Cancer discovery The Pancreatic Cancer Microbiome Promotes Oncogenesis

Cancer discovery The Pancreatic Cancer Microbiome Promotes Oncogenesis by Induction of Innate and Adaptive Immune Suppression Gastroenterology Gut Microbiota Promotes Tumor Growth in Mice by Modulating Immune Response R 3 강 승경 / Pf. 류 지 곤
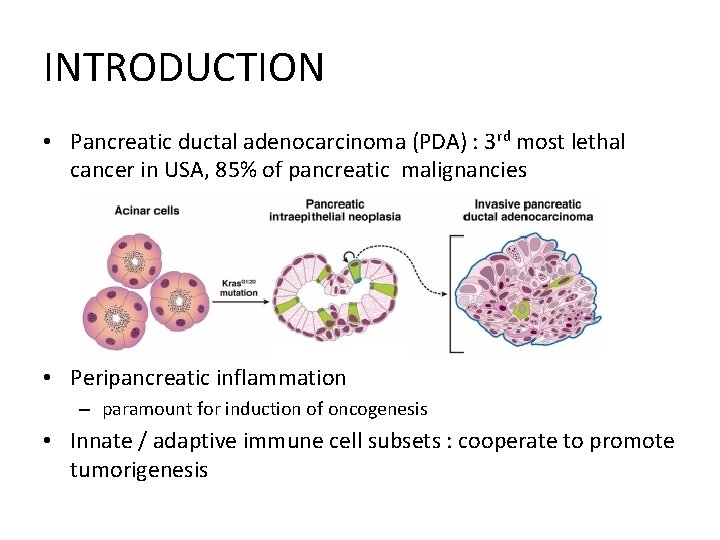
INTRODUCTION • Pancreatic ductal adenocarcinoma (PDA) : 3 rd most lethal cancer in USA, 85% of pancreatic malignancies • Peripancreatic inflammation – paramount for induction of oncogenesis • Innate / adaptive immune cell subsets : cooperate to promote tumorigenesis
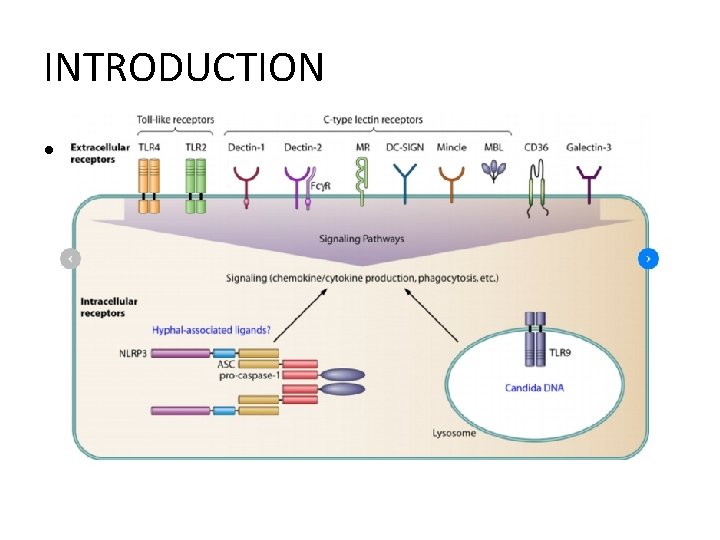
INTRODUCTION • Pattern Recognition Receptors (PRR) – Transmit inflammation, tumorigenesis↑ – Deficient in select PRR signaling • Toll-like receptor 4 (TLR 4), TLR 7, TLR 9, Mincle • Slower progression of PDA – Protumorigenic effects of PRR ligation in PDA Bacterial dysbiosis influences PDA progression.

INTRODUCTION Microbial dysbiosis, Disrupted epithelial barrier function Translocation of bacteria Neoplastic transformation • Microbiome – Contributor to oncogenesis in intestinal tract malignancies • laryngeal, esophageal, gastric, colorectal, primary liver cancer – Direct contact, recipient of portal venous drainage(liver cancer) – Remote from the gastrointestinal lumen or its drainage. – Etiologic relationship? • intrapancreatic microbiota PDA immune-suppressive inflammation in


