Capacity Fade with Aqueous Soluble Organics Its Measurement

Capacity Fade with Aqueous Soluble Organics: Its Measurement, Minimization, and Reversal Research supported by U. S. DOE award DE-AC 05 -76 RL 01830 through PNNL subcontract 428977 PI: Michael J. Aziz, Harvard School of Engineering & Applied Sciences co-I’s: Roy Gordon, Shmuel Rubinstein, Christopher Rycroft • Methods for measuring extremely low capacity fade rates (HIDDEN) • “Methuselah” family of anthraquinone negolytes: record-breaking capacity retention rate • Bringing dead redox-active molecules back to life: inexpensive dihydroxyanthraquinone • Porous electrode science & engineering: mapping the mass transport coefficient • Review of ASO capacity fade rates from all published studies (HIDDEN) DOE/OE Energy Storage Peer Review, Albuquerque NM, 2019 -09 -25

Harvard Milestones and Deliverables, 9/10/18 -9/9/19 Aqueous Soluble Organic electrolyte development: Year 1 Milestones: (1) Demonstrate Aqueous-Soluble Organic cycling capacity of at least 1. 0 moles of transferrable electrons per liter. (2) Determine the degradation mechanism for the demonstrated ASO electrolyte. Porous Electrode Science & Engineering: Year 1 Milestone: Two-dimensional mapping of mass transport coefficient in two or more commercial carbon papers or felts at four or more flow rates. 2
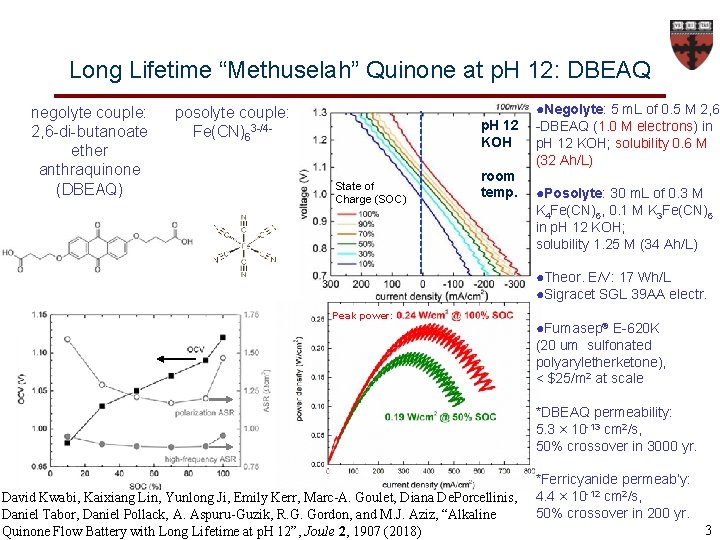
