Capillary electrophoresis Instrumentation and control of separation Sayed






























- Slides: 34

Capillary electrophoresis Instrumentation and control of separation Sayed M Derayea, Ph. D Minia University – Faculty of Pharmacy Department of Analytical Chemistry Sayed_derayea@yahoo. com 1

Variables affecting EOF

Variables affecting EOF n n EOF can be controlled using different variables which in the same time produce some of the complexity of CE relative to HPLC. These variables include: 1. Buffer p. H and strength 2. Electric field 3. Temperature 4. Organic Modifier 5. Surfactant 6. Covalent wall coating

Variables affecting EOF Buffer p. H n Buffer p. H is the most convenient method for controlling EOF but has to be balanced against effects on the charge on the analyte as EOF increases with p. H. Buffer strength: n EOF decreases with increasing buffer strength however: (i) Increased ionic strength means increased electric current flow through the capillary, which can cause heating. (ii) At low ionic strength more sample absorption onto the capillary walls occurs. (iii) Low buffer concentrations reduce sample stacking following injection.

Variables affecting EOF 2. Electric field n Increased electric field increases EOF. Lowering the applied electric field may reduce separation efficiency and raising the field strength may cause heating. 3. Temperature n Increased temperature decreases viscosity and thus increases flow this factor is easy to control. 4. Organic Modifier n Changes potential at capillary wall » the dielectric constant of the running buffer and the viscosity. Usually decreases EOF. n Complex effects - can be useful but best determined experimentally

Variables affecting EOF 5. Surfactant n Surfactant absorbs onto the surface of the capillary wall (i) Cationic surfactants n n have a high affinity for the silanol groups and thus block access by the smaller cations in solution reducing EOF At high concentration they form a double layer giving the capillary wall an effective positive charge and causing EOF to reverse flow towards the anode (ii) Anionic surfactants reduce the access of the smaller ions in the running buffer to the positive potential at the wall thus increasing the zeta potential and thus EOF

Variables affecting EOF 6. Covalent wall coating n The coating of the wall can raise or lower EOF depending on the coating (i) Neutral coatings reduce negative charge of the capillary wall thus reducing EOF but (ii) ionic coatings will have marked effects on EOF.

Capillary electrophoresis Instrumentation

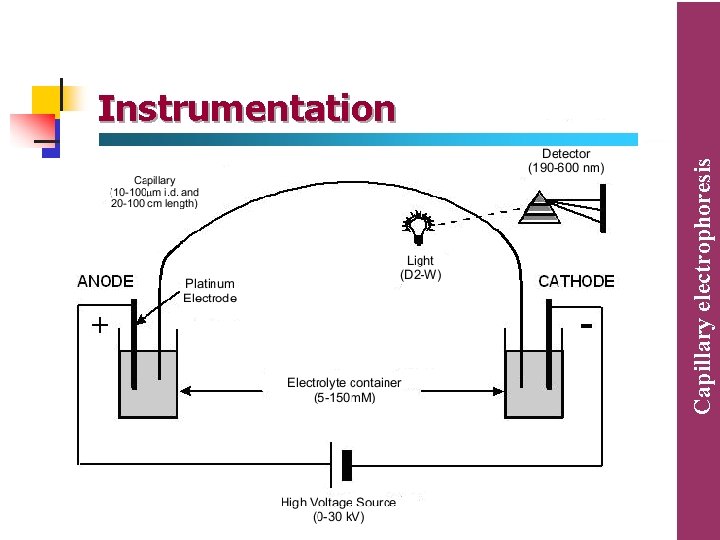
The fundamentals of CE system (i) Injection is commonly automated and is usually accomplished by pressuring the vial containing the sample with air. (ii) Having loaded the sample the capillary is switched to a vial containing running buffer. The flow rate of the running buffer through the capillary is in the low nanolitres/min range. (iii) The capillary are like those used in capillary gas chromatography with polyamide coating on the outside. The length of the capillary is about 100 cm with internal diameter of 0. 025 – 0. 05 mm. they are wound together to be easily place inside the instrument. (iv) At the detector end the capillary has a window burnt into it so that it is transparent to the radiation used for detection of the analyte. (v) The most commonly used detector is a diode array or rapid scanning UV detector although fluorometric, conductimetric and mass spectrometric detectors are available.