METHOD • Cell lines – KPC cells • spontaneous pancreatic tumors originating in Kras ; Trp 53 ; Pdx-1 cre mice G 12 D/þ R 172 H/þ – Braf-Pten melanoma cells • topical tamoxifen induced tumors arising in Tyr-Cre. ER; Braf. V 600 E/þ; Ptenfl/fl mice – MC 38 colon cancer cells • Flow Cytometry • 16 S Ribosomal RNA Gene Sequencing • Statistical Analyses – Unpaired Student t test with Welch’s correction – Mann-Whitney test, and 1 -way analysis of variance with Tukey’s multiple comparison test for comparisons

• Establish cancer models – By subcutaneous injection of KPC pancreatic cancer cells or melanoma cells – By splenic injection of KPC cells, B 16 -F 10 melanoma cells, or MC 38 colon cancer cells to induce liver metastases. • Objective – Evaluate the Impact of gut microbiome depletion on tumor growth
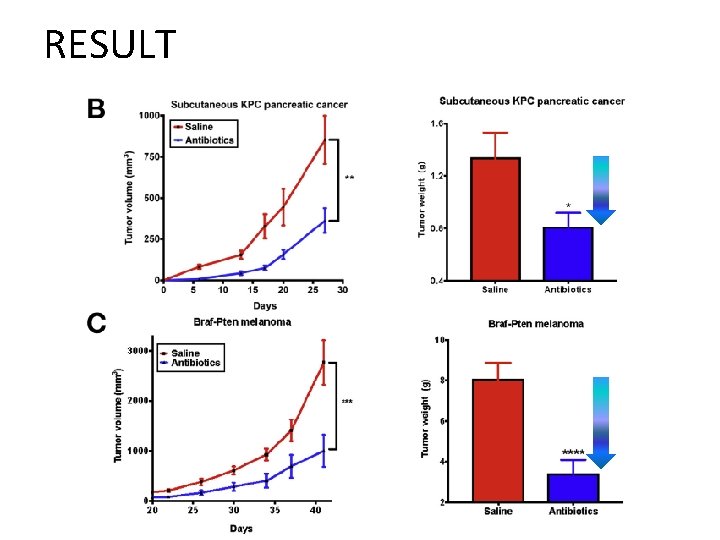
RESULT • Gut microbiome depletion Subcutaneous tumor burden ↓↓ in pancreatic cancer and melanoma models Liver metastases burden ↓↓ in pancreatic cancer, colon cancer, and melanoma models
RESULT
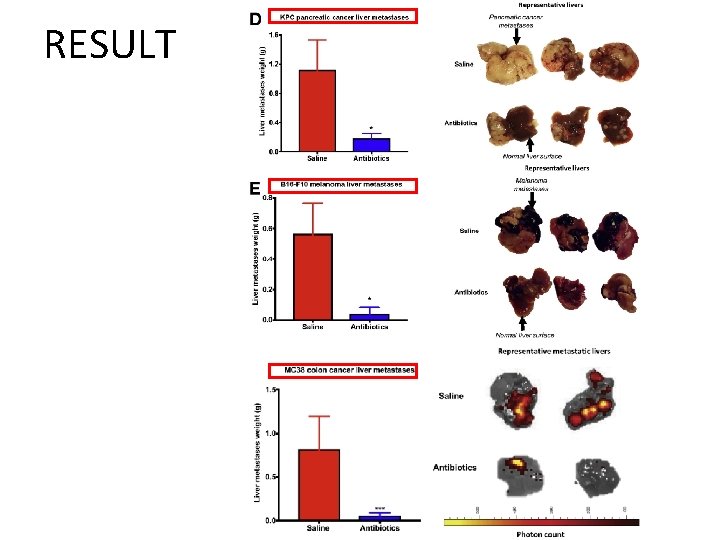
RESULT
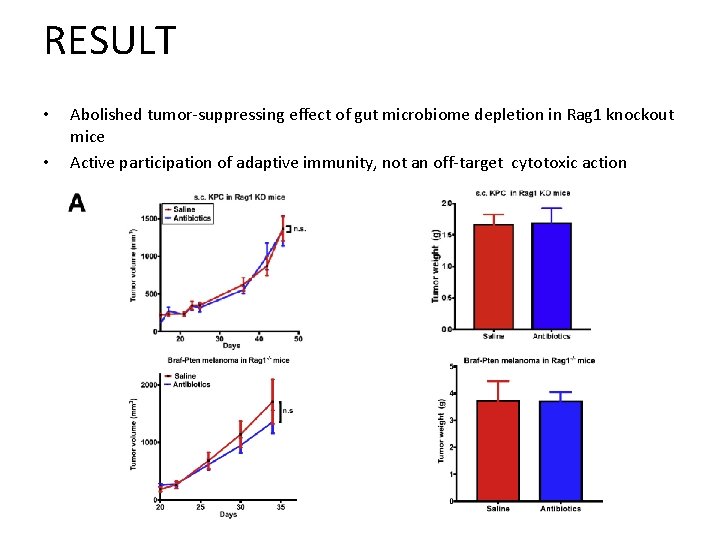
RESULT • • Abolished tumor-suppressing effect of gut microbiome depletion in Rag 1 knockout mice Active participation of adaptive immunity, not an off-target cytotoxic action

RESULT • Impact of gut microbiome depletion on T cells in TME – naïve helper T cells (Th 0) mature into Th 1, Th 2, regulatory T cell, Th 17 lineage – Th 1 - cytokine (IFN-ɣ): anti-tumorigenic role – Th 2/ Treg - cytokines IL 4, IL 5, IL 10 : pro-tumorigenic role • High Th 1/Th 2 ratio in TME : survival ↑ in pancreatic cancer • Th 17 cells – Pro-tumorigenic in pancreatic cancer, melanoma, colorectal cancer. • IL 17 a – Linked to the gut microbiome and plays a key role in defending against fungal and bacterial pathogens.
