CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman

CAMPBELL BIOLOGY TENTH EDITION Reece • Urry • Cain • Wasserman • Minorsky • Jackson 2 The Chemical Context of Life Clicker Questions by Tara Stoulig © 2014 Pearson Education, Inc.
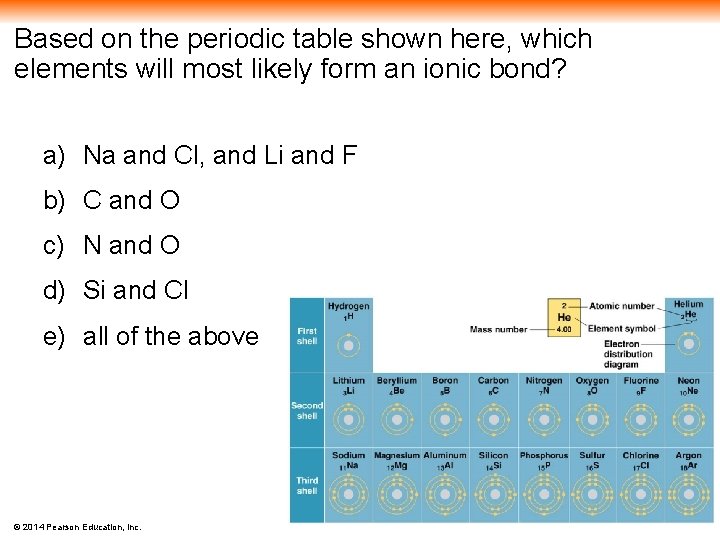
Based on the periodic table shown here, which elements will most likely form an ionic bond? a) Na and Cl, and Li and F b) C and O c) N and O d) Si and Cl e) all of the above © 2014 Pearson Education, Inc.
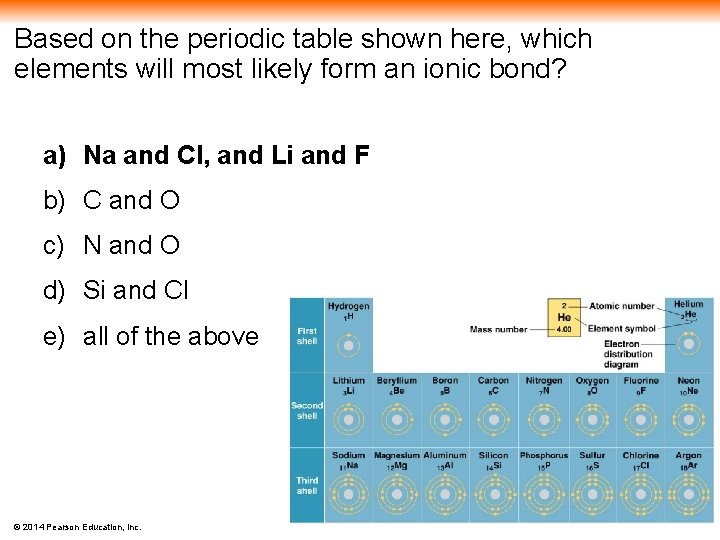
Based on the periodic table shown here, which elements will most likely form an ionic bond? a) Na and Cl, and Li and F b) C and O c) N and O d) Si and Cl e) all of the above © 2014 Pearson Education, Inc.
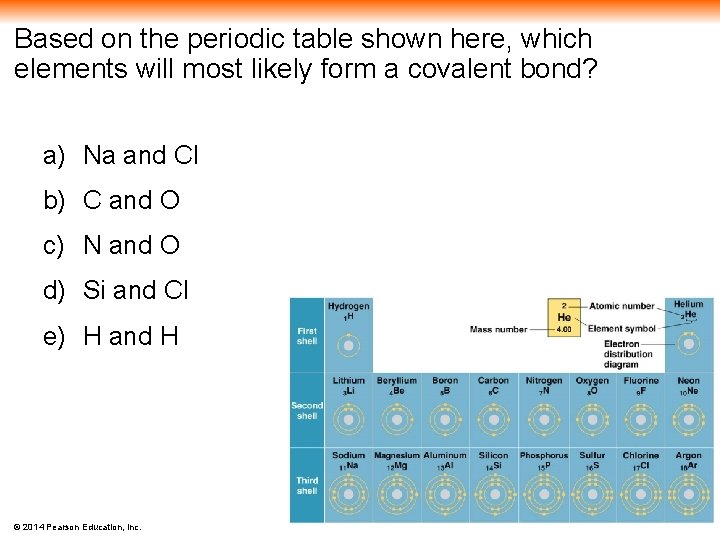
Based on the periodic table shown here, which elements will most likely form a covalent bond? a) Na and Cl b) C and O c) N and O d) Si and Cl e) H and H © 2014 Pearson Education, Inc.
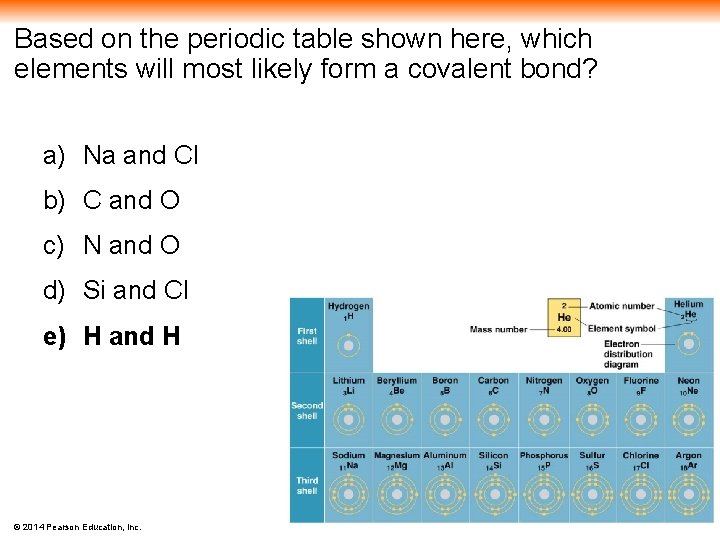
Based on the periodic table shown here, which elements will most likely form a covalent bond? a) Na and Cl b) C and O c) N and O d) Si and Cl e) H and H © 2014 Pearson Education, Inc.