Long Lifetime “Methuselah” Quinone at p. H 12: DBEAQ negolyte couple: 2, 6 -di-butanoate ether anthraquinone (DBEAQ) posolyte couple: Fe(CN)63 -/4 - p. H 12 KOH State of Charge (SOC) room temp. ●Negolyte: 5 m. L of 0. 5 M 2, 6 -DBEAQ (1. 0 M electrons) in p. H 12 KOH; solubility 0. 6 M (32 Ah/L) ●Posolyte: 30 m. L of 0. 3 M K 4 Fe(CN)6, 0. 1 M K 3 Fe(CN)6 in p. H 12 KOH; solubility 1. 25 M (34 Ah/L) ●Theor. E/V: 17 Wh/L ●Sigracet SGL 39 AA electr. Peak power: ●Fumasep® E-620 K (20 um sulfonated polyaryletherketone), < $25/m 2 at scale *DBEAQ permeability: 5. 3 × 10 -13 cm 2/s, 50% crossover in 3000 yr. David Kwabi, Kaixiang Lin, Yunlong Ji, Emily Kerr, Marc-A. Goulet, Diana De. Porcellinis, Daniel Tabor, Daniel Pollack, A. Aspuru-Guzik, R. G. Gordon, and M. J. Aziz, “Alkaline Quinone Flow Battery with Long Lifetime at p. H 12”, Joule 2, 1907 (2018) *Ferricyanide permeab’y: 4. 4 × 10 -12 cm 2/s, 50% crossover in 200 yr. 3
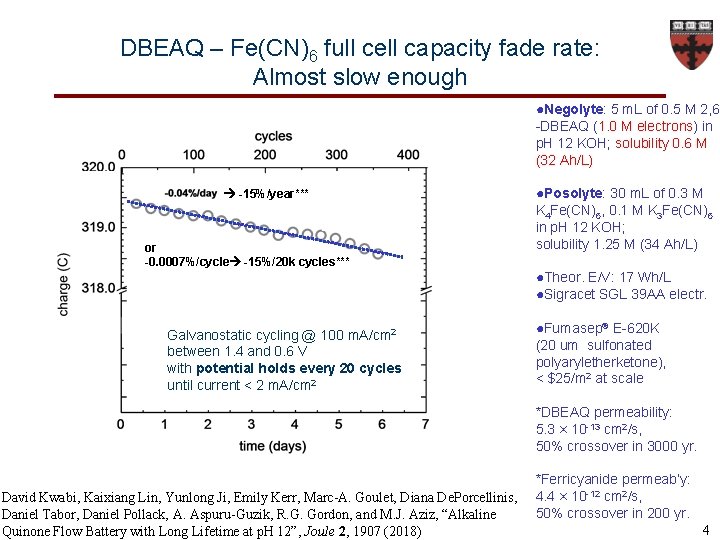
DBEAQ – Fe(CN)6 full cell capacity fade rate: Almost slow enough ●Negolyte: 5 m. L of 0. 5 M 2, 6 -DBEAQ (1. 0 M electrons) in p. H 12 KOH; solubility 0. 6 M (32 Ah/L) -15%/year*** or -0. 0007%/cycle -15%/20 k cycles*** ●Posolyte: 30 m. L of 0. 3 M K 4 Fe(CN)6, 0. 1 M K 3 Fe(CN)6 in p. H 12 KOH; solubility 1. 25 M (34 Ah/L) ●Theor. E/V: 17 Wh/L ●Sigracet SGL 39 AA electr. Galvanostatic cycling @ 100 m. A/cm 2 between 1. 4 and 0. 6 V with potential holds every 20 cycles until current < 2 m. A/cm 2 ●Fumasep® E-620 K (20 um sulfonated polyaryletherketone), < $25/m 2 at scale *DBEAQ permeability: 5. 3 × 10 -13 cm 2/s, 50% crossover in 3000 yr. David Kwabi, Kaixiang Lin, Yunlong Ji, Emily Kerr, Marc-A. Goulet, Diana De. Porcellinis, Daniel Tabor, Daniel Pollack, A. Aspuru-Guzik, R. G. Gordon, and M. J. Aziz, “Alkaline Quinone Flow Battery with Long Lifetime at p. H 12”, Joule 2, 1907 (2018) *Ferricyanide permeab’y: 4. 4 × 10 -12 cm 2/s, 50% crossover in 200 yr. 4