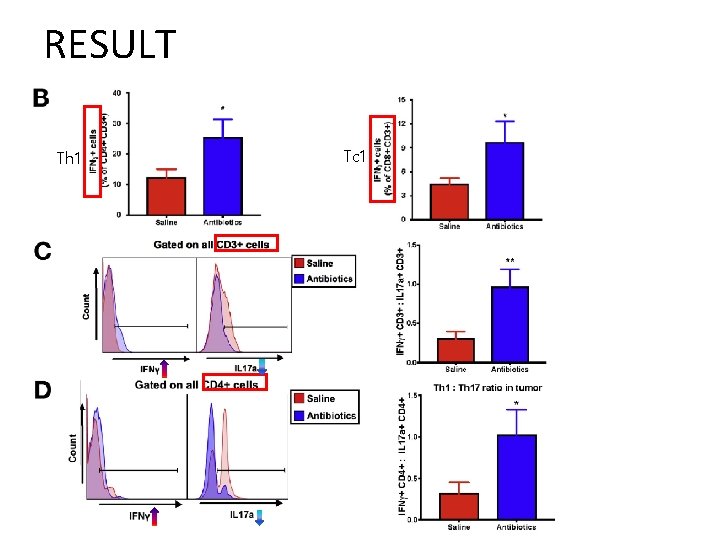
RESULT Th 1 Tc 1
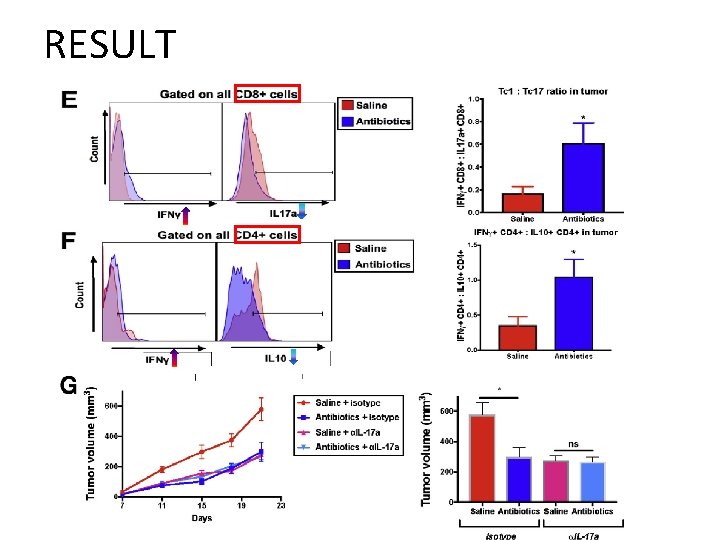
RESULT
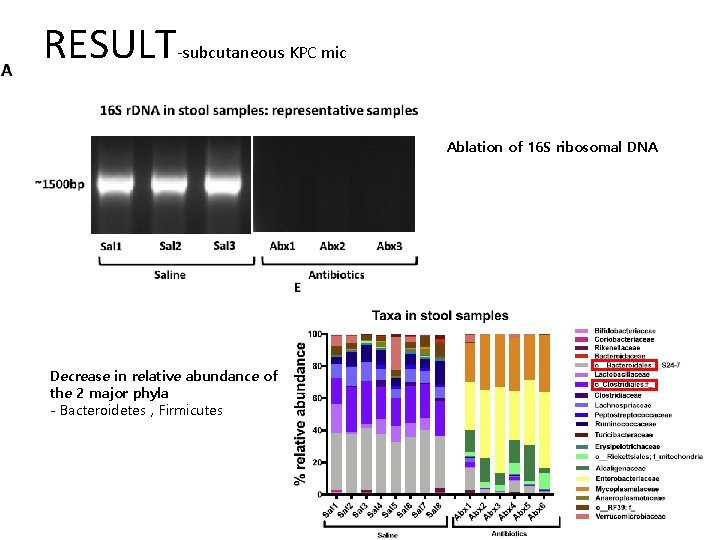
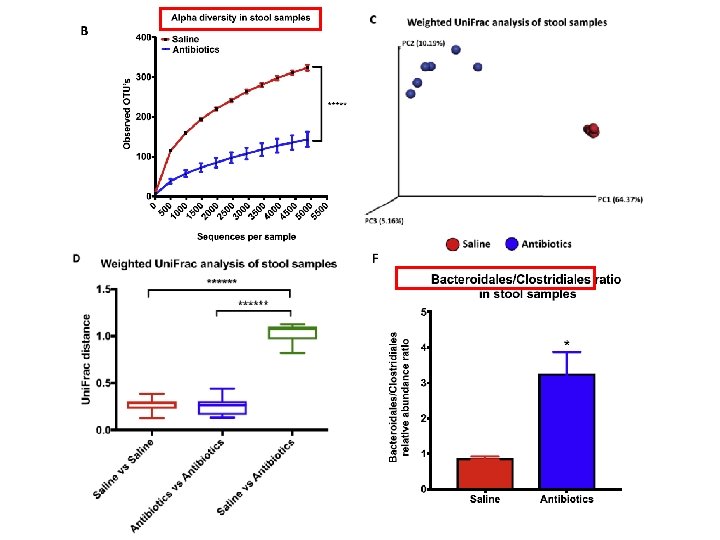
RESULT-subcutaneous KPC mic Ablation of 16 S ribosomal DNA Decrease in relative abundance of the 2 major phyla - Bacteroidetes , Firmicutes

DISCUSSION • Mechanism of interaction with microbiome and immune system : Unclear , some inferences • Gut microbes interact with immune system via PRR in pancreatic and other cancers • Rag 1 knockout / IL 17 a-neutralizing antibody not cytotoxic effect of antibiotics in anti-tumor phenomenon • Depleting the gut microbiome ↑ infiltration of pancreatic tumors with effector T cells. • Future studies – gut microbial modulation strategy potentiate the efficacy of checkpoint inhibitors or cytotoxic drugs

Cancer discovery April, 2018 • 1 Department • 2 S. • • • of Basic Science and Craniofacial Biology, New York University College of Dentistry, New York. Arthur Localio Laboratory, Department of Surgery, New York University School of Medicine, New York. 3 National Gnotobiotic Rodent Research Center, University of North Carolina, Chapel Hill, North Carolina. 4 Department of Epidemiology and Health Promotion, NYU College of Dentistry, New York. 5 Department of Biology, Brooklyn College and the Graduate Center (CUNY), Brooklyn, New York. 6 Department of Medicine, New York University School of Medicine, New York. 7 Department of Medicine, Microbiology, and Immunology, University of North Carolina, Chapel Hill, North Carolina. 8 Department of Cell Biology, New York University School of Medicine, New York.

