Carbohydrate MetabolismI Glycolysis TCA Cycle Hexose Monophosphate Shunt

Carbohydrate Metabolism–I Glycolysis, TCA Cycle & Hexose Monophosphate Shunt Pathway

LEARNING OBJECTIVES • Describe the steps of Glycolysis. Add a note on its Energetics • Enumerate the steps of TCA cycle with inhibitors and energetics • Describe the HMP shunt pathway and state its significance

Metabolism of Carbohydrate 1. Energy (ATP) producing pathways/reactions (in all cells) • Glycolysis • Oxidation of pyruvate • Tricarboxylic acid cycle (TCA cycle) 2. Production of NADPH and ribose • Hexose monophosphate(HMP) shunt pathway

3. Pathways involved in blood glucose homeostasis • Gluconeogenesis • Metabolism of glycogen- glycogenesis & glycogenolysis 4. Synthesis of uronic acids • Uronic acid pathway 5. Metabolism of other hexoses • Metabolism of fructose and galactose

Clinical Importance/Disorders of Carbohydrate Metabolism • Obesity • Diabetes mellitus • Glycogen storage diseases • Galactosemia • Fructose intolerance and fructosuria

Glycolysis • Definition • Tissue location • Intracellular location • Types • End product • Reactions (Pathway) • Energetics • Regulation • Functions • Inhibitors of glycolysis • Clinical significance

Glycolysis Synonym: Embden-Meyerhof pathway • Definition Glycolysis is an oxidative pathway for the catabolism of glucose to pyruvate or lactate accompanied by the formation of ATP. Tissue location All the cells of the body, RBC

Intracellular location: Cytosol Types: Aerobic in the presence of oxygen Anaerobic lack of oxygen End product: Pyruvate, during aerobic conditions Lactate/lactic acid, RBC under anaerobic conditions in skeletal muscles during prolonged contraction

Reaction Sequence/Pathway It can be divided in to 2 phases I. Energy consuming/investment phase glucose → glyceraldehyde-3 -phosphate II. Energy generation/payoff phase – glyceraldehyde-3 -phosphate → pyruvate
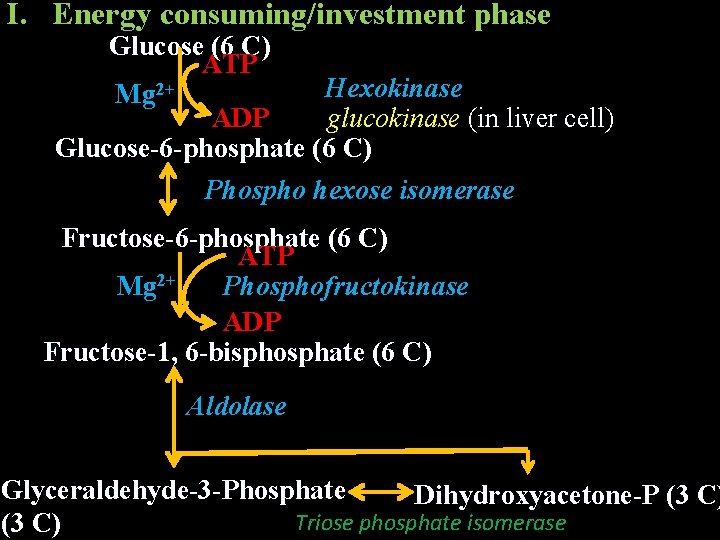
I. Energy consuming/investment phase Glucose (6 C) ATP Hexokinase Mg 2+ ADP glucokinase (in liver cell) Glucose-6 -phosphate (6 C) Phospho hexose isomerase Fructose-6 -phosphate (6 C) ATP Mg 2+ Phosphofructokinase ADP Fructose-1, 6 -bisphosphate (6 C) Aldolase Glyceraldehyde-3 -Phosphate Dihydroxyacetone-P (3 C) Triose phosphate isomerase (3 C)