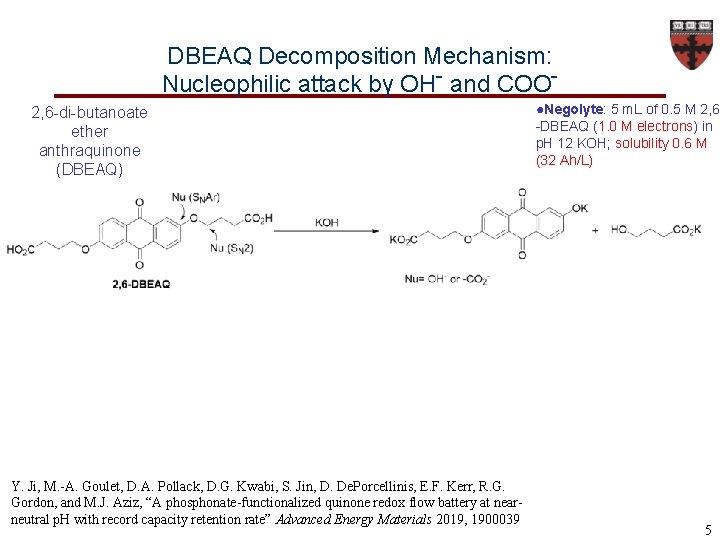
DBEAQ Decomposition Mechanism: Nucleophilic attack by OH- and COO 2, 6 -di-butanoate ether anthraquinone (DBEAQ) Y. Ji, M. -A. Goulet, D. A. Pollack, D. G. Kwabi, S. Jin, D. De. Porcellinis, E. F. Kerr, R. G. Gordon, and M. J. Aziz, “A phosphonate-functionalized quinone redox flow battery at nearneutral p. H with record capacity retention rate” Advanced Energy Materials 2019, 1900039 ●Negolyte: 5 m. L of 0. 5 M 2, 6 -DBEAQ (1. 0 M electrons) in p. H 12 KOH; solubility 0. 6 M (32 Ah/L) 5
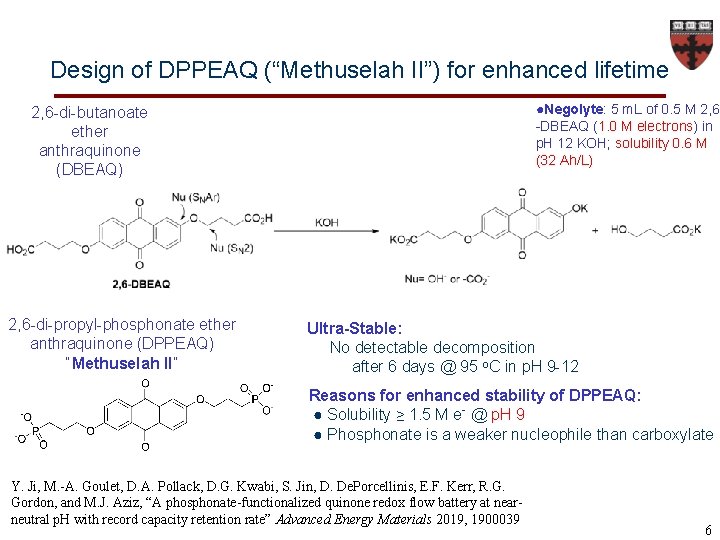
Design of DPPEAQ (“Methuselah II”) for enhanced lifetime ●Negolyte: 5 m. L of 0. 5 M 2, 6 -DBEAQ (1. 0 M electrons) in p. H 12 KOH; solubility 0. 6 M (32 Ah/L) 2, 6 -di-butanoate ether anthraquinone (DBEAQ) 2, 6 -di-propyl-phosphonate ether anthraquinone (DPPEAQ) “Methuselah II” Ultra-Stable: No detectable decomposition after 6 days @ 95 o. C in p. H 9 -12 Reasons for enhanced stability of DPPEAQ: ● Solubility ≥ 1. 5 M e- @ p. H 9 ● Phosphonate is a weaker nucleophile than carboxylate Y. Ji, M. -A. Goulet, D. A. Pollack, D. G. Kwabi, S. Jin, D. De. Porcellinis, E. F. Kerr, R. G. Gordon, and M. J. Aziz, “A phosphonate-functionalized quinone redox flow battery at nearneutral p. H with record capacity retention rate” Advanced Energy Materials 2019, 1900039 6
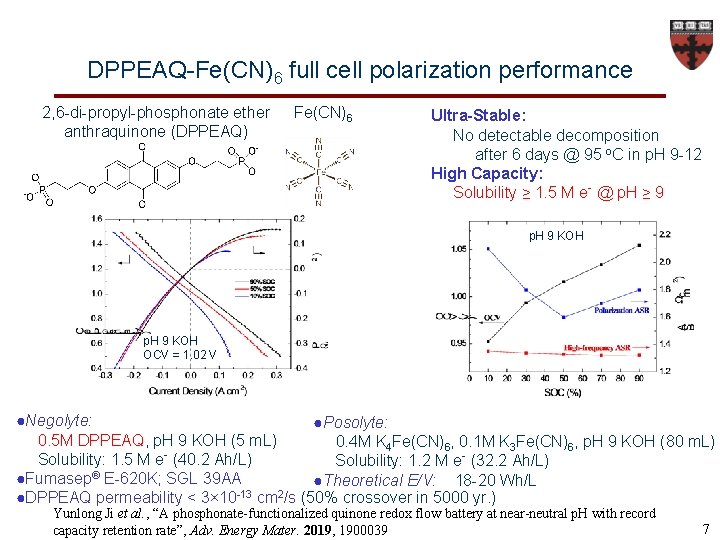