METHOD • Animals and in vivo models – KC mice: develop spontaneous pancreatic neoplasia by targeted expression of mutant Kras in the pancreas – KPC mice: express mutant intrapancreatic Kras ; Trp 53 ; Pdx 1 cre mice G 12 D/þ R 172 H/þ • Antibiotic treatment, fecal, and bacterial transfer experiments – Ablation of the gut microbiome – 6 -week-old WT or KC mice , antibiotic cocktail by oral gavage daily for 5 d. Controls-PBS. * Broad-spectrum antibiotics cocktail : vancomycin, neomycin, metronidazole, ampicillin and amphotericin B

METHOD • • • Murine cellular isolation, Flow cytometry, In vitro experiments Histology, Immunohistochemistry, RNA analysis Quantitative PCR FISH Statistical considerations for tumor size and immunologic analyses – measurements of tumor size : Student t test
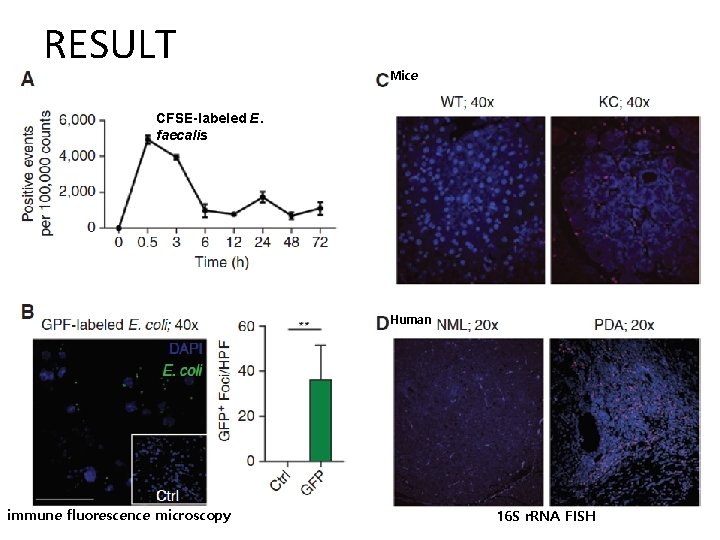
RESULT Mice CFSE-labeled E. faecalis Human immune fluorescence microscopy 16 S r. RNA FISH
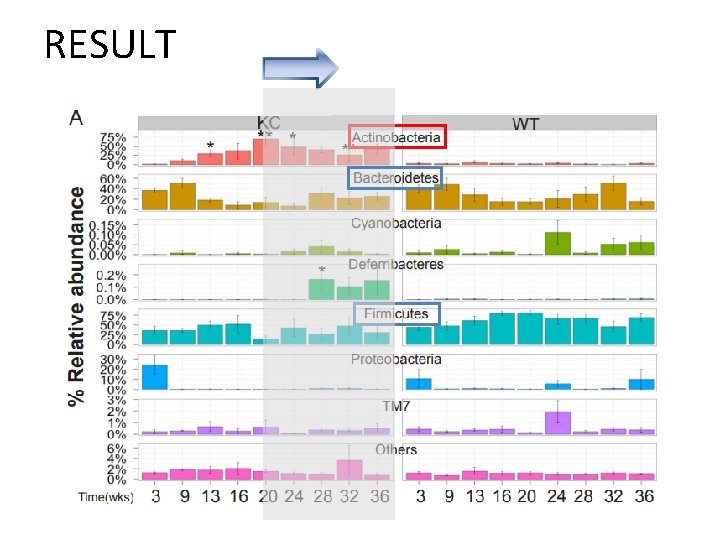
RESULT
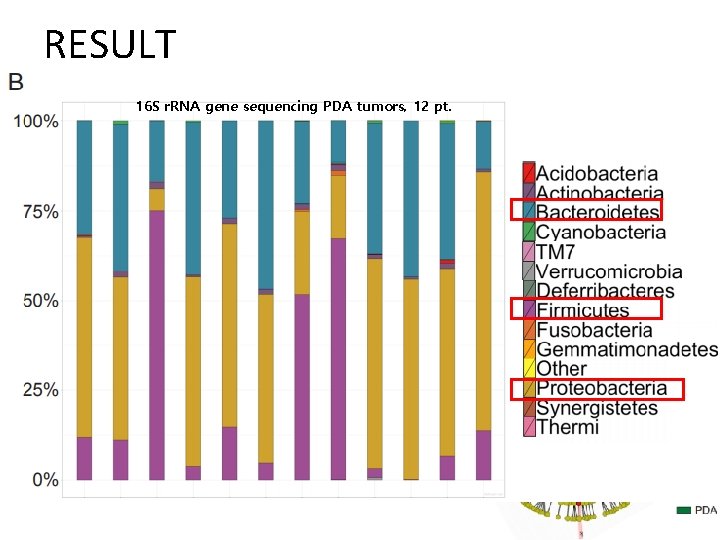
RESULT 16 S r. RNA gene sequencing PDA tumors, 12 pt.
