CALYPSO Trial Carboplatin Pegylated Liposomal Doxorubicin PLD versus

CALYPSO Trial: Carboplatin & Pegylated Liposomal Doxorubicin (PLD) versus Carboplatin & Paclitaxel in Relapsed, Platinum. Sensitive Ovarian Cancer Pujade-Lauraine E et al. ASCO 2009; Abstract LBA 5509. (Oral Presentation)

Introduction l Choice of treatment for relapsed ovarian cancer is dependent upon the interval since prior platinum-based therapy – Relapse within six months: platinum-resistant disease – Relapse > six months: platinum-sensitive disease l Carboplatin/paclitaxel (CP) is a standard treatment in platinum-sensitive, relapsed ovarian cancer l Due to risk of cumulative neuropathy and hair loss, other carboplatin-based combinations are needed l Pegylated liposomal doxorubicin (PLD) is a rationale partner for carboplatin – PLD resulted in significant improvements in PFS and OS compared to topotecan in platinum-sensitive ovarian cancer (JCO 2001; 19: 3312) – Safety and efficacy (median PFS: 9. 4 months) of carboplatin/PLD was demonstrated in a phase II trial (Ann Oncol 2007; 18: 263) l Current study objectives: – Evaluate PFS and safety of carboplatin/PLD (CD) versus CP in patients with platinum-sensitive, relapsed ovarian cancer Source: Pujade-Lauraine E et al. ASCO 2009; Abstract LBA 5509.
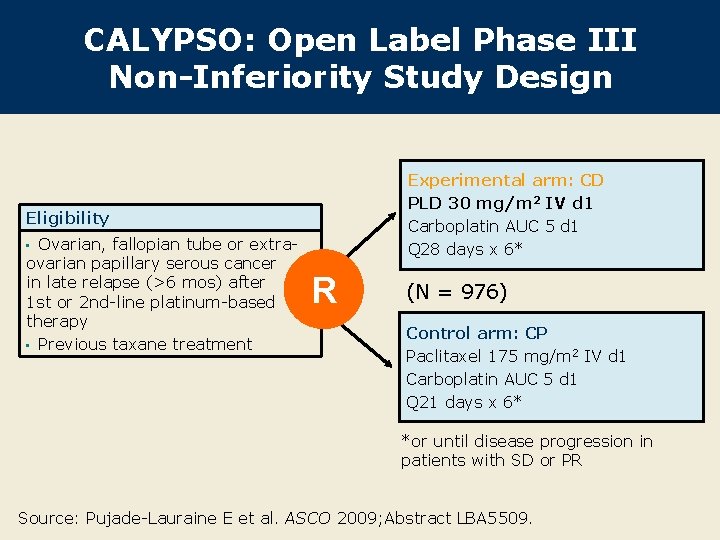
CALYPSO: Open Label Phase III Non-Inferiority Study Design Experimental arm: CD PLD 30 mg/m 2 IV d 1 Carboplatin AUC 5 d 1 Q 28 days x 6* Eligibility Ovarian, fallopian tube or extraovarian papillary serous cancer in late relapse (>6 mos) after 1 st or 2 nd-line platinum-based therapy • Previous taxane treatment • R (N = 976) Control arm: CP Paclitaxel 175 mg/m 2 IV d 1 Carboplatin AUC 5 d 1 Q 21 days x 6* *or until disease progression in patients with SD or PR Source: Pujade-Lauraine E et al. ASCO 2009; Abstract LBA 5509.