II. Energy generation/payoff phase – Glyceraldehyde-3 -Phosphate (3 C) Pi NAD+ Glyceraldehyde-3 - phosphate dehydrogenase NADH + H+ 1, 3 -bisphsophoglycerate (3 C) ADP Mg 2+ 1, 3 -bisphsopho-glycerate kinase Substrate level phosphorylation ATP 3 -phosphoglycerate (3 C) Phosphoglycerate mutase 2 -phosphoglycerate (3 C) Mg 2+ H 2 O Enolase Phosphoenol pyruvate (3 C)
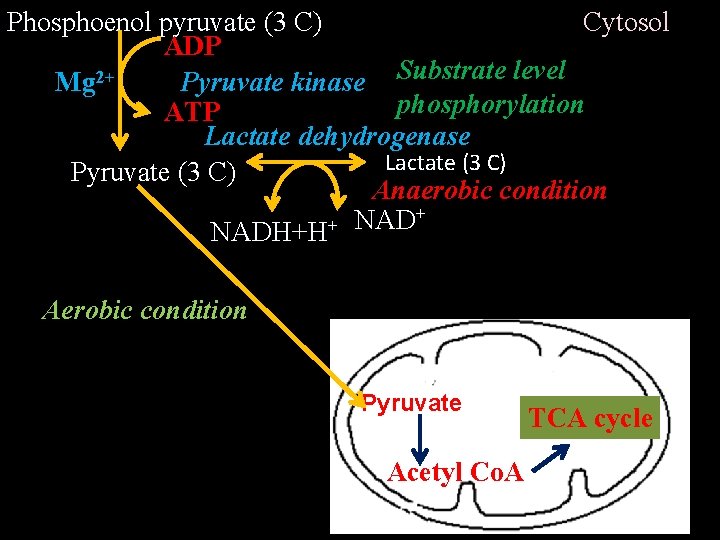
Cytosol Phosphoenol pyruvate (3 C) ADP Pyruvate kinase Substrate level Mg 2+ phosphorylation ATP Lactate dehydrogenase Lactate (3 C) Pyruvate (3 C) Anaerobic condition + NADH+H Aerobic condition Pyruvate Acetyl Co. A TCA cycle

glucose hexokinase/ glucokinase glucose-6 - phosphate Cytoplasm Phosphohexose isomerase Fructose -6 - phosphate Phosphofructo kinase Fructose-1, 6 -bisphosphate Aldolase Glyceraldehyde – 3 - phosphate Glyceraldehyde-3 - phosphate dehydrogenase 1, 3 -bisphoglycerate Dihydroxy acetone 1, 3 -bisphsopho-glycerate kinase phosphate 3 -phosphoglycerate Phosphoglycerate mutase 2 -phosphoglycerate Enolase Phosphoenolpyruvate Pyruvate kinase Lactate dehydrogenase Lactate Pyruvate

Energetics of Aerobic Glycolysis Energy produced Reaction catalysed by Glyceraldehyde -3 -phosphate dehydrogenase Phosphoglycerate kinase Pyruvate kinase Method of ATP per formation mol of glucose Respiratory chain oxidation of 2 NADH Substrate level phosphorylation 5 2 2 Total = 9

Energy utilised Reaction catalysed by ATP per mol of glucose utilised Hexokinase Phosphofructokinase 1 1 Total =2 Net energy produced in Aerobic Glycolysis = energy produced – energy utilized = 9 - 2 = 7 ATP

Energetics of Anaerobic Glycolysis Energy produced Reaction catalysed by Method of ATP per formation mol of glucose Phosphoglycerate Substrate level kinase phosphorylation Pyruvate kinase Substrate level phosphorylation 2 2 Total= 4

Energy utilised Reaction catalysed by ATP per mol of glucose utilised Hexokinase Phosphofructokinase 1 1 Total= 2 Net energy produced in Anaerobic Glycolysis = energy produced – energy utilized = 4 - 2 = 2 ATP

Regulation of Glycolysis is controlled by the energy state of the cell and by hormones insulin and glucagon. The 3 key glycolytic enzymes hexokinase, Phosphofructo kinase, Pyruvate kinase are regulated.
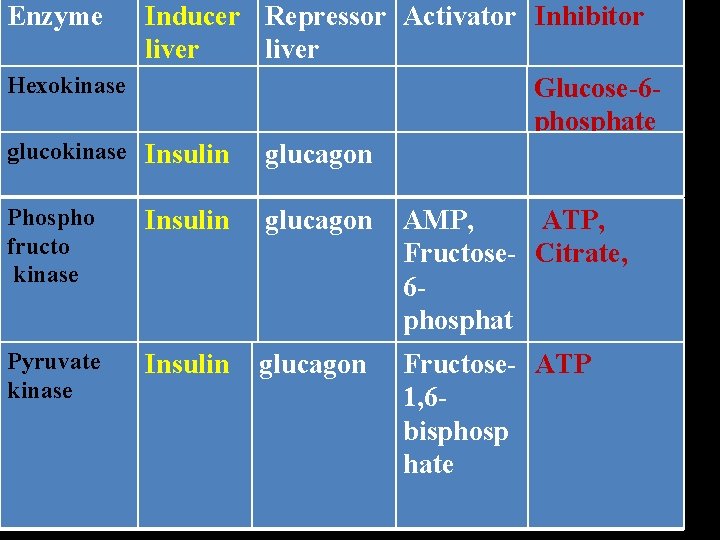
Enzyme Inducer Repressor Activator Inhibitor liver Hexokinase Glucose-6 phosphate glucokinase Insulin glucagon Phospho fructo kinase Insulin glucagon Pyruvate kinase Insulin glucagon AMP, ATP, Fructose- Citrate, 6 phosphat e. Fructose- ATP 1, 6 bisphosp hate

