CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman

CAMPBELL BIOLOGY TENTH EDITION Reece • Urry • Cain • Wasserman • Minorsky • Jackson 4 Carbon and the Molecular Diversity of Life Clicker Questions by Lisa M. Flick, Ph. D. © 2014 Pearson Education, Inc.

What was the first organic molecule to be synthesized in the laboratory? a) ammonium cyanate b) hydrogen cyanide c) urea d) acetic acid e) methane © 2014 Pearson Education, Inc.

What was the first organic molecule to be synthesized in the laboratory? a) ammonium cyanate b) hydrogen cyanide c) urea d) acetic acid e) methane © 2014 Pearson Education, Inc.

Carbon is an unusual atom in that it can form multiple bonds. Which statement is NOT true? a) A carbon-to-carbon cis double bond is the type found in nature and is associated with cardiovascular health. b) A carbon-to-carbon trans double bond is made artificially in food processing and is associated with poor cardiovascular health. c) Multiple carbon-to-carbon double bonds located near each other can absorb light, so they are found in molecules in the eye or in chloroplasts. d) Multiple carbon-to-carbon bonds are stronger than single bonds. e) Saturated fats are those that have a carbon-to-carbon double bond are associated with good health. © 2014 Pearson Education, Inc.

Carbon is an unusual atom in that it can form multiple bonds. Which statement is NOT true? a) A carbon-to-carbon cis double bond is the type found in nature and is associated with cardiovascular health. b) A carbon-to-carbon trans double bond is made artificially in food processing and is associated with poor cardiovascular health. c) Multiple carbon-to-carbon double bonds located near each other can absorb light, so they are found in molecules in the eye or in chloroplasts. d) Multiple carbon-to-carbon bonds are stronger than single bonds. e) Saturated fats are those that have a carbon-to-carbon double bond are associated with good health. © 2014 Pearson Education, Inc.

What type of chemical bond joins a functional group to the carbon skeleton of a large molecule? a) covalent bond b) hydrogen bond c) ionic bond d) double bond e) disulfide bond © 2014 Pearson Education, Inc.

What type of chemical bond joins a functional group to the carbon skeleton of a large molecule? a) covalent bond b) hydrogen bond c) ionic bond d) double bond e) disulfide bond © 2014 Pearson Education, Inc.

Which of the following is NOT one of the seven functional groups found in biological molecules? a) amino b) hydroxyl c) carboxyl d) cyanate e) phosphate © 2014 Pearson Education, Inc.

Which of the following is NOT one of the seven functional groups found in biological molecules? a) amino b) hydroxyl c) carboxyl d) cyanate e) phosphate © 2014 Pearson Education, Inc.

Which functional group behaves as a weak acid in organic molecules? a) amino b) carboxyl c) carbonyl d) sulfhydryl e) hydroxyl © 2014 Pearson Education, Inc.

Which functional group behaves as a weak acid in organic molecules? a) amino b) carboxyl c) carbonyl d) sulfhydryl e) hydroxyl © 2014 Pearson Education, Inc.

Which functional group behaves as a weak base in organic molecules? a) amino b) carboxyl c) carbonyl d) sulfhydryl e) hydroxyl © 2014 Pearson Education, Inc.

Which functional group behaves as a weak base in organic molecules? a) amino b) carboxyl c) carbonyl d) sulfhydryl e) hydroxyl © 2014 Pearson Education, Inc.

Which type of molecule always contains phosphate groups? a) carbohydrates b) lipids c) proteins d) nucleic acids e) none of the above © 2014 Pearson Education, Inc.