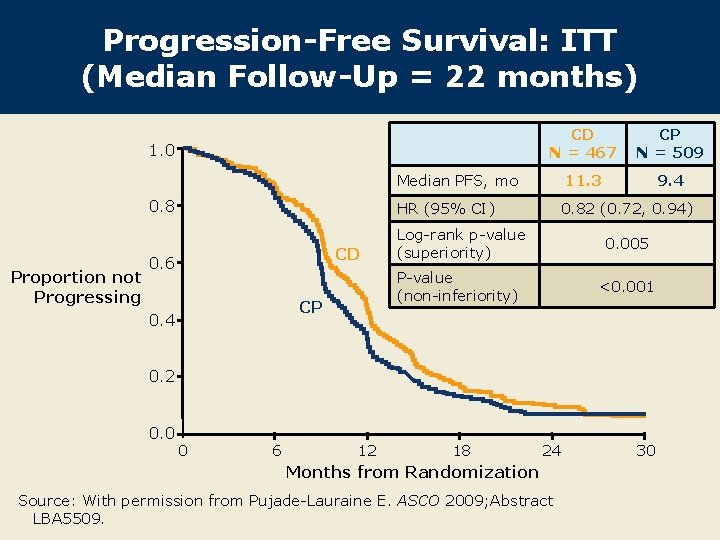
Progression-Free Survival: ITT (Median Follow-Up = 22 months) CD N = 467 CP N = 509 Median PFS, mo 11. 3 9. 4 HR (95% CI) 0. 82 (0. 72, 0. 94) 1. 0 0. 8 Proportion not Progressing CD 0. 6 CP 0. 4 Log-rank p-value (superiority) 0. 005 P-value (non-inferiority) <0. 001 0. 2 0. 0 0 6 12 18 24 Months from Randomization Source: With permission from Pujade-Lauraine E. ASCO 2009; Abstract LBA 5509. 30
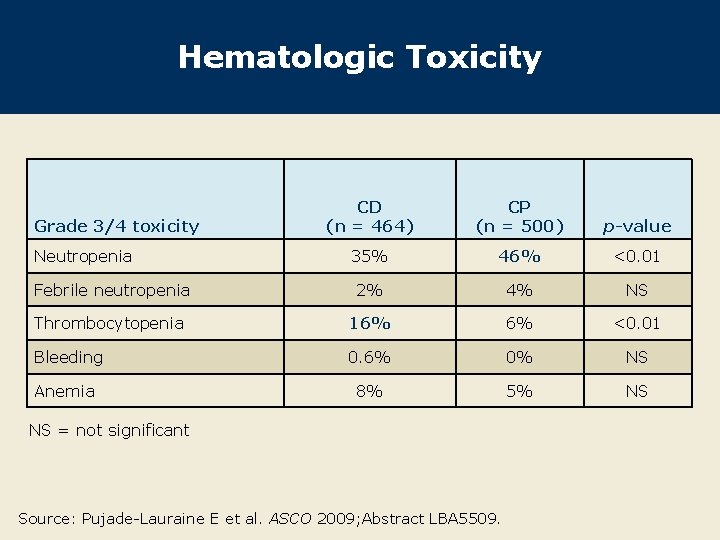
Hematologic Toxicity CD (n = 464) CP (n = 500) p-value 35% 46% <0. 01 Febrile neutropenia 2% 4% NS Thrombocytopenia 16% 6% <0. 01 Bleeding 0. 6% 0% NS Anemia 8% 5% NS Grade 3/4 toxicity Neutropenia NS = not significant Source: Pujade-Lauraine E et al. ASCO 2009; Abstract LBA 5509.