• High-voltage DC (direct current) power supplies, providing voltages up to +30 k. V, are used in capillary electrophoresis. • The operating current is often kept below 100 µA to reduce joule heating effects. • Current as high as 250 µA are possible but not desirable since they may lead to unstable, irreproducible operating conditions. Capillary electrophoresis Power supply

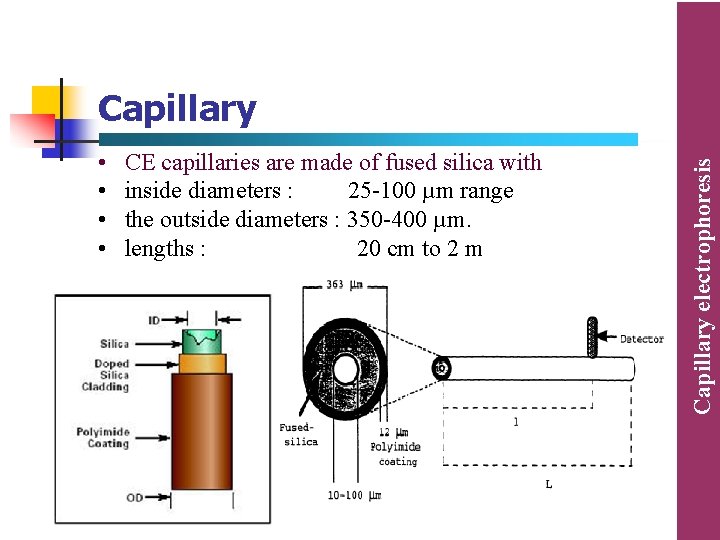
• • CE capillaries are made of fused silica with inside diameters : 25 -100 µm range the outside diameters : 350 -400 µm. lengths : 20 cm to 2 m Capillary electrophoresis Capillary

• The relatively thick capillary wall when compared to its inner diameter functions as an effective heat sink. It is capable of dissipating the heat that is generated in the capillary as a result of Joule heating, thus allowing the use of high electric fields. . • To make thin silica capillaries more durable and flexible, they are typically coated with a thin layer of polyimide coating. • The intended use determines whether to choose an uncoated or coated capillary for the analysis. • Uncoated fused silica capillaries are the most common mainly, because the internal surface is reproducible making the separation predictable. Capillary electrophoresis Capillary

Ø To make an injection, the inlet reservoir vial containing the running buffer is removed and the sample vial is put in its place. Ø A small amount of the sample is injected into the end of the capillary either electrokinetically or hydrodynamically (by vacuum or pressure difference), the later being the most commonly used. Ø The inlet vial is then returned to its place and the high voltage is turned on. Capillary electrophoresis Sample injection

1. Hydrodynamic sample injection Ø Hydrodynamic injection is commonly induced by applying Ø pressure or Ø vacuum. Ø however also includes siphoning. Ø Hydrodynamic injection produce reproducible sample injections with good relative standard deviations (%RSD<2%) and can provide a small injection volume and incremental increases in volumeis, Capillary electrophoresis Sample injection

2. Electrokinetic sample injection Ø In electrokinetic sample injection, both the capillary end and the electrode are removed from the buffer vial and placed into the sample vial, then Ø a potential is applied across the two electrode for a few seconds, then Ø the electrode and the capillary end are returned to their places Capillary electrophoresis Sample injection

n n The commonly used detectors are absorbance, fluorescence and mass spectrometry. In addition to these, there also several other detection systems adapted for use such as electrochemical conductivity and amperometric detections. Capillary electrophoresis Detector

Method Conc. detection limit Mass detection limit (molar) (moles) UV-VIS 10 -5 -10 -8 10 -13 -10 -16 Fluorescence 10 -7 -10 -9 10 -15 -10 -17 Amperometry 10 -10 -10 -11 10 -18 -10 -19 Indirect UV 10 -3 -10 -6 10 -11 -10 -14 Indirect fluorescence 10 -5 -10 -7 10 -13 -10 -15 Indirect amperometry 10 -8 -10 -9 10 -16 -10 -17 LIF 10 -14 -10 -17 10 -18 -10 -21 Conductivity 10 -7 -10 -8 10 -15 -10 -16 MS 10 -8 -10 -9 10 -16 -10 -17 LIF = lesser induced fluorescence Capillary electrophoresis Detection systems applied in CE

n n n These detectors are the most widely used in CE and in HPLC because of : n universal suitability. n relatively insensitive to temperature changes. n non-destructive and n respond only to compounds that absorb radiation at the range of wavelengths from the source light. They typically cover the range of 190 -800 nm. UV-Vis detectors are commonly used In pharmaceutical analysis, because most of drugs contain chromophore, and those without chromphore could be chemically converted to compound that can absorb light. Disadvantage: the short optical path gives poor sensitivity which is often not good enough for the analysis of biological samples Capillary electrophoresis UV-Visible detectors