DPPEAQ-Fe(CN)6 full cell polarization performance 2, 6 -di-propyl-phosphonate ether anthraquinone (DPPEAQ) Fe(CN)6 Ultra-Stable: No detectable decomposition after 6 days @ 95 o. C in p. H 9 -12 High Capacity: Solubility ≥ 1. 5 M e- @ p. H ≥ 9 p. H 9 KOH OCV = 1. 02 V ●Negolyte: ●Posolyte: 0. 5 M DPPEAQ, p. H 9 KOH (5 m. L) 0. 4 M K 4 Fe(CN)6, 0. 1 M K 3 Fe(CN)6, p. H 9 KOH (80 m. L) Solubility: 1. 5 M e (40. 2 Ah/L) Solubility: 1. 2 M e- (32. 2 Ah/L) ●Fumasep® E-620 K; SGL 39 AA ●Theoretical E/V: 18 -20 Wh/L -13 2 ●DPPEAQ permeability < 3× 10 cm /s (50% crossover in 5000 yr. ) Yunlong Ji et al. , “A phosphonate-functionalized quinone redox flow battery at near-neutral p. H with record capacity retention rate”, Adv. Energy Mater. 2019, 1900039 7
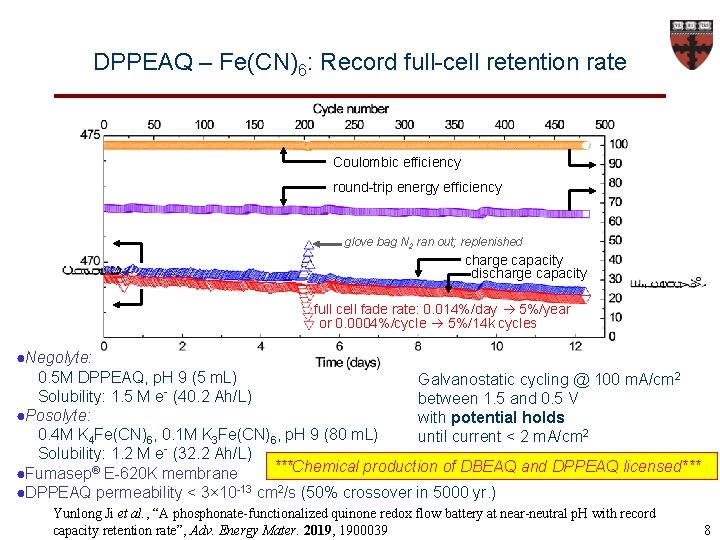
DPPEAQ – Fe(CN)6: Record full-cell retention rate Coulombic efficiency round-trip energy efficiency glove bag N 2 ran out; replenished charge capacity discharge capacity full cell fade rate: 0. 014%/day 5%/year or 0. 0004%/cycle 5%/14 k cycles ●Negolyte: 0. 5 M DPPEAQ, p. H 9 (5 m. L) Galvanostatic cycling @ 100 m. A/cm 2 Solubility: 1. 5 M e- (40. 2 Ah/L) between 1. 5 and 0. 5 V ●Posolyte: with potential holds 0. 4 M K 4 Fe(CN)6, 0. 1 M K 3 Fe(CN)6, p. H 9 (80 m. L) until current < 2 m. A/cm 2 Solubility: 1. 2 M e- (32. 2 Ah/L) ***Chemical production of DBEAQ and DPPEAQ licensed*** ●Fumasep® E-620 K membrane ●DPPEAQ permeability < 3× 10 -13 cm 2/s (50% crossover in 5000 yr. ) Yunlong Ji et al. , “A phosphonate-functionalized quinone redox flow battery at near-neutral p. H with record capacity retention rate”, Adv. Energy Mater. 2019, 1900039 8
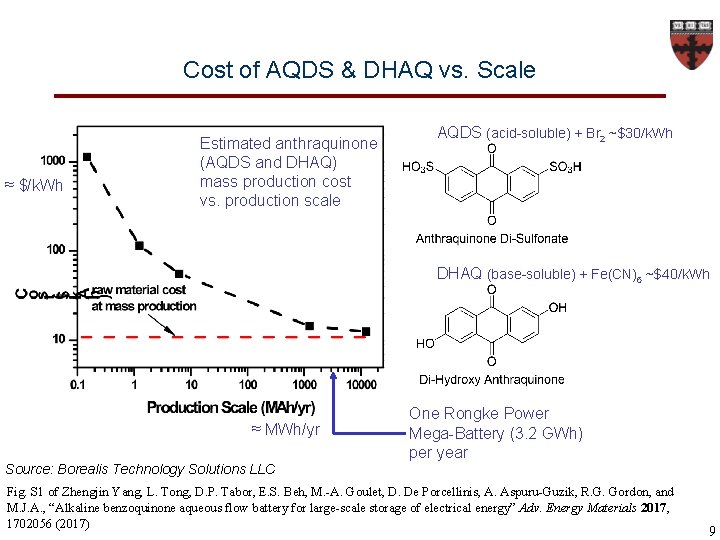
