakfast cereals 31

Digestion of Carbohydrates Mouth: Physically broken by teeth. Salivary Amylase breaks Starch into Maltose. Stomach: Physically churned up. Intestine: Pancreatic juice (Pancreas) Amylase breaks Starch into Maltose. Intestinal Juice: (Ileum) Maltase breaks Maltose into Glucose. Sucrase breaks Sucrose into Glucose & Fructose. Lactase breaks Lactose into Glucose and Galactose. 32

Absorption: Monosaccharides are absorbed through the villi of the small intestine into the blood stream and are carried to the liver in the portal vein. 33

Utilasation of Carbohydrates Monosaccharides can be oxidised to produce energy (cellular respiration). q Some monosaccharides are changed to glycogen and stored in liver and muscle as an energy reserve. q Excess carbohydrate is changed to body fat and stored in the adipose tissue under the skin. q Vitamin B 1, B 2 and Pyrodoxine are needed to metabolise carbohydrates. q 34
- Slides: 34