Capillary electrophoresis Control of separation in Capillary electrophoresis 19

n n As discussed earlier, cations move most quickly towards the point of detection and time has to be allowed for separations to develop and the EOF should not exceed the cationic mobility by an amount which is incompatible with achieving separation. The factors which can be used to control EOF have been discussed earlier. Another factor in allowing separation to develop which is simply controlled is the length of the capillary; however, the longer the capillary in relation to a fixed applied potential the lower the electric field which is in volts/cm, Since the detection system is mounted before the column outlet, it is important that the distance between the detector and the outlet is not too great since the effective length of the capillary is reduced. Capillary electrophoresis 1. Migration time

n n Longitudinal diffusion is generally the most important cause of peak broadening in CE because of the absence of mass transfer and eddy diffusion (multiple path) seen in other types of chromatography which contain packed stationary phase. Thus to some extent CE resembles capillary gas chromatography but with less mass transfer effects and lower longitudinal diffusion since the sample is in the liquid phase. Longitudinal diffusion depends on the length of time an analyte spends in the capillary and also on the diffusion coefficient of the analyte in the mobile phase. Large analytes such as proteins and oligonucleotides have low diffusion coefficients and thus CE can produce very efficient separations of these types of analyte. Capillary electrophoresis 2. Longitudinal diffusion

n n The capillaries used in CE have narrow internal diameters. For a l 00 cm >< 50 pm i. d. capillary an injection of 0. 02 ul would occupy a l cm length of capillary space. Automatic injection can overcome difficulties in reproducible injection of such small volumes but often detection limits require that larger amounts of sample are injected. Typically the injection is accomplished by applying pressure at the sample loading end of the capillary. An important element in accomplishing efficient sample loading, particularly if detection limits are a problem and a larger volume of sample has to be loaded, is stacking. Capillary electrophoresis 3. Injection plug length

n n n A simple method for achieving stacking is to dissolve the sample in water or low conductivity buffer. The greater resistance of the water plug causes a localised increase in electrical potential across the plug width and the sample ions dissolved in the plug will migrate rapidly until the boundary of the running buffer is reached. By using this method, longer plugs up to 10% of the capillary length can be injected, resulting in an increase in detection limit. Capillary electrophoresis .

n n n The strength of the electric field which can be applied across the capillary is limited by conversion of electrical energy into heat. Localised heating can cause changes in the viscosity of the running buffer and a localised increase in analyte diffusion. Heat generation can be minimised by using narrow capillaries where heat dissipation is rapid and by providing a temperature-controlled environment for the capillary Capillary electrophoresis 4. Joule heating

n n n Analytes may absorb onto thc wall of thc capillary either by interaction with the negatively charged silanol groups or by hydrophobic intcraction. High ionic strength buffers block the negative charge on the capillary wall and reduce the EOF but also increase heating. If only analysis of cations is required, the p. H of the running buffer can be lowered, e. g. to p. H 2. The low p. H suppresses the charge on the silanol groups, reduces EOF to a low level but ensures full ionic mobility of the cations, which will migrate to the cathode without the aid of the EOE Full ionisation of the analytes does not allow for differences in p. Ka to be used in producing separation. Capillary electrophoresis 5. Solute wal. I interactions

n n n The mobility of the running buffer has to be fairly similar to the mobility of the ions in the sample zone. If the mobility of the analyte ions is greater than the mobility of the buffer ions, a fronting peak will result since the ions at the front of the sample zone tend to diffuse into the running buffer solution where they experience a greater applied electric field (due to the higher resistance of the buffer compared with the sample) and accelerate away from the sample zone. This effect will be less if the concentration of the running buffer is much greater than that of the sample. Capillary electrophoresis 6. Electro-dispersion

n n Conversely, if the mobility of the sample ions is lower than that ofthe running buffer ions, a tailing peak will be produced because the ions in the rear of the sample zone will tend to diffuse into the buffer where they will experience a lower applied electric field (due to the lower resistance of the buffer compared with the sample) and will thus lag further behind the sample zone. This effect will be less if the concentration of the running buffer is much lower than that of the sample zone. Capillary electrophoresis .