Cost of AQDS & DHAQ vs. Scale ≈ $/k. Wh Estimated anthraquinone (AQDS and DHAQ) mass production cost vs. production scale AQDS (acid-soluble) + Br 2 ~$30/k. Wh DHAQ (base-soluble) + Fe(CN)6 ~$40/k. Wh ≈ MWh/yr One Rongke Power Mega-Battery (3. 2 GWh) per year Source: Borealis Technology Solutions LLC Fig. S 1 of Zhengjin Yang, L. Tong, D. P. Tabor, E. S. Beh, M. -A. Goulet, D. De Porcellinis, A. Aspuru-Guzik, R. G. Gordon, and M. J. A. , “Alkaline benzoquinone aqueous flow battery for large-scale storage of electrical energy” Adv. Energy Materials 2017, 1702056 (2017) 9
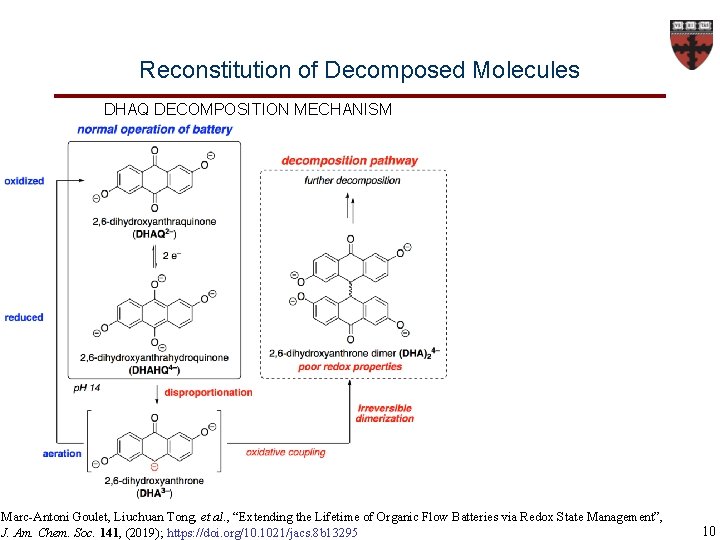
Reconstitution of Decomposed Molecules DHAQ DECOMPOSITION MECHANISM Marc-Antoni Goulet, Liuchuan Tong, et al. , “Extending the Lifetime of Organic Flow Batteries via Redox State Management”, J. Am. Chem. Soc. 141, (2019); https: //doi. org/10. 1021/jacs. 8 b 13295 10
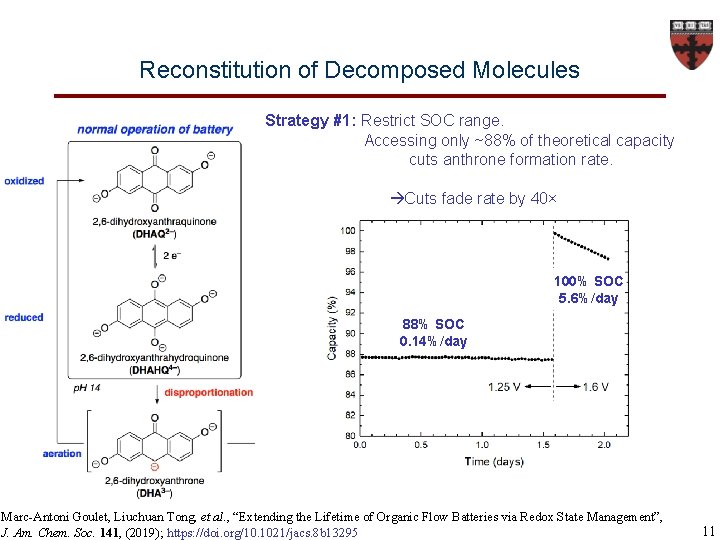
Reconstitution of Decomposed Molecules Strategy #1: Restrict SOC range. Accessing only ~88% of theoretical capacity cuts anthrone formation rate. Cuts fade rate by 40× 100% SOC 5. 6%/day 88% SOC 0. 14%/day Marc-Antoni Goulet, Liuchuan Tong, et al. , “Extending the Lifetime of Organic Flow Batteries via Redox State Management”, J. Am. Chem. Soc. 141, (2019); https: //doi. org/10. 1021/jacs. 8 b 13295 11
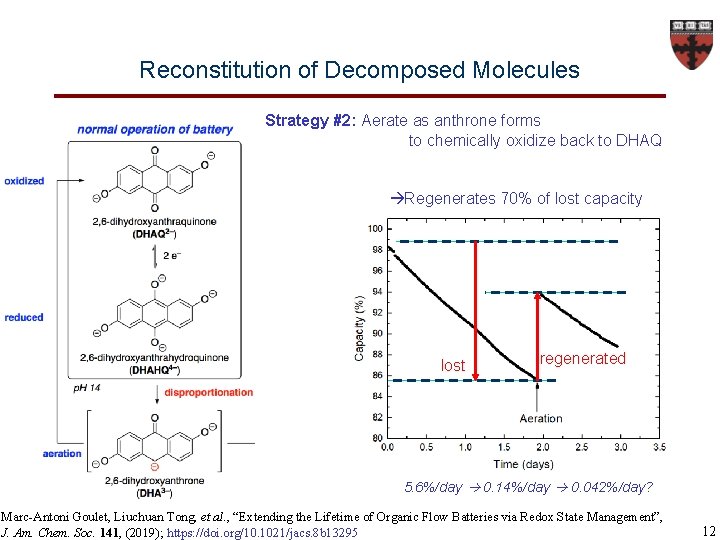
Reconstitution of Decomposed Molecules Strategy #2: Aerate as anthrone forms to chemically oxidize back to DHAQ Regenerates 70% of lost capacity lost regenerated 5. 6%/day 0. 14%/day 0. 042%/day? Marc-Antoni Goulet, Liuchuan Tong, et al. , “Extending the Lifetime of Organic Flow Batteries via Redox State Management”, J. Am. Chem. Soc. 141, (2019); https: //doi. org/10. 1021/jacs. 8 b 13295 12