Functions/Significance of Glycolytic Pathway 1. Glycolysis is a major pathway for ATP synthesis a)In all the cells of the body. b)In tissues lacking mitochondria, eg. RBC. c)During strenuous exercise, anaerobic glycolysis is the source of energy for muscles. 2. 2, 3 - bisphoglycerate formed from 1, 3 - bisphoglycerate in RBC help to release oxygen from Hb in the peripheral tissues.

3. Glycolysis serves as pathway for catabolism of fructose, galactose and some pentoses derived from the diet 4. It provides carbon skeletons for the synthesis of certain non-essential amino acids 5. Most of the reactions of glycolysis are reversible used for gluconeogenesis in liver and kidneys.

Rapaport Luebering Cycle in RBC In this pathway the reaction catalyzed by 1, 3 bisphoglycerate kinase is bypassed involves no net yield of ATP. It takes place in RBC.
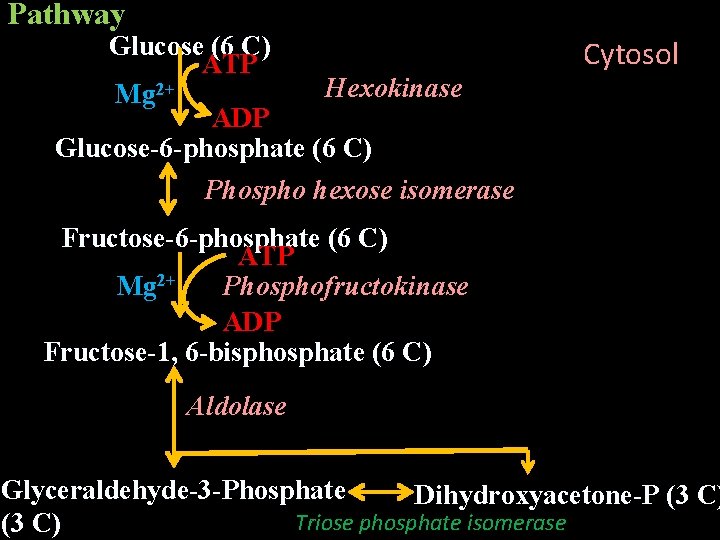
Pathway Glucose (6 C) ATP Hexokinase Mg 2+ ADP Glucose-6 -phosphate (6 C) Phospho hexose isomerase Cytosol Fructose-6 -phosphate (6 C) ATP Mg 2+ Phosphofructokinase ADP Fructose-1, 6 -bisphosphate (6 C) Aldolase Glyceraldehyde-3 -Phosphate Dihydroxyacetone-P (3 C) Triose phosphate isomerase (3 C)

Glyceraldehyde-3 -Phosphate (3 C) Pi NAD+ Glyceraldehyde-3 - phosphate dehydrogenase NADH + H+ 1, 3 -bisphsophoglycerate (3 C) Bisphoglycerate mutase 3 -phsophoglycerate (3 C) 2, 3 Bisphoglycerate (2, 3 BPG) Phosphatase Pi lactate
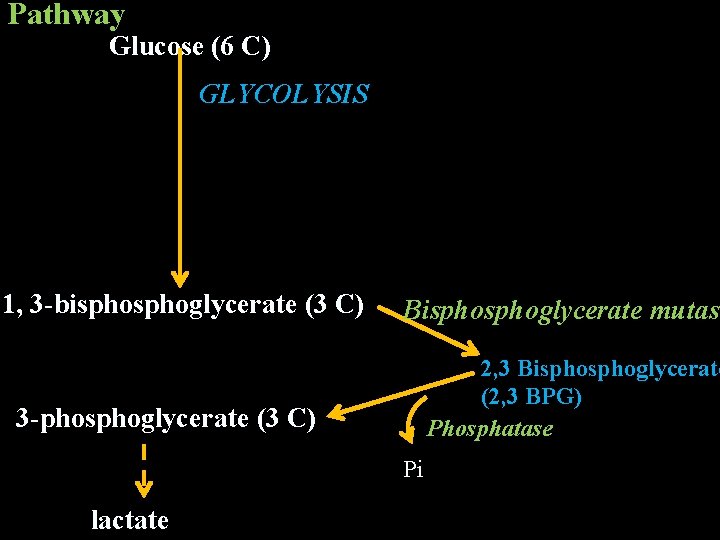
Pathway Glucose (6 C) GLYCOLYSIS 1, 3 -bisphoglycerate (3 C) 3 -phosphoglycerate (3 C) Bisphoglycerate mutase 2, 3 Bisphoglycerate (2, 3 BPG) Phosphatase Pi lactate