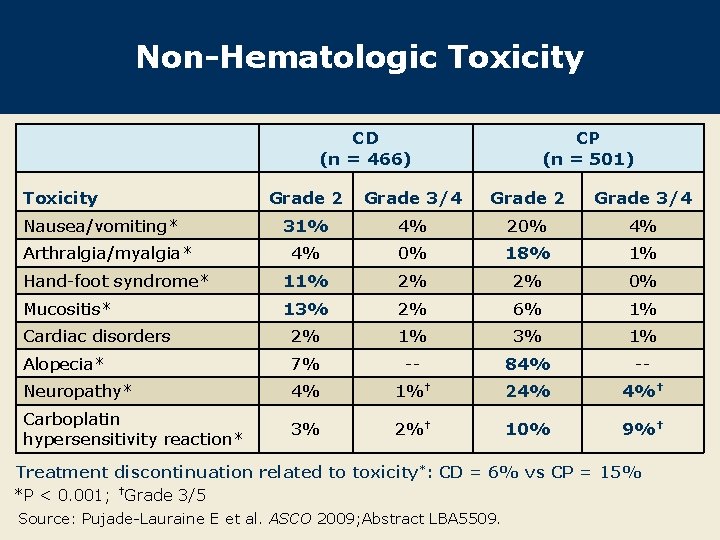
Non-Hematologic Toxicity CD (n = 466) Toxicity CP (n = 501) Grade 2 Grade 3/4 31% 4% 20% 4% 4% 0% 18% 1% Hand-foot syndrome* 11% 2% 2% 0% Mucositis* 13% 2% 6% 1% Cardiac disorders 2% 1% 3% 1% Alopecia* 7% -- 84% -- Neuropathy* 4% 1%† 24% 4%† Carboplatin hypersensitivity reaction* 3% 2%† 10% 9%† Nausea/vomiting* Arthralgia/myalgia* Treatment discontinuation related to toxicity*: CD = 6% vs CP = 15% *P < 0. 001; †Grade 3/5 Source: Pujade-Lauraine E et al. ASCO 2009; Abstract LBA 5509.
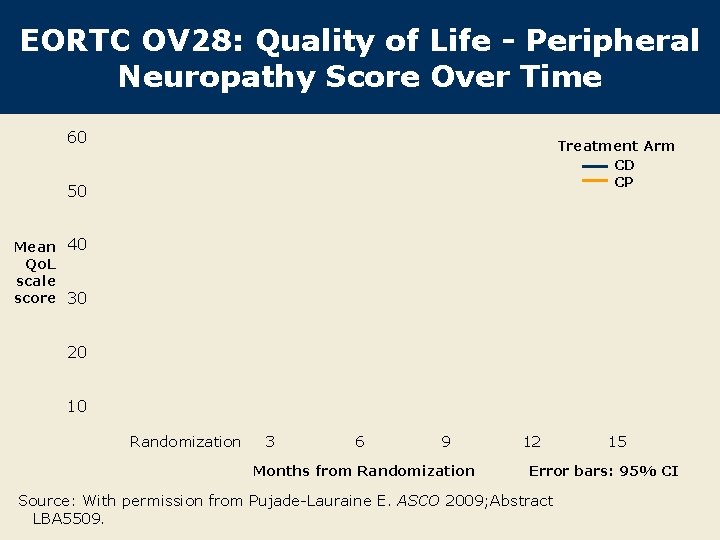
EORTC OV 28: Quality of Life - Peripheral Neuropathy Score Over Time 60 Treatment Arm CD CP 50 Mean 40 Qo. L scale score 30 20 10 Randomization 3 6 9 Months from Randomization 12 15 Error bars: 95% CI Source: With permission from Pujade-Lauraine E. ASCO 2009; Abstract LBA 5509.

Summary and Conclusions l In relapsed, platinum-sensitive ovarian cancer carboplatin/pegylated liposomal doxorubicin (CD) was non-inferior to CP – 18% reduction in risk of progression (HR = 0. 82, p = 0. 005) l Overall survival data is immature l CP associated with more severe toxicity (carboplatin hypersensitivity), alopecia and long-lasting toxicity (neuropathy) — more toxicity-related treatment discontinuation – CP = 15% vs CD = 6% (p < 0. 001) l CD associated with moderate, reversible hand-foot syndrome, more mucositis and more nausea/vomiting l Overall, CD demonstrated a superior therapeutic index (benefit/risk ratio) versus the current standard CP l CD offers an evidence-based option for patients with relapsed, platinumsensitive ovarian cancer Source: Pujade-Lauraine E et al. ASCO 2009; Abstract LBA 5509.
- Slides: 8