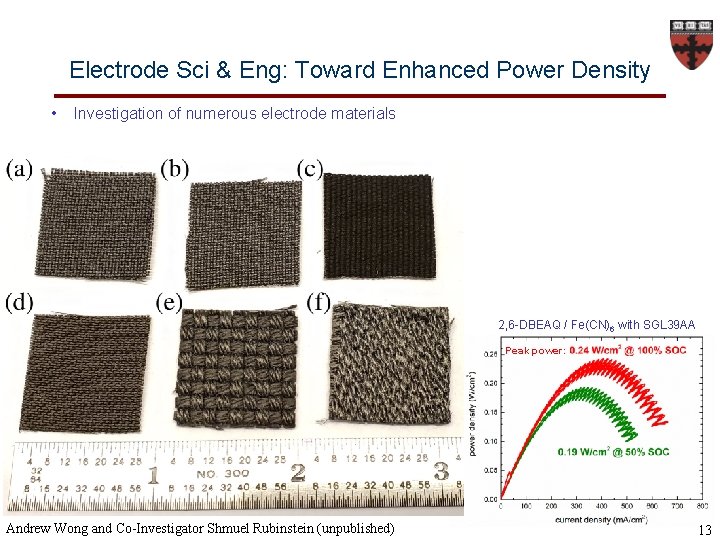
Electrode Sci & Eng: Toward Enhanced Power Density • Investigation of numerous electrode materials 2, 6 -DBEAQ / Fe(CN)6 with SGL 39 AA Peak power: Andrew Wong and Co-Investigator Shmuel Rubinstein (unpublished) 13
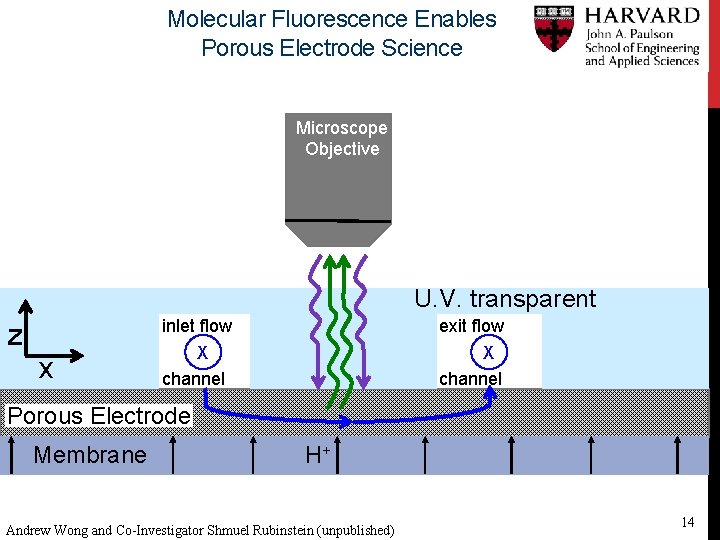
Molecular Fluorescence Enables Porous Electrode Science Microscope Objective U. V. transparent z x inlet flow X channel exit flow X channel Porous Electrode Membrane H+ Andrew Wong and Co-Investigator Shmuel Rubinstein (unpublished) 14
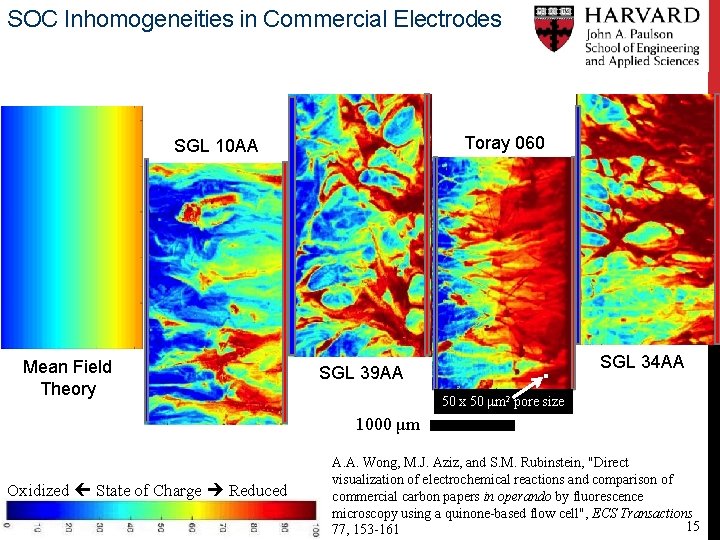
SOC Inhomogeneities in Commercial Electrodes Toray 060 SGL 10 AA Mean Field Theory SGL 34 AA SGL 39 AA 50 x 50 μm 2 pore size 1000 μm Oxidized State of Charge Reduced A. A. Wong, M. J. Aziz, and S. M. Rubinstein, "Direct visualization of electrochemical reactions and comparison of commercial carbon papers in operando by fluorescence microscopy using a quinone-based flow cell", ECS Transactions 15 77, 153 -161
![Mapping Mass Transport Coefficient, km mass transfer rate [mol/s] concentration difference [M] mass transfer Mapping Mass Transport Coefficient, km mass transfer rate [mol/s] concentration difference [M] mass transfer](http://slidetodoc.com/presentation_image/3cf7cae366b0e522dec3b4136b5887d8/image-16.jpg)
Mapping Mass Transport Coefficient, km mass transfer rate [mol/s] concentration difference [M] mass transfer area [m 2] Quinone concentration during reduction (a) Quinone concentration difference (b) Reciprocal of concentration differential (c) 1 mm Andrew Wong and Co-Investigator Shmuel Rubinstein (unpublished) Mass Transport Coefficient Map (d) 1 mm 16
