CALORIMETRY ENTHALPY CHANGES be able to define enthalpy

CALORIMETRY

ENTHALPY CHANGES be able to define enthalpy and enthalpy change (ΔH) be able to define standard enthalpy chemical reactions involve the making and breaking of bonds be able to determine bond enthalpies from data use mean bond enthalpies to estimate ΔH for reactions be able to calculate enthalpy change from experimental data use Hess’s law to calculate enthalpy changes in reactions
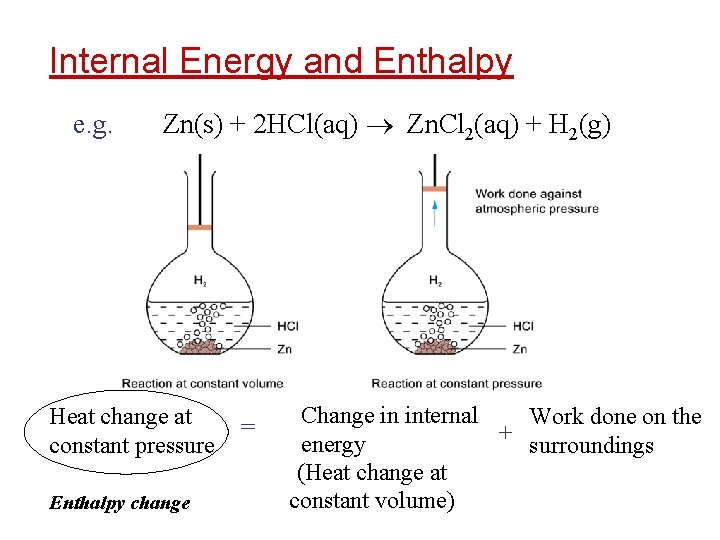
Internal Energy and Enthalpy e. g. Zn(s) + 2 HCl(aq) Zn. Cl 2(aq) + H 2(g) Heat change at constant pressure Enthalpy change = Change in internal Work done on the + energy surroundings (Heat change at constant volume)

First Law THERMODYNAMICS Energy can be neither created nor destroyed but It can be converted from one form to another Second Law Energy flows from hot to cold Third Law A perfectly ordered crystal has zero entropy. Chemical reactions either absorb or give off energy Exothermic Energy is given out Endothermic Energy is absorbed Every Day Examples Exothermic combustion of fuels respiration (oxidation of carbohydrates) Endothermic photosynthesis thermal decomposition of calcium carbonate
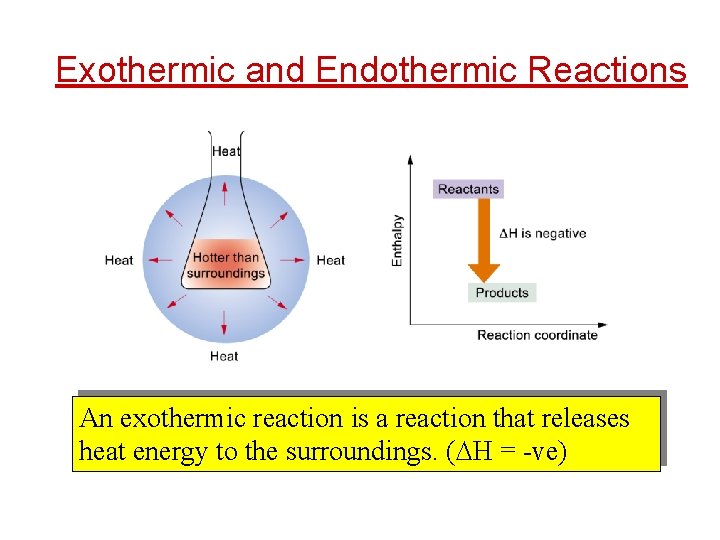
Exothermic and Endothermic Reactions An exothermic reaction is a reaction that releases heat energy to the surroundings. ( H = -ve)
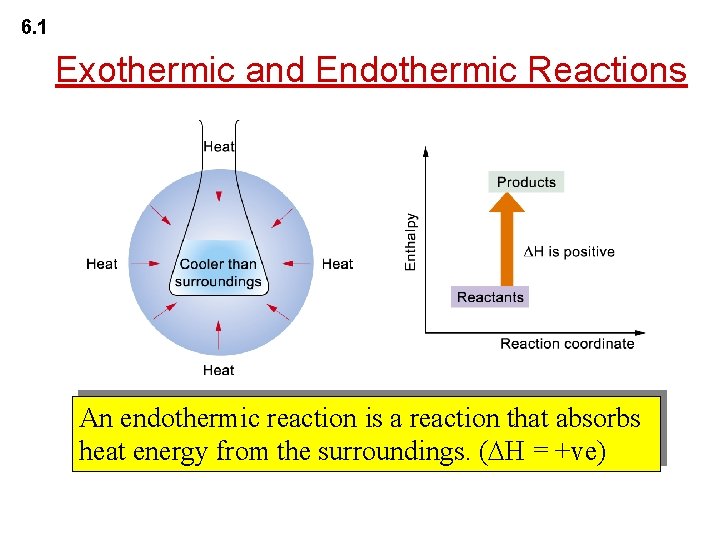
6. 1 Exothermic and Endothermic Reactions An endothermic reaction is a reaction that absorbs heat energy from the surroundings. ( H = +ve)

Calorimetry Fuse school ( calorimeters and heats of combustion ) https: //www. youtube. com/watch? v=if. Ttdv. F 98 T 8

Introduction to Thermodynamics is the study of the interconversion of heat and other kinds of energy. In thermodynamics, there are three types of systems: An open system can exchange mass and energy with the surroundings. A closed system allows the transfer of energy but not mass. An isolated system does not exchange either mass or energy with its surroundings.

1 g of water 1 ºC hotter Energy required = 4. 18 J 1 g of water 2 ºC hotter Energy required = 2 x 4. 18 J = 8. 36 J

1 g of water 1 ºC hotter Energy required = 4. 18 J 5 g of water 1 ºC hotter Energy required = 5 x 4. 18 J = 20. 9 J

1 g of water 1 ºC hotter Energy required = 4. 18 J 3 g of water 10 ºC hotter Energy required = 3 x 10 x 4. 18 = 125. 4 J

1 g of copper 1 ºC hotter Energy required = 0. 39 J 10 g of copper 5 ºC hotter Energy required = 10 x 5 x 0. 39 = 19. 5 J