Function 2, 3 -BPG binds to Hb decreases its affinity for oxygen leading to release of oxygen in the peripheral tissues.

Clinical Significance of Glycolysis 1. Hemolytic anemia Deficiency of pyruvate kinase leads to decreased synthesis of ATP in RBCs in turn leading to instability of membrane and hemolysis and hence, also anemia 2. Muscle cramp seen during excessive physical exercise due to anaerobic glycolysis leading to accumulation of lactic acid in muscles.

Thank you

Metabolic Fate of Pyruvate • Pyruvate generated in the cytoplasm is transported to the mitochondria by pyruvate transporter. • Pyruvate is converted to acetyl Co. A by oxidative decarboxylation by pyruvate dehydrogenase complex. • This is an irreversible reaction. • Pyruvate dehydrogenase complex is a multi-enzyme complex containing 3 enzymes activities. It also contains 5 coenzymes. thiamine pyrophosphate (TPP), lipoamide, coenzyme A (Co. ASH), FAD and NAD+
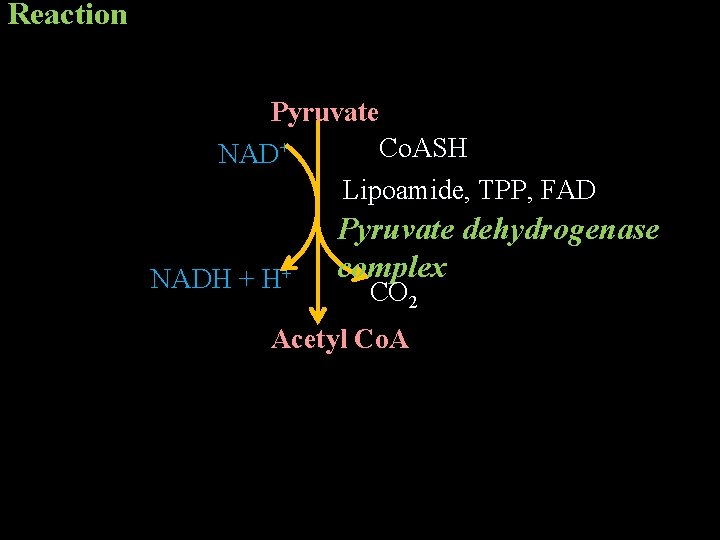
Reaction Pyruvate Co. ASH NAD+ Lipoamide, TPP, FAD NADH + H+ Pyruvate dehydrogenase complex CO 2 Acetyl Co. A

Energetics 1 molecule of glucose produces 2 molecules of pyruvate and hence, 5 molecules of ATP at this step.
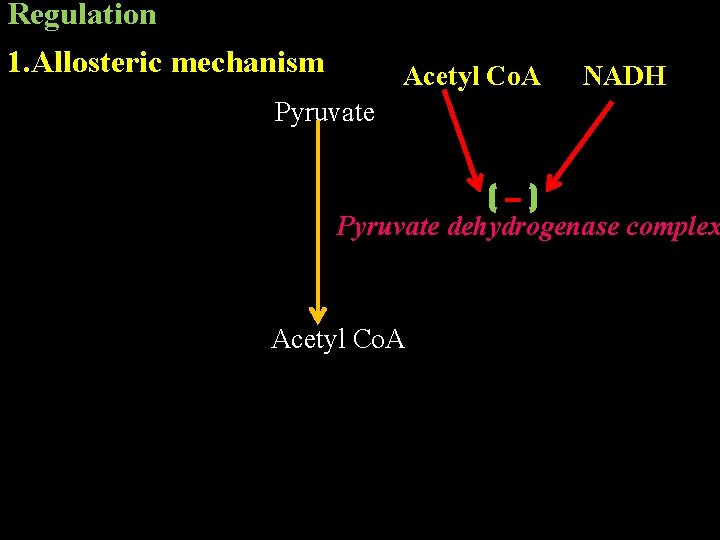
Regulation 1. Allosteric mechanism Acetyl Co. A NADH Pyruvate dehydrogenase complex Acetyl Co. A