Which type of molecule always contains phosphate groups? a) carbohydrates b) lipids c) proteins d) nucleic acids e) none of the above © 2014 Pearson Education, Inc.
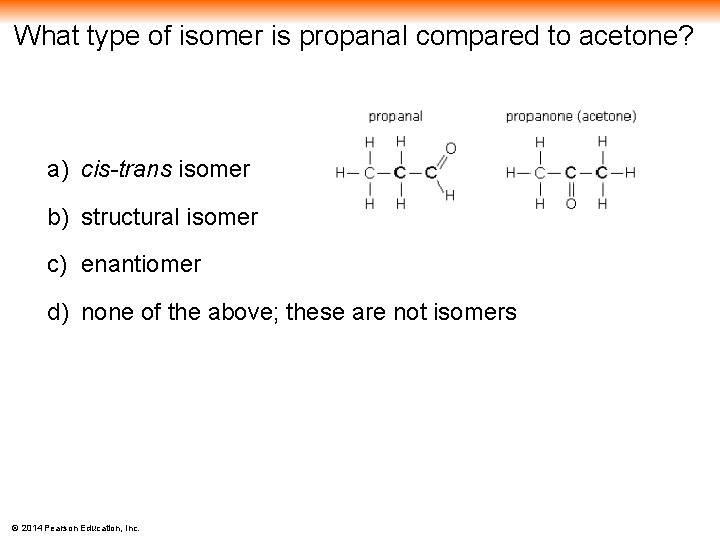
What type of isomer is propanal compared to acetone? a) cis-trans isomer b) structural isomer c) enantiomer d) none of the above; these are not isomers © 2014 Pearson Education, Inc.
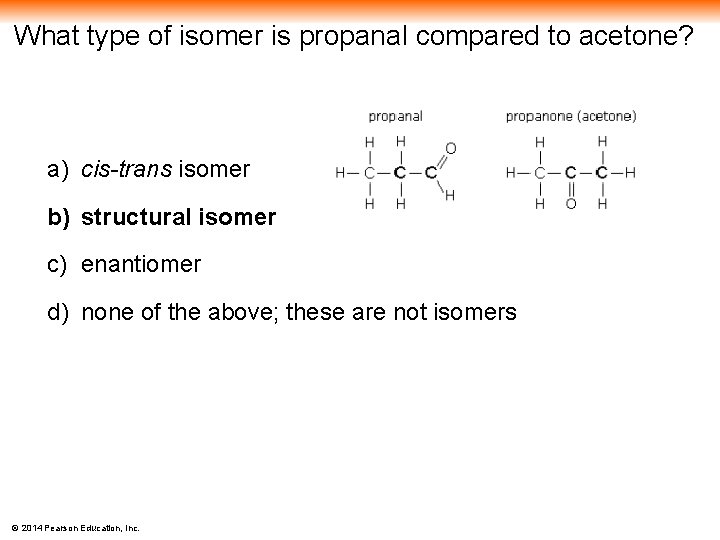
What type of isomer is propanal compared to acetone? a) cis-trans isomer b) structural isomer c) enantiomer d) none of the above; these are not isomers © 2014 Pearson Education, Inc.

Which type of molecule may contain sulfhydryl groups? a) carbohydrate b) lipid c) protein d) nucleic acid e) all of the above © 2014 Pearson Education, Inc.

Which type of molecule may contain sulfhydryl groups? a) carbohydrate b) lipid c) protein d) nucleic acid e) all of the above © 2014 Pearson Education, Inc.

Which functional group is best known for its ability to change the shape of a molecule without affecting its reactivity? a) amino b) carboxyl c) sulfhydryl d) phosphate e) methyl © 2014 Pearson Education, Inc.

Which functional group is best known for its ability to change the shape of a molecule without affecting its reactivity? a) amino b) carboxyl c) sulfhydryl d) phosphate e) methyl © 2014 Pearson Education, Inc.

Scientific Skills Questions © 2014 Pearson Education, Inc.

The table below gives the molar ratios of some of the products from Stanley Miller’s abiotic synthesis of organic molecules experiment. What is the molar ratio of serine? a) 1 mole of serine per mole of glycine b) 3. 0 x 10 2 moles of serine per mole of glycine c) 3. 0 x 10 2 moles of glycine per mole of serine d) 1 mole of serine per 3. 0 x 10 2 moles of glycine © 2014 Pearson Education, Inc.

The table below gives the molar ratios of some of the products from Stanley Miller’s abiotic synthesis of organic molecules experiment. What is the molar ratio of serine? a) 1 mole of serine per mole of glycine b) 3. 0 x 10 2 moles of serine per mole of glycine c) 3. 0 x 10 2 moles of glycine per mole of serine d) 1 mole of serine per 3. 0 x 10 2 moles of glycine © 2014 Pearson Education, Inc.

The table below gives the molar ratios of some of the products from Stanley Miller’s abiotic synthesis of organic molecules experiment. Which amino acid is present in higher amounts than glycine? a) serine b) methionine c) alanine d) serine and methionine © 2014 Pearson Education, Inc.

The table below gives the molar ratios of some of the products from Stanley Miller’s abiotic synthesis of organic molecules experiment. Which amino acid is present in higher amounts than glycine? a) serine b) methionine c) alanine d) serine and methionine © 2014 Pearson Education, Inc.

Based on these results, how many molecules of methionine are present per mole of glycine? a) 1. 08 x 10 70 molecules b) 1. 8 x 10 3 molecules c) 1. 08 x 10 21 molecules d) 6. 02 x 10 23 molecules © 2014 Pearson Education, Inc.

Based on these results, how many molecules of methionine are present per mole of glycine? a) 1. 08 x 10 70 molecules b) 1. 8 x 10 3 molecules c) 1. 08 x 10 21 molecules d) 6. 02 x 10 23 molecules © 2014 Pearson Education, Inc.

The synthetic atmosphere in this experiment contained H 2 S instead of water vapor. Which of these amino acids could not have been produced in Miller’s original abiotic synthesis experiment? a) serine b) methionine c) alanine d) glycine © 2014 Pearson Education, Inc.

The synthetic atmosphere in this experiment contained H 2 S instead of water vapor. Which of these amino acids could not have been produced in Miller’s original abiotic synthesis experiment? a) serine b) methionine c) alanine d) glycine © 2014 Pearson Education, Inc.
- Slides: 30