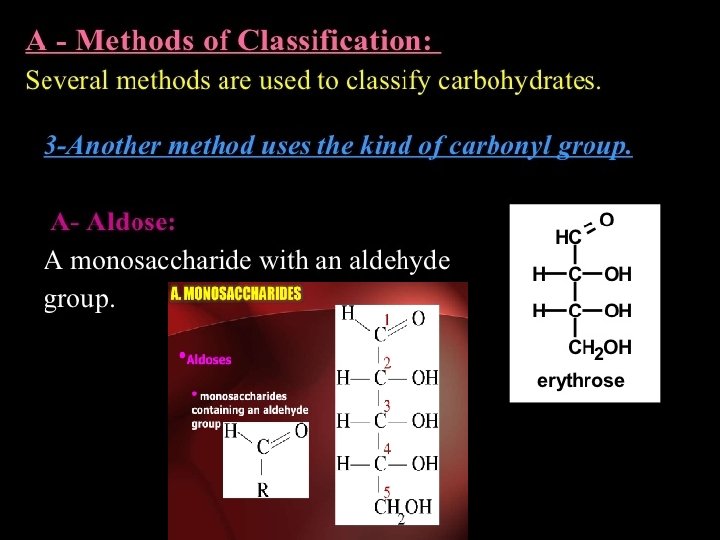
Carbohydrate Chemistry Structural representation of sugars Monosaccharides can

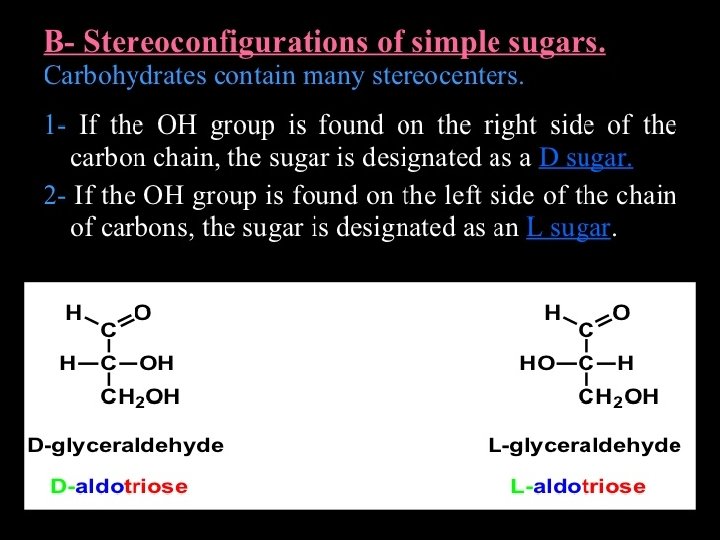
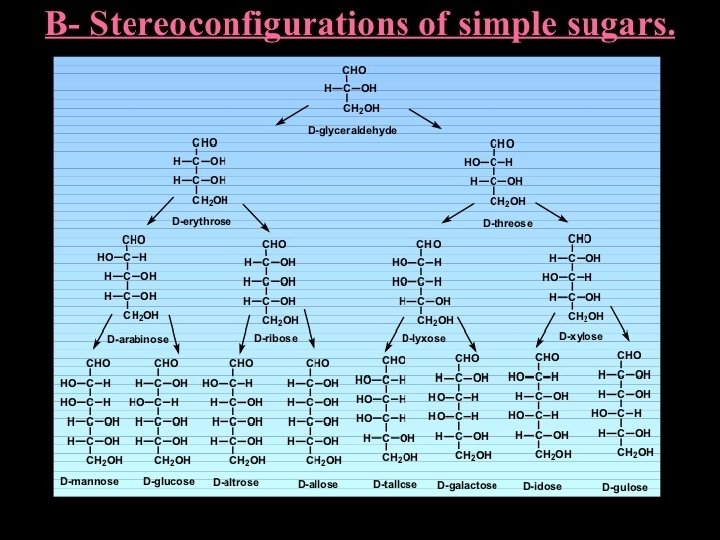
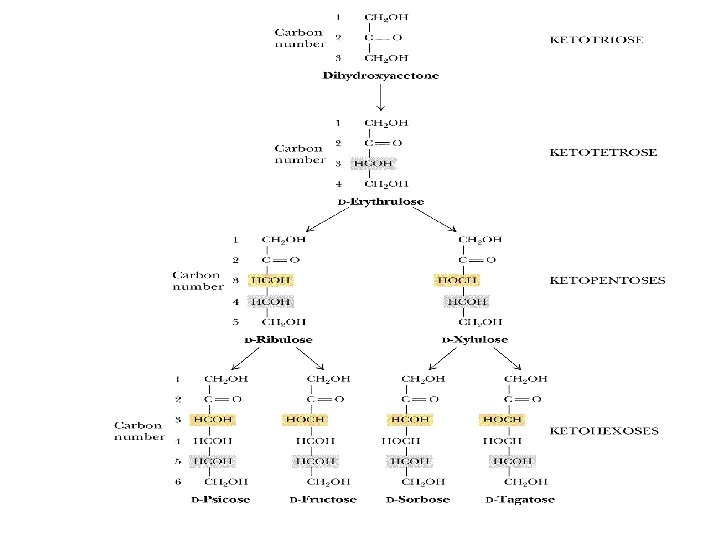
• Carbohydrate Chemistry
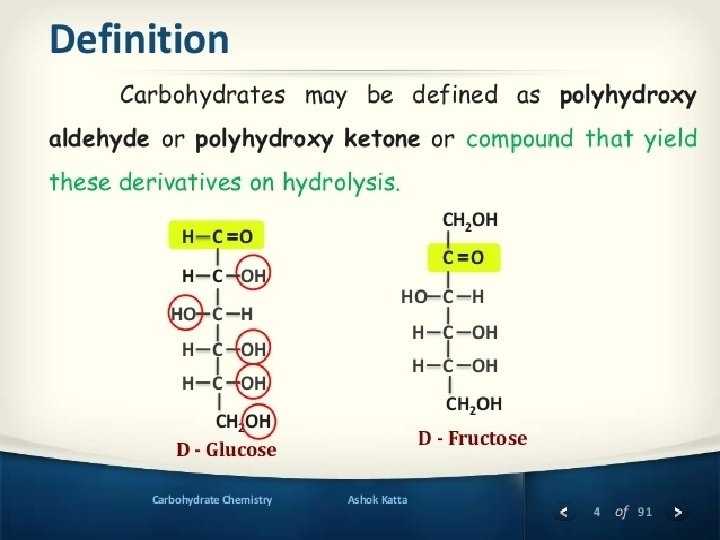




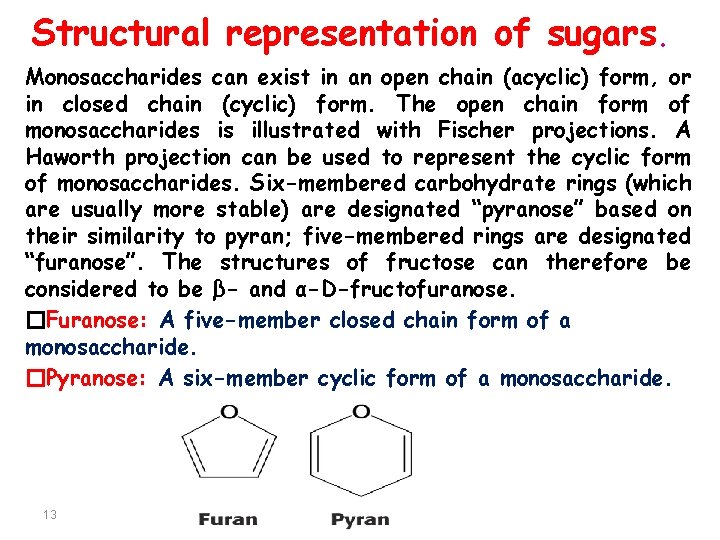
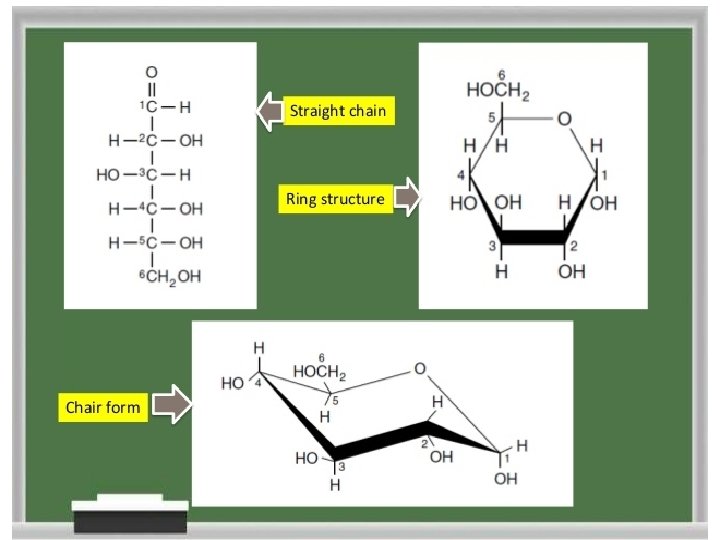
Structural representation of sugars. Monosaccharides can exist in an open chain (acyclic) form, or in closed