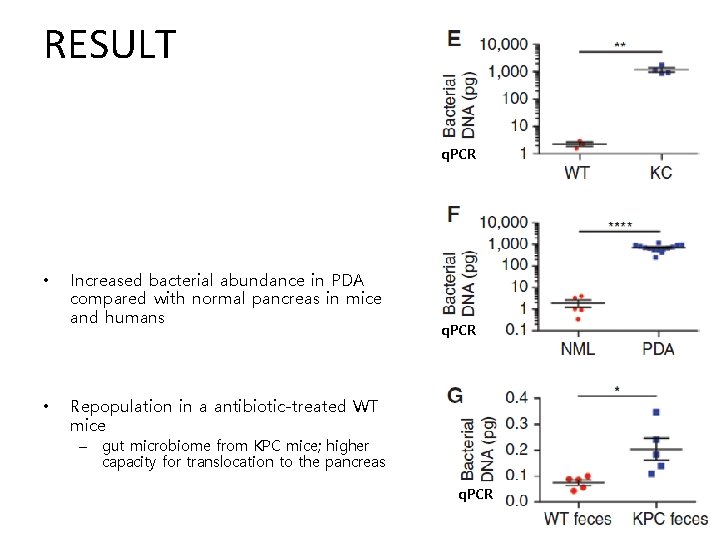
RESULT q. PCR • • Increased bacterial abundance in PDA compared with normal pancreas in mice and humans q. PCR Repopulation in a antibiotic-treated WT mice – gut microbiome from KPC mice; higher capacity for translocation to the pancreas q. PCR
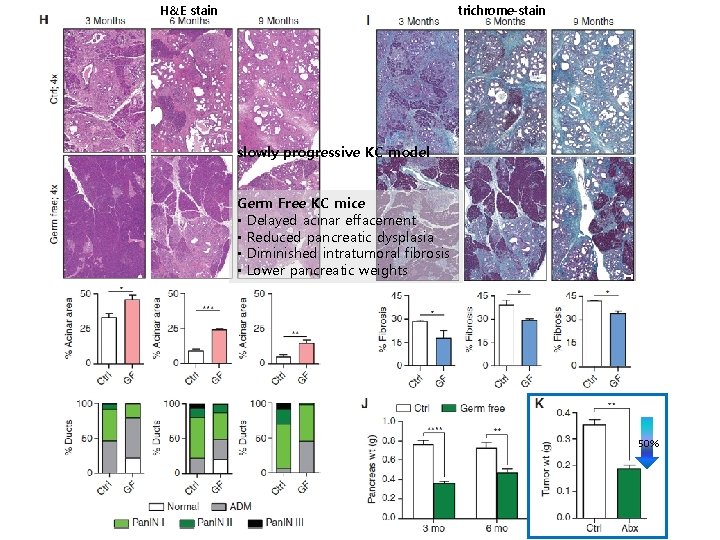
H&E stain trichrome-stain slowly progressive KC model Germ Free KC mice • Delayed acinar effacement • Reduced pancreatic dysplasia • Diminished intratumoral fibrosis • Lower pancreatic weights 50%
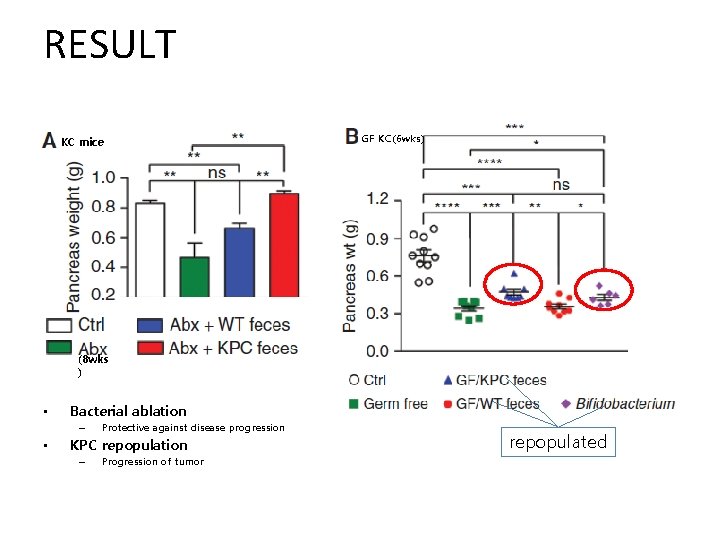
RESULT KC mice GF KC(6 wks) (8 wks ) • Bacterial ablation – • Protective against disease progression KPC repopulation – Progression of tumor repopulated
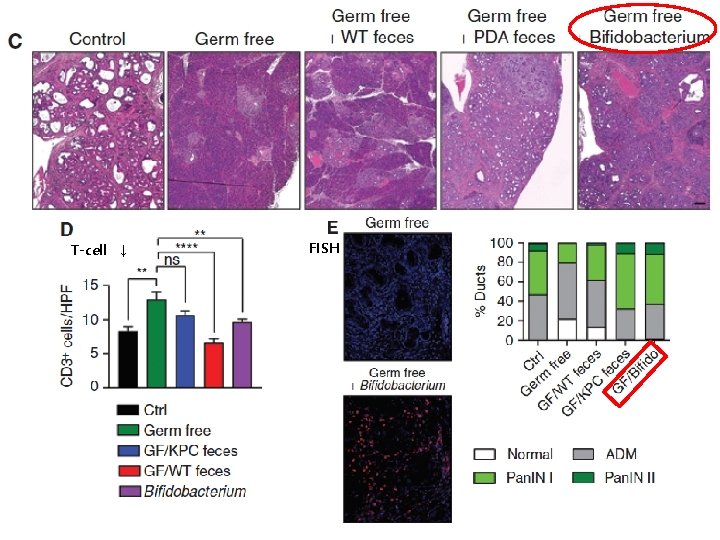
RESULT T-cell ↓ FISH
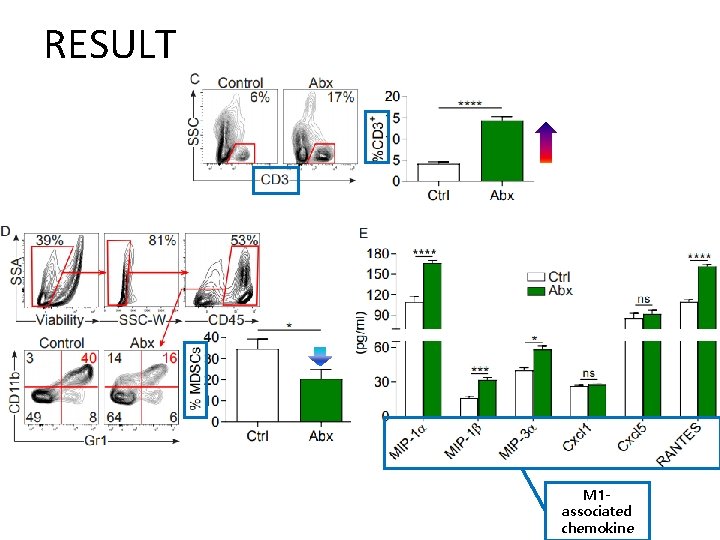
RESULT M 1 associated chemokine