Titanium has an atomic number of 22. How many protons, neutrons, and electrons are in an isotope of titanium with mass number of 48? a) p 22, n 26, e 22 b) p 11, n 26, e 11 c) p 11, n 11, e 70 d) p 11, n 22, e 48 e) p 22, n 22, e 48 © 2014 Pearson Education, Inc.

Titanium has an atomic number of 22. How many protons, neutrons, and electrons are in an isotope of titanium with mass number of 48? a) p 22, n 26, e 22 b) p 11, n 26, e 11 c) p 11, n 11, e 70 d) p 11, n 22, e 48 e) p 22, n 22, e 48 © 2014 Pearson Education, Inc.

H 2 O can be considered ______, but H 2 can only be considered ______. a) an isotope and a molecule; a molecule b) an isotope and a molecule; an isotope c) a compound an isotope; a molecule d) a molecule and a compound; a compound e) a molecule and a compound; a molecule © 2014 Pearson Education, Inc.

H 2 O can be considered ______, but H 2 can only be considered ______. a) an isotope and a molecule; a molecule b) an isotope and a molecule; an isotope c) a compound an isotope; a molecule d) a molecule and a compound; a compound e) a molecule and a compound; a molecule © 2014 Pearson Education, Inc.

What do elements with atomic numbers 6, 14, and 22 have in common? a) same number of electrons b) same atomic mass c) same number of valence electrons and will form the same number of covalent bonds d) all of the above e) none of the above © 2014 Pearson Education, Inc.

What do elements with atomic numbers 6, 14, and 22 have in common? a) same number of electrons b) same atomic mass c) same number of valence electrons and will form the same number of covalent bonds d) all of the above e) none of the above © 2014 Pearson Education, Inc.

What type of bond is very prevalent in lipids and gives lipids their properties? a) polar covalent b) nonpolar covalent c) strong ionic d) weak ionic e) hydrogen © 2014 Pearson Education, Inc.

What type of bond is very prevalent in lipids and gives lipids their properties? a) polar covalent b) nonpolar covalent c) strong ionic d) weak ionic e) hydrogen © 2014 Pearson Education, Inc.