2. Covalent modification Pyruvate dehydrogenase complex is regulated by covalent modification. The enzyme active in dephosphorylated form

Clinical Aspects Lactic acidosis: Pyruvate is converted to lactate and its accumulation in the blood leads to lactic acidosis. Causes : Inherited deficiency of pyruvate dehydrogenase Arsenite, and mercuric ions, inhibit the enzyme

Tricarboxylic Acid Cycle (TCA Cycle) • Contents: • • • Definition Tissue location Intracellular location Reactions (Pathway) Energetics Regulation Functions Inhibitors Clinical significance

Tricarboxylic Acid Cycle (TCA Cycle) Synonym: Krebs cycle or Citric acid cycle • Definition TCA cycle is the final common pathway for oxidation of carbohydrate (glucose), fat (fatty acid) and protein (amino acid) for production of energy. Tissue location All tissues Intracellular location: Mitochondrial matrix
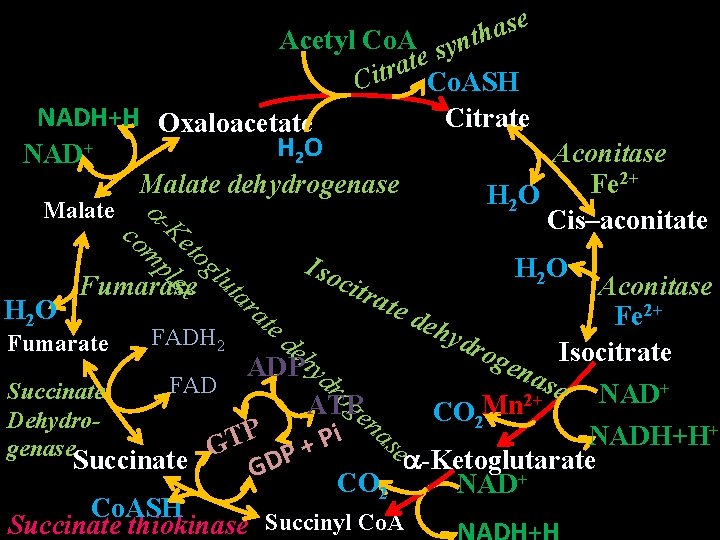
e s a h t yn Acetyl Co. A s e t a r Cit Co. ASH NADH+H Oxaloacetate Citrate H 2 O Aconitase NAD+ 2+ Malate dehydrogenase Fe H 2 O Malate Cis–aconitate co -Ke m tog pl l Iso H 2 O u e c x ta Fumarase Aconitase itra ra te d 2+ H 2 O Fe te e hyd FADH 2 de Fumarate rog h ena Isocitrate ADP yd se NAD+ ro FAD Succinate 2+ ge Mn ATP CO 2 Dehydron + as P i NADH+H P T e + genase G P Succinate -Ketoglutarate GD CO 2 NAD+ Co. ASH Succinate thiokinase Succinyl Co. A

Energetics of TCA Cycle Reaction No. of Reduced Method of catalyzed by Coenzyme ATP production Oxidative phosphorylation Oxidative -Ketoglutarate 1 NADH phosphorylation DH Substrate level – – – Succinate thiokinase phosphorylation Oxidative 1 FADH 2 Succinate phosphorylation DH 1 NADH Oxidative Malate DH phosphorylation Isocitrate DH 1 NADH No. of ATP molecules formed 2. 5 1 1. 5 2. 5 Total 10