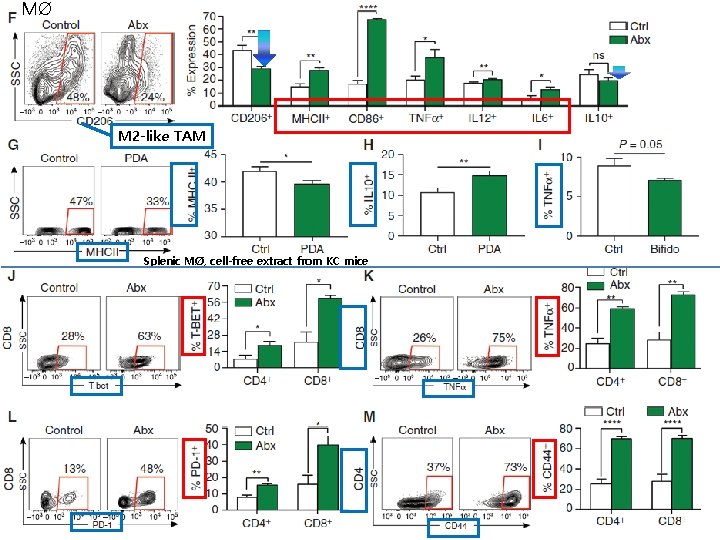
MØ M 2 -like TAM Splenic MØ, cell-free extract from KC mice
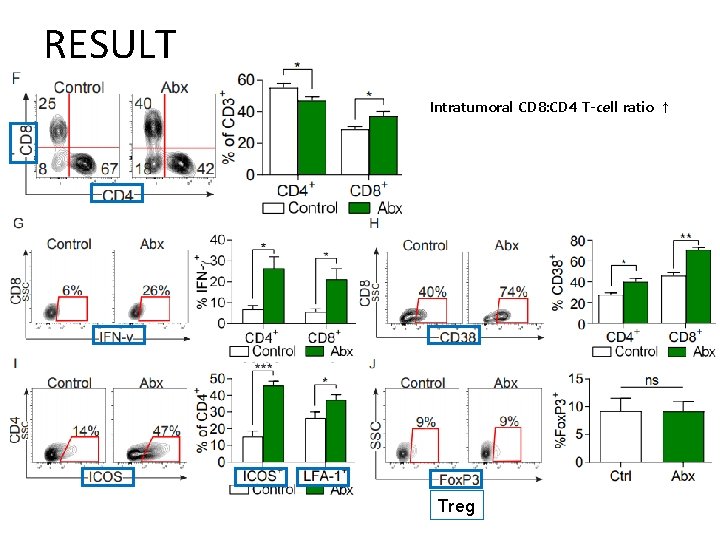
RESULT Intratumoral CD 8: CD 4 T-cell ratio ↑ Treg
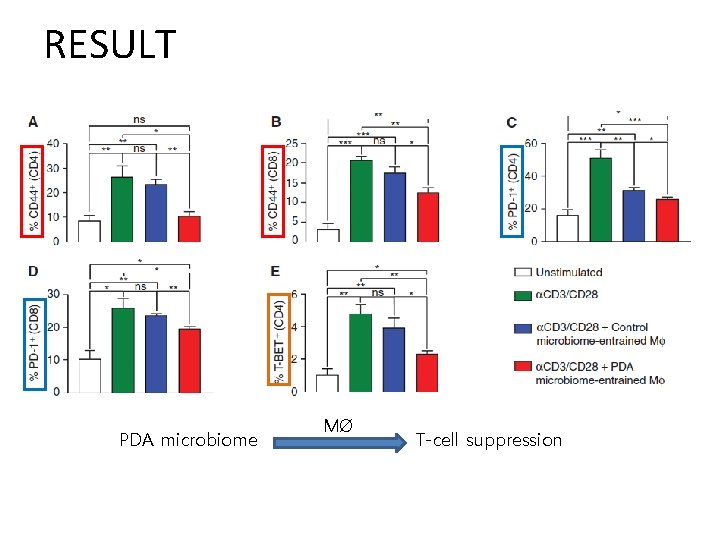
RESULT PDA microbiome MØ T-cell suppression
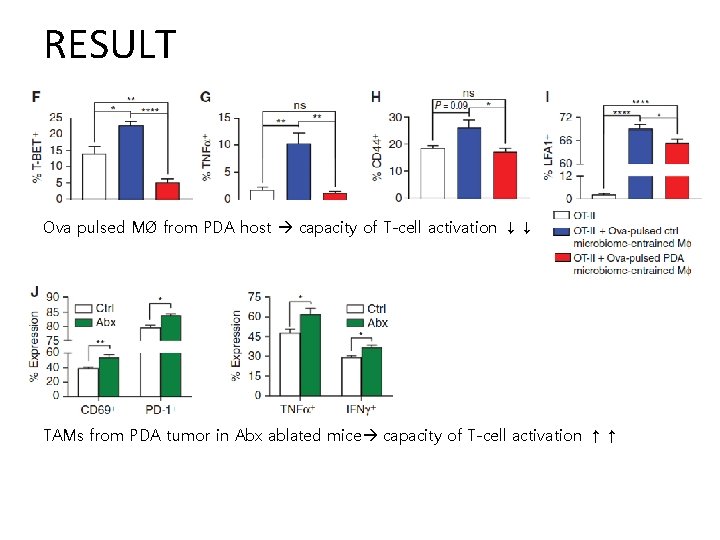
RESULT Ova pulsed MØ from PDA host capacity of T-cell activation ↓↓ TAMs from PDA tumor in Abx ablated mice capacity of T-cell activation ↑↑
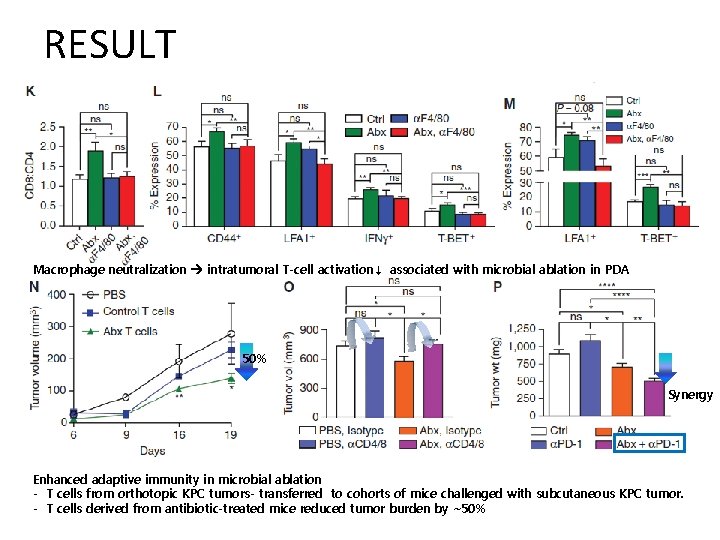
RESULT Macrophage neutralization intratumoral T-cell activation↓ associated with microbial ablation in PDA 50% Synergy Enhanced adaptive immunity in microbial ablation - T cells from orthotopic KPC tumors- transferred to cohorts of mice challenged with subcutaneous KPC tumor. - T cells derived from antibiotic-treated mice reduced tumor burden by ∼ 50%