chain (cyclic) form. The open chain form of monosaccharides is illustrated with Fischer projections. A Haworth projection can be used to represent the cyclic form of monosaccharides. Six-membered carbohydrate rings (which are usually more stable) are designated “pyranose” based on their similarity to pyran; five-membered rings are designated “furanose”. The structures of fructose can therefore be considered to be β- and α-D-fructofuranose. �Furanose: A five-member closed chain form of a monosaccharide. �Pyranose: A six-member cyclic form of a monosaccharide. 13

Structural representation of sugars • Fisher projection: straight chain representation. • Haworth projection: simple ring in perspective • Conformational representation: chair and boat configurations
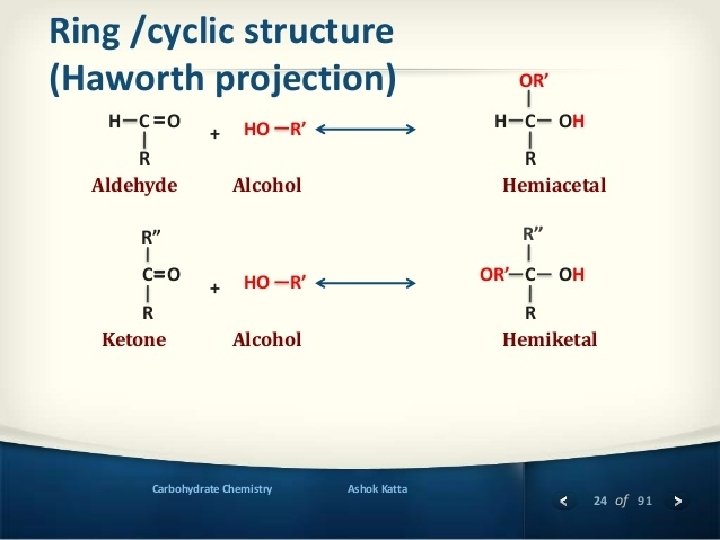
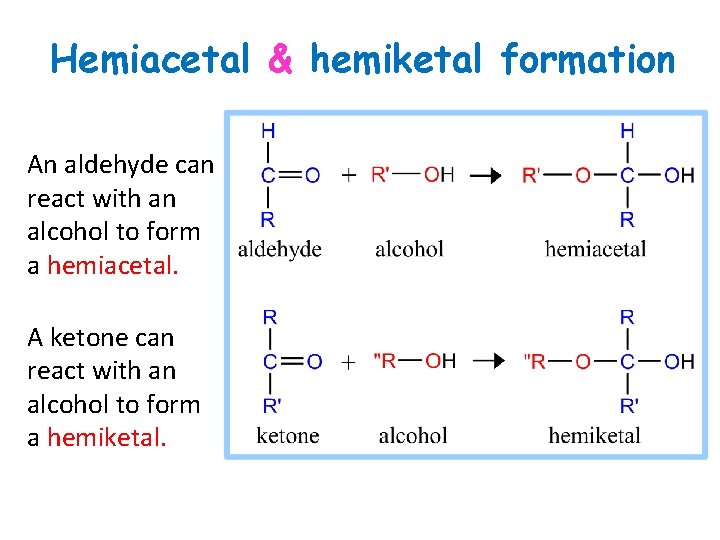
Hemiacetal & hemiketal formation An aldehyde can react with an alcohol to form a hemiacetal. A ketone can react with an alcohol to form a hemiketal.