Regulation of of TCA Cycle • The cellular needs of ATP. When energy charge of the cell is low, indicated by high level of NAD+ and FAD, cycle operates at a faster rate. • Sites for regulation • Citrate synthase • Isocitrate dehydrogenase • -Ketoglutarate dehydrogenase
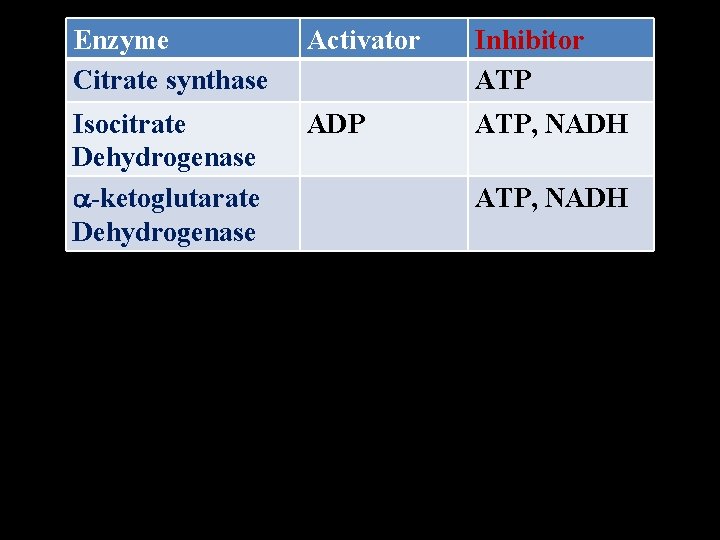
Enzyme Citrate synthase Activator Inhibitor ATP Isocitrate Dehydrogenase -ketoglutarate Dehydrogenase ADP ATP, NADH

Inhibitors (synthetic) of TCA cycle Enzyme Inhibitor mechanism Citrate Fluroacetate Suicide synthase inhibition Succinate Malonate Competitive dehydrogen inhibition ase

Amphibolic Role of TCA Cycle TCA cycle has a dual role. 1. Catabolic Oxidation of acetyl-Co. A produced from metabolism of carbohydrates, fats and proteins to CO 2, H 2 O and energy. 2. Anabolic Intermediates of TCA are utilised for the synthesis of various compounds like glucose, non –essential amino acids, fatty acid, heme.
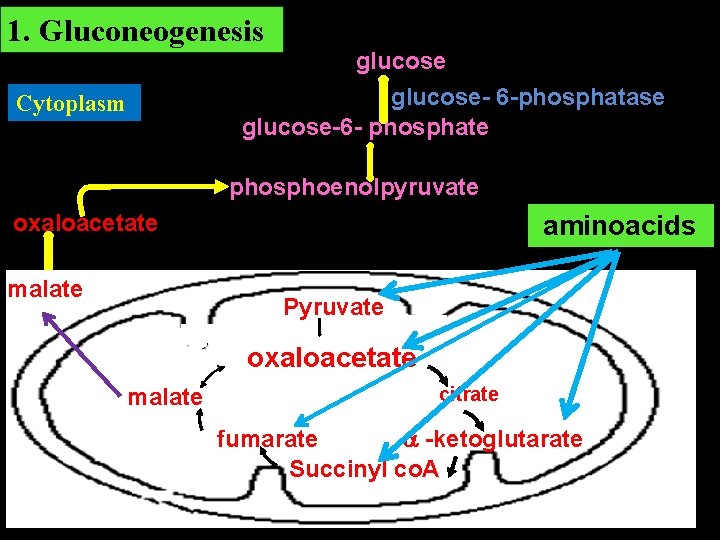
1. Gluconeogenesis Cytoplasm glucose- 6 -phosphatase glucose-6 - phosphate phosphoenolpyruvate oxaloacetate malate aminoacids Pyruvate oxaloacetate malate citrate fumarate -ketoglutarate Succinyl co. A
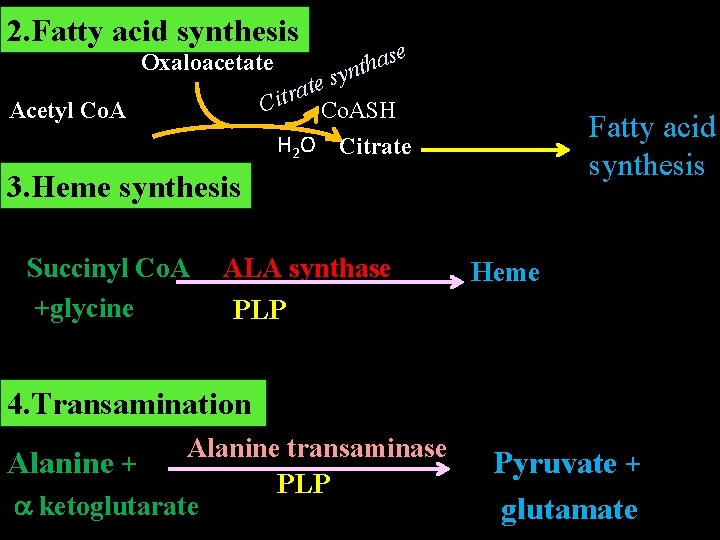
2. Fatty acid synthesis Oxaloacetate se a h t syn ate r t i C Co. ASH Acetyl Co. A Fatty acid synthesis H 2 O Citrate 3. Heme synthesis Succinyl Co. A ALA synthase +glycine PLP Heme 4. Transamination Alanine transaminase Alanine + PLP ketoglutarate Pyruvate + glutamate