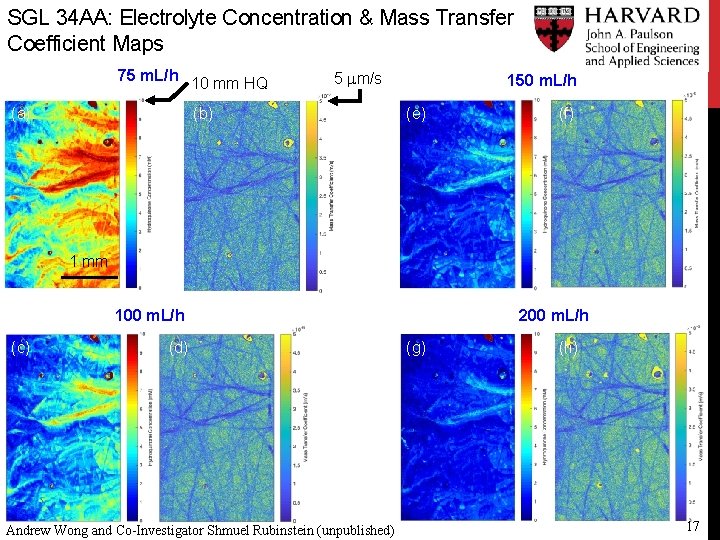
SGL 34 AA: Electrolyte Concentration & Mass Transfer Coefficient Maps 75 m. L/h (a) 10 mm HQ 5 mm/s (b) 150 m. L/h (e) (f) 1 mm 100 m. L/h (c) (d) Andrew Wong and Co-Investigator Shmuel Rubinstein (unpublished) 200 m. L/h (g) (h) 17
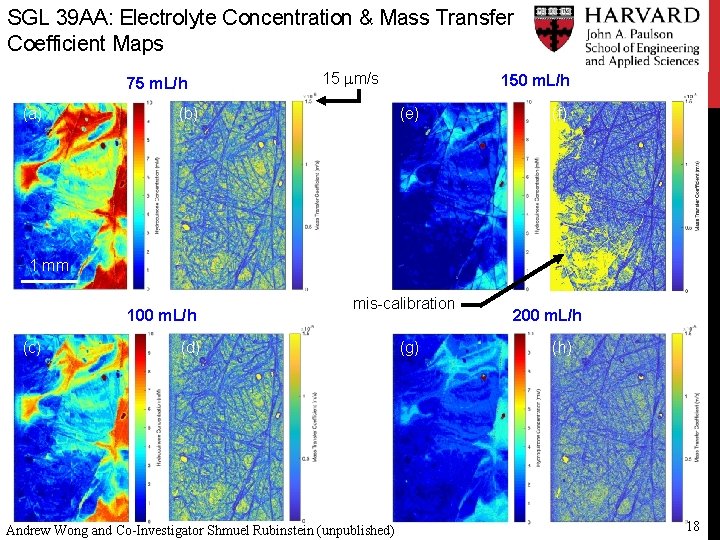
SGL 39 AA: Electrolyte Concentration & Mass Transfer Coefficient Maps 75 m. L/h (a) 15 mm/s (b) 150 m. L/h (e) (f) 1 mm 100 m. L/h (c) mis-calibration (d) Andrew Wong and Co-Investigator Shmuel Rubinstein (unpublished) (g) 200 m. L/h (h) 18
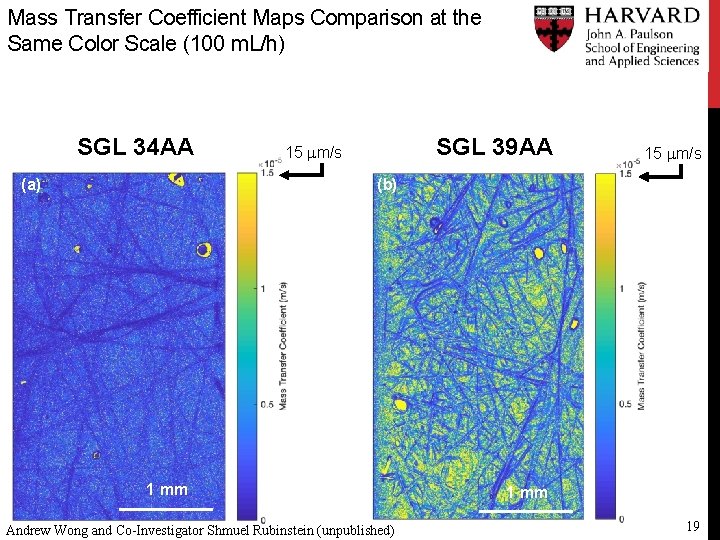
Mass Transfer Coefficient Maps Comparison at the Same Color Scale (100 m. L/h) SGL 34 AA (a) SGL 39 AA 15 mm/s (b) 1 mm Andrew Wong and Co-Investigator Shmuel Rubinstein (unpublished) 1 mm 19
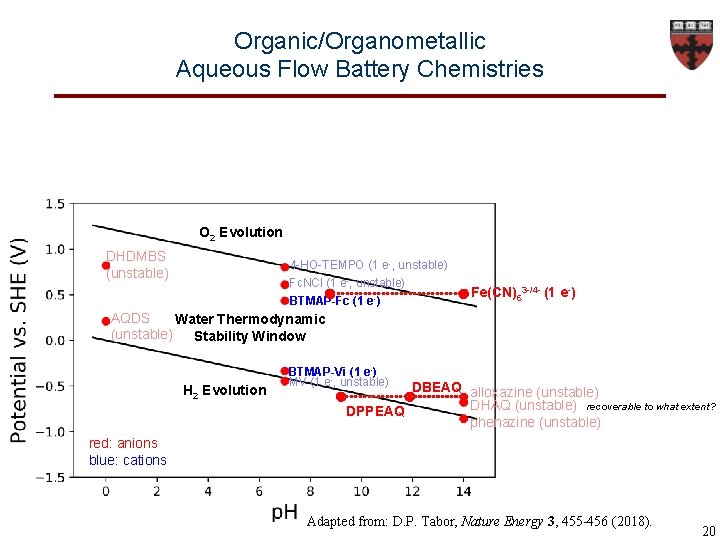
Organic/Organometallic Aqueous Flow Battery Chemistries O 2 Evolution DHDMBS (unstable) 4 -HO-TEMPO (1 e-, unstable) Fc. NCl (1 e-, unstable) BTMAP-Fc (1 e-) Fe(CN)63 -/4 - (1 e-) AQDS Water Thermodynamic (unstable) Stability Window H 2 Evolution BTMAP-Vi (1 e-) MV (1 e-, unstable) DBEAQ alloxazine (unstable) DHAQ (unstable) recoverable to what extent? DPPEAQ phenazine (unstable) red: anions blue: cations Adapted from: D. P. Tabor, Nature Energy 3, 455 -456 (2018). 20