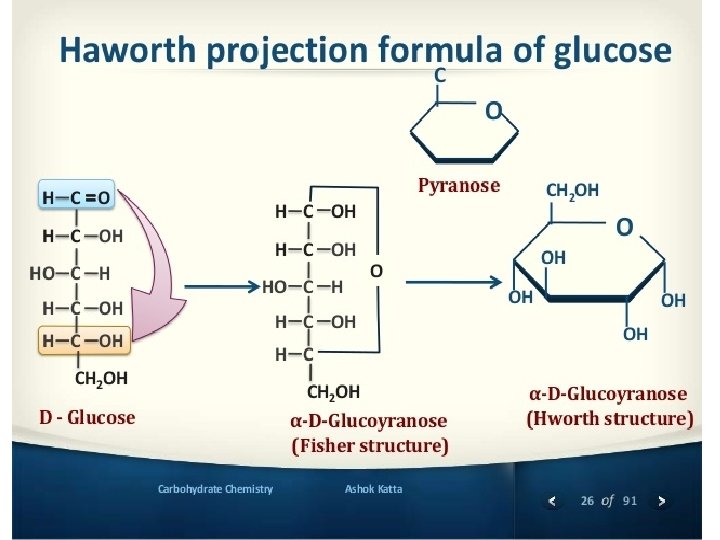
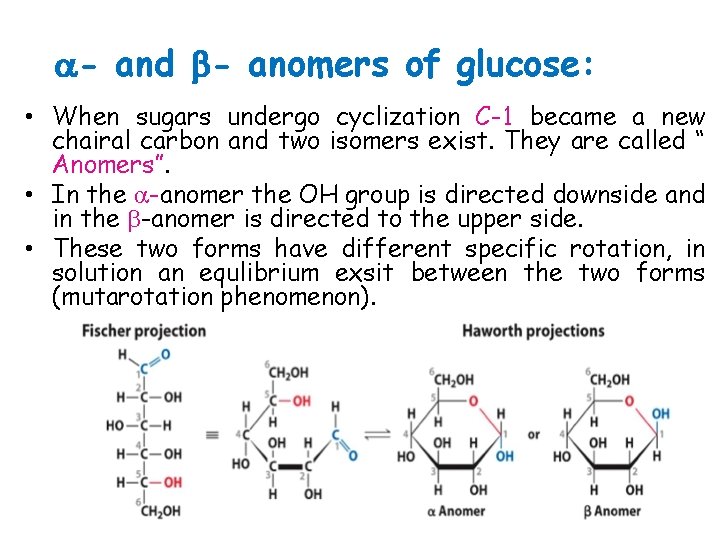
- and - anomers of glucose: • When sugars undergo cyclization C-1 became a new chairal carbon and two isomers exist. They are called “ Anomers”. • In the -anomer the OH group is directed downside and in the -anomer is directed to the upper side. • These two forms have different specific rotation, in solution an equlibrium exsit between the two forms (mutarotation phenomenon).
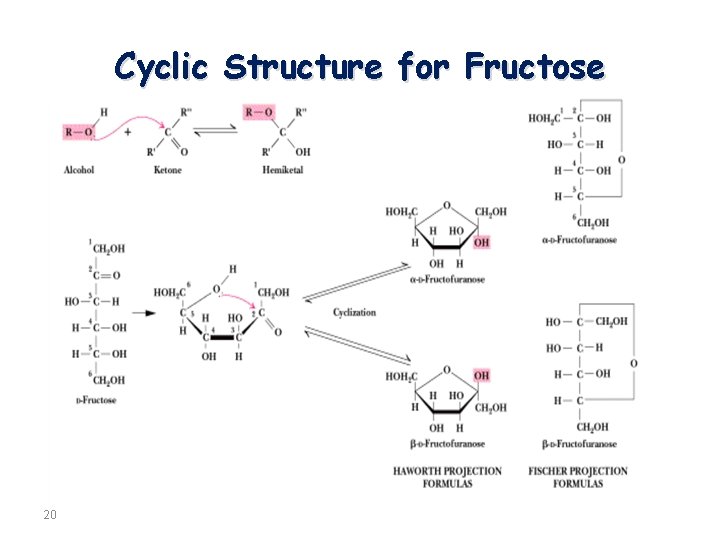
Cyclic Structure for Fructose 20
- Slides: 20