Anaplerosis or Anaplerotic Reactions • Definition Metabolic reactions, which replenish the intermediates of TCA cycle, are called anaplerotic reactions or anaplerosis. • Reactions 1. Pyruvate+CO 2 Pyruvate carboxylase Biotin ATP ADP +Pi Oxaloacetate
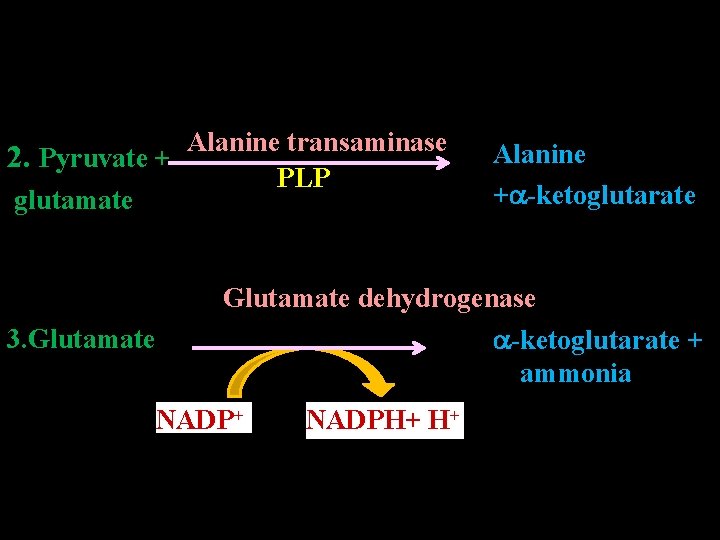
Alanine transaminase 2. Pyruvate + PLP glutamate Alanine + -ketoglutarate Glutamate 3. Glutamate dehydrogenase -ketoglutarate + ammonia NADP+ NADPH+ H+

Thank you

Hexose Monophosphate Shunt Pathway (HMP Shunt Pathway) • • • Definition Tissue location Intracellular location Regulation Functions Clinical significance

Hexose Monophosphate Shunt Pathway (HMP Shunt Pathway) Synonyms: Pentose phosphate pathway; Direct oxidative pathway • Definition This is a pathway for oxidation of glucose alternative to glycolysis, for production of NADPH and ribose (ribose-5 -P). Tissue location RBC, adrenal cortex, liver, testis, thyroid, adipose tissue, etc.