An atom of oxygen has an atomic number of 8. How many electrons are in the first, second, and third electron shells, respectively? a) 2, 3, 3 b) 2, 6, 0 c) 8, 0, 0 d) 0, 4, 4 e) none of the above © 2014 Pearson Education, Inc.

An atom of oxygen has an atomic number of 8. How many electrons are in the first, second, and third electron shells, respectively? a) 2, 3, 3 b) 2, 6, 0 c) 8, 0, 0 d) 0, 4, 4 e) none of the above © 2014 Pearson Education, Inc.

What numbers must be placed as coefficients in the blanks for the chemical reaction below in order to ensure that matter is conserved? Fe 3 O 4 __C __Fe __CO a) 1; 1; 1 b) 4; 3; 4 c) 2; 1; 2 d) 3; 4; 3 e) 0; 3; 4 © 2014 Pearson Education, Inc.

What numbers must be placed as coefficients in the blanks for the chemical reaction below in order to ensure that matter is conserved? Fe 3 O 4 __C __Fe __CO a) 1; 1; 1 b) 4; 3; 4 c) 2; 1; 2 d) 3; 4; 3 e) 0; 3; 4 © 2014 Pearson Education, Inc.

Of the four most commonly found elements in the human body, which has the most valence electrons? a) O b) C c) H d) N © 2014 Pearson Education, Inc.

Of the four most commonly found elements in the human body, which has the most valence electrons? a) O b) C c) H d) N © 2014 Pearson Education, Inc.

Lithium and fluorine are most likely to form which type of bond? a) polar covalent b) nonpolar covalent c) ionic d) hydrogen e) A and B are equally likely © 2014 Pearson Education, Inc.

Lithium and fluorine are most likely to form which type of bond? a) polar covalent b) nonpolar covalent c) ionic d) hydrogen e) A and B are equally likely © 2014 Pearson Education, Inc.

Carbon-14 dating works for fossils up to about 75, 000 years old. Most dinosaurs went extinct 65. 5 million years ago. Can 14 C be used to date dinosaur bones? a) Yes; the bones continued to take in 14 C, even after the dinosaur died. b) No; the 14 C present in the dinosaur when it died would decay too much to be measured after 65. 5 million years. c) No; 14 C can only be used to date dinosaur teeth, which are much stronger than bones. d) Yes; the bones contained 14 C when the dinosaur died so it can be measured to determine the fossil’s age. © 2014 Pearson Education, Inc.

Carbon-14 dating works for fossils up to about 75, 000 years old. Most dinosaurs went extinct 65. 5 million years ago. Can 14 C be used to date dinosaur bones? a) Yes; the bones continued to take in 14 C, even after the dinosaur died. b) No; the 14 C present in the dinosaur when it died would decay too much to be measured after 65. 5 million years. c) No; 14 C can only be used to date dinosaur teeth, which are much stronger than bones. d) Yes; the bones contained 14 C when the dinosaur died so it can be measured to determine the fossil’s age. © 2014 Pearson Education, Inc.

Radioactive uranium-235 has a half-life of 704 million years. If it was incorporated into dinosaur bones, could it be used to date the dinosaur fossils? a) Yes; after 65. 5 million years only about one-tenth of the 235 U would have decayed, leaving plenty to measure in the fossils. b) No; the dinosaurs went extinct too recently to use a radioisotope with a half-life of 704 million years. c) No; only about one tenth of the 235 U would have decayed after 65. 5 million years, not leaving enough to measure in the fossils. d) Yes; but only for dinosaurs that lived more than 704 million years ago. © 2014 Pearson Education, Inc.

Radioactive uranium-235 has a half-life of 704 million years. If it was incorporated into dinosaur bones, could it be used to date the dinosaur fossils? a) Yes; after 65. 5 million years only about one-tenth of the 235 U would have decayed, leaving plenty to measure in the fossils. b) No; the dinosaurs went extinct too recently to use a radioisotope with a half-life of 704 million years. c) No; only about one tenth of the 235 U would have decayed after 65. 5 million years, not leaving enough to measure in the fossils. d) Yes; but only for dinosaurs that lived more than 704 million years ago. © 2014 Pearson Education, Inc.
- Slides: 25