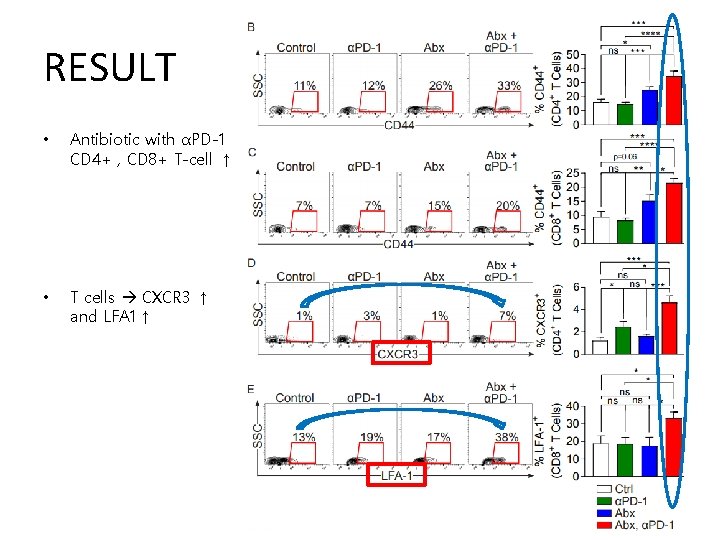
RESULT • Antibiotic with αPD-1 CD 4+ , CD 8+ T-cell ↑ • T cells CXCR 3 ↑ and LFA 1↑
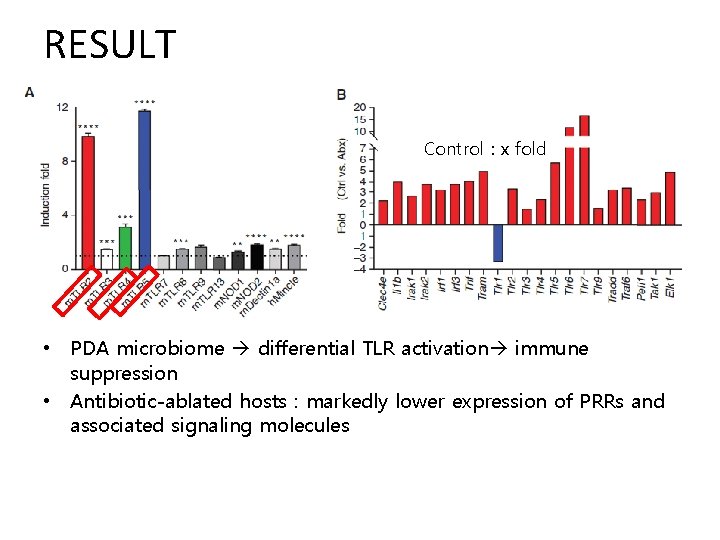
RESULT Control : x fold • PDA microbiome differential TLR activation immune suppression • Antibiotic-ablated hosts : markedly lower expression of PRRs and associated signaling molecules
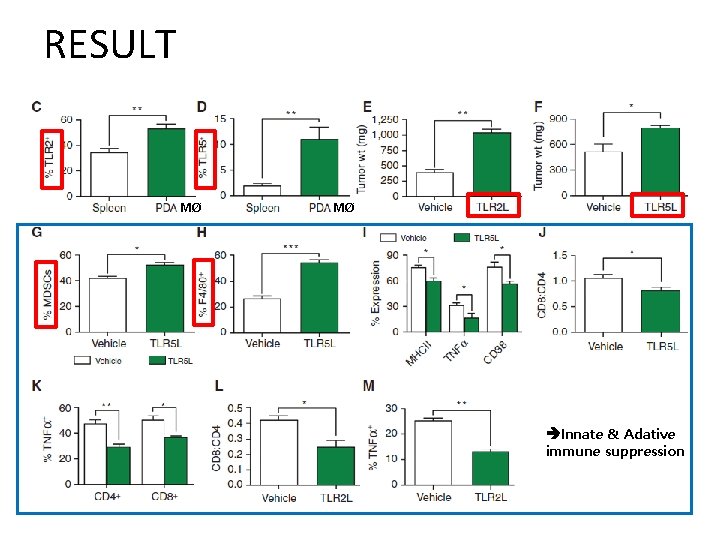
RESULT MØ MØ Innate & Adative immune suppression
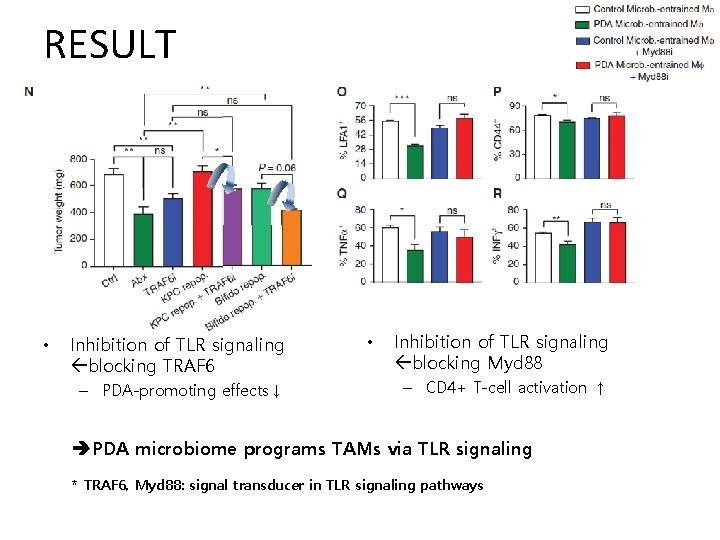
RESULT • Inhibition of TLR signaling blocking TRAF 6 – PDA-promoting effects↓ • Inhibition of TLR signaling blocking Myd 88 – CD 4+ T-cell activation ↑ PDA microbiome programs TAMs via TLR signaling * TRAF 6, Myd 88: signal transducer in TLR signaling pathways

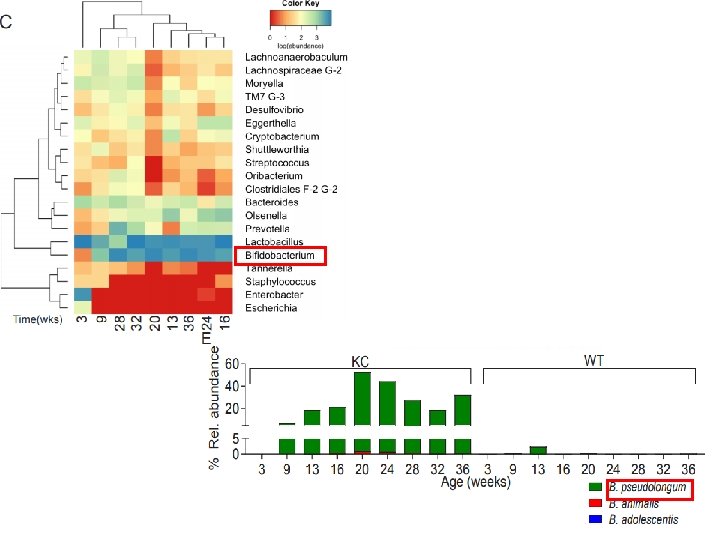
DISCUSSION • Distinct stagespecific gut , pancreatic microbiome intratumoral immune suppression disease progression • Species or signatures that promotes disease pathogenesis : B. pseudolongum • abundant in gut and tumor, accelerated oncogenesis via TLR • Targeting the microbiome – Protected against PDA – Enhanced antitumor immunity – Susceptibility ↑ to immunotherapy • Elements of the microbiome : useful in early diagnosis and risk stratification. • Microbial-targeted therapies(oral antibiotics, probiotics) – reduce risk in preinvasive disease – adjuvant to standard therapies / synergy with checkpoint directed immunotherapy in invasive disease.

CONCLUSION • Bi-aspect of antibiotics Protumorigenic? • Change of diversity • Additional research – FMT of favorable gut microbiome – Antibiotics : dose, duration of use – Synergistic effect in Cytotoxic chemotherapy?

Thank You
- Slides: 41