Intracellular location: Cytosol Reactions/Pathway: HMP shunt pathway has two phases. 1. Oxidative phase (irreversible) 2. Non-oxidative phase (reversible
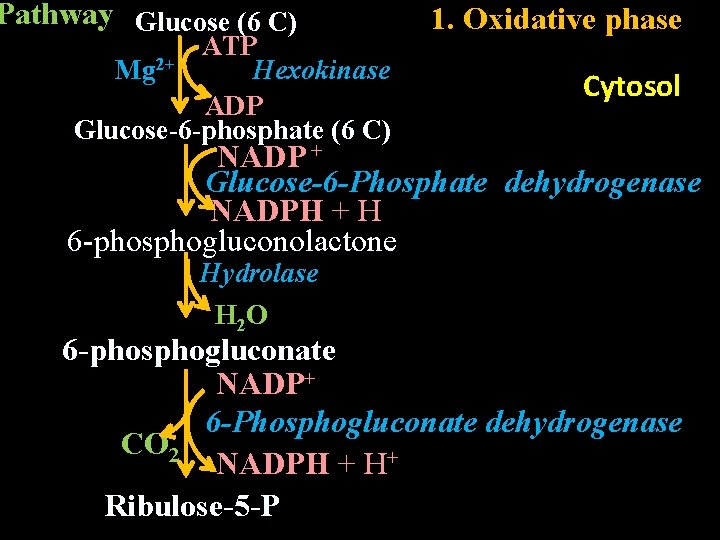
Pathway Glucose (6 C) ATP Mg 2+ Hexokinase ADP Glucose-6 -phosphate (6 C) 1. Oxidative phase Cytosol NADP + Glucose-6 -Phosphate dehydrogenase NADPH + H 6 -phosphogluconolactone Hydrolase H 2 O 6 -phosphogluconate NADP+ 6 -Phosphogluconate dehydrogenase CO 2 NADPH + H+ Ribulose-5 -P
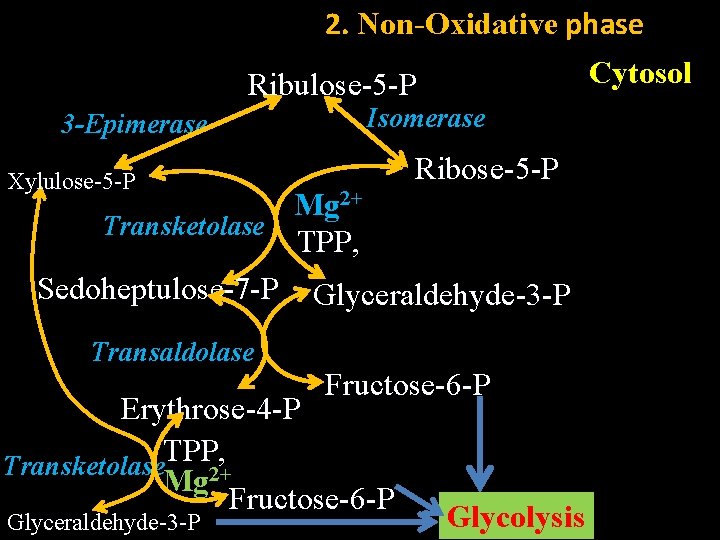
2. Non-Oxidative phase Cytosol Ribulose-5 -P 3 -Epimerase Isomerase Ribose-5 -P Xylulose-5 -P Mg 2+ Transketolase TPP, Sedoheptulose-7 -P Glyceraldehyde-3 -P Transaldolase Fructose-6 -P Erythrose-4 -P TPP, Transketolase 2+ Mg Fructose-6 -P Glyceraldehyde-3 -P Glycolysis

Regulation of HMP Shunt Pathway Glucose-6 -phosphate dehydrogenase is the ratelimiting enzyme of this pathway. This enzyme is inhibited by NADPH and induced by insulin.
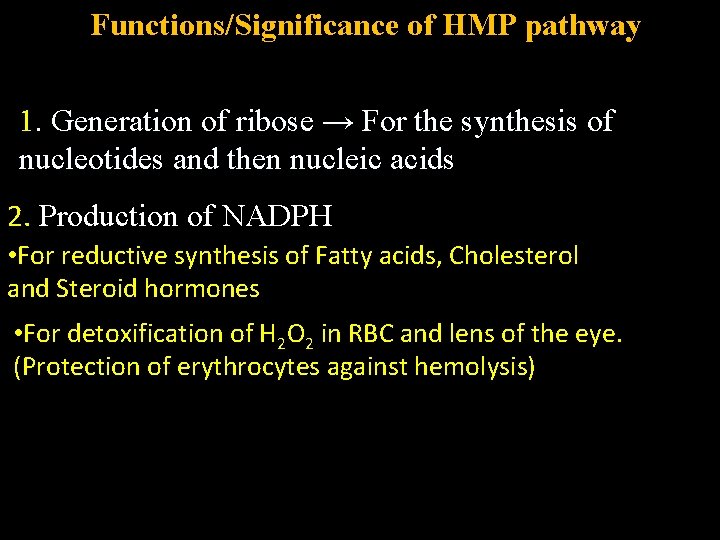
Functions/Significance of HMP pathway 1. Generation of ribose → For the synthesis of nucleotides and then nucleic acids 2. Production of NADPH • For reductive synthesis of Fatty acids, Cholesterol and Steroid hormones • For detoxification of H 2 O 2 in RBC and lens of the eye. (Protection of erythrocytes against hemolysis)

Clinical Significance of HMP pathway Glucose-6 -phophate dehydrogenase (GPD) deficiency There is decreased generation of NADPH Manifestations: jaundice and severe anemia.

Thank you

Anabolic Pathways: Intermediates of TCA cycle involved Gluconeogenesis Oxaloacetate Fatty acid synthesis Citrate Heme synthesis Succinyl Co. A Synthesis of Glutamate by -Ketoglutarate, transamination Synthesis of Aspartate by transamination Oxaloacetate Catabolic Pathways: Oxidation of acetyl-Co. A produced from metabolism of carbohydrates, fats and proteins to CO 2, H 2 O and energy




















- Slides: 77