Publications *D. G. Kwabi, K. Lin, Y. Ji, E. F. Kerr, M. -A. Goulet, D. De. Porcellinis, D. P. Tabor, D. A. Pollack, A. Aspuru-Guzik, R. G. Gordon, and M. J. Aziz, “Alkaline Quinone Flow Battery with Long Lifetime at p. H 12” Joule 2, 1907 (2018) *Y. Ji, M. -A. Goulet, D. A. Pollack, D. G. Kwabi, S. Jin, D. De. Porcellinis, E. F. Kerr, R. G. Gordon, and M. J. Aziz, “A phosphonate-functionalized quinone redox flow battery at near-neutral p. H with record capacity retention rate” Advanced Energy Materials 2019, 1900039 *M. -A. Goulet, L. Tong, D. A. Pollack, D. P. Tabor, S. A. Odom, A. Aspuru-Guzik, E. E. Kwan, R. G. Gordon, and M. J. Aziz, “Extending the lifetime of organic flow batteries via redox state management” Journal of the American Chemical Society 141, 8014 (2019) D. G. Kwabi, A. A. Wong, and M. J. Aziz, "Rational Evaluation and Cycle Life Improvement of Quinone-Based Aqueous Flow Batteries Guided by In-Line Optical Spectrophotometry”, J. Electrochem. Soc. 165, A 1770 -1776 (2018) Y. Liu, M. -A. Goulet, L. Tong, Y. Liu, Y. Ji, L. Wu, R. G. Gordon, M. J. Aziz, Z. Yang, and T. Xu, “A long lifetime allorganic aqueous flow battery utilizing TMAP-TEMPO Radical” Chem 5, 1861 (2019) S. Jin, Y. Jing, D. G. Kwabi, Y. Ji, L. Tong, D. De Porcellinis, M. -A. Goulet, D. A. Pollack, R. G. Gordon, and M. J. Aziz, “A water-miscible quinone flow battery with high volumetric capacity and energy density” ACS Energy Letters, 6 1342 (2019) M. Park, E. S. Beh, E. M. Fell, Y. Jing, E. F. Kerr, D. De. Porcellinis, M. -A. Goulet, J. Ryu, A. A. Wong, R. G. Gordon, J. Cho, and M. J. Aziz, “A high voltage aqueous zinc-organic hybrid flow battery” Advanced Energy Materials, 2019, 9, 1900694 L. Tong, Y. Jing, R. G. Gordon, and M. J. Aziz, “Symmetric All-Quinone Aqueous Battery” ACS Applied Energy Materials 6, 4016 (2019) 25
- Slides: 21