Capillary electrophoresis Comparing CE with HPLC or slab electrophoresis 28

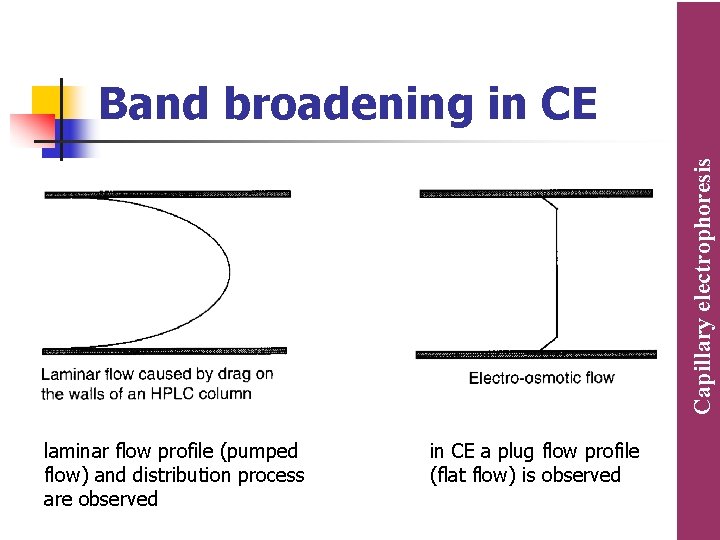
n n Separation efficiency is higher in CE, because in HPLC, additional peak broadening by the laminar flow and distribution process are observed while in CE a plug flow profile (flat flow) is observed. Due to this higher efficiency, systematic errors by peak overlapping are less likely in CE In CE, the capillaries can be easily cleaned and replaced (as they are much cheaper compared to HPLC columns). Samples containing complex matrix components (plasma samples, polymer solutions or plant extract) can be directly injected onto a capillary. The overall cost of running samples and sample preparation is strongly reduced compared to HPLC. Capillary electrophoresis Comparison between CE and HPLC

Capillary electrophoresis Band broadening in CE laminar flow profile (pumped flow) and distribution process are observed in CE a plug flow profile (flat flow) is observed

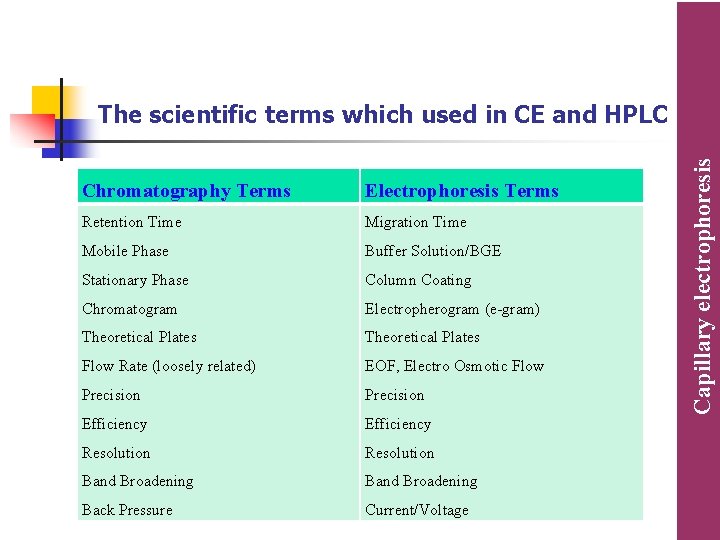
Chromatography Terms Electrophoresis Terms Retention Time Migration Time Mobile Phase Buffer Solution/BGE Stationary Phase Column Coating Chromatogram Electropherogram (e-gram) Theoretical Plates Flow Rate (loosely related) EOF, Electro Osmotic Flow Precision Efficiency Resolution Band Broadening Back Pressure Current/Voltage Capillary electrophoresis The scientific terms which used in CE and HPLC

n CE is a development of SGE but offers much higher resolution, which is due to two effects: n Because of the very small inner diameter, the temperature gradient within the capillary is small, leading to very low convection and therefore a negligible disturbance of the bands/peaks. n Because of the small dimensions, capillaries offer a good heat dissipation, which allows the operation of high electric fields (up to 1000 V/cm and more). These fields allow a much reduced analysis time, which means less diffusion of the bands. Capillary electrophoresis Comparison with conventional Slab Gel Electrophoresis

n n n CE offers higher speed than slab gel techniques. Capillaries are available in a variety of inner diameters (about 10 to 300 um) and lengths (from a few centimeters to several meters)making them useful for many different needs(e. g. fast separation, high-resolution separation). CE can be automated as it offers on-line detection. CE is compatible with a number of detection methods, such as UV -absorbance, laser-induced fluorescence (LIF), mass spectrometry (MS), chemiluminescence and voltametry. The sample volume can be very small (e. g. 1µl). The range of electrolyte combinations is large and includes the addition of volatile additives. Capillary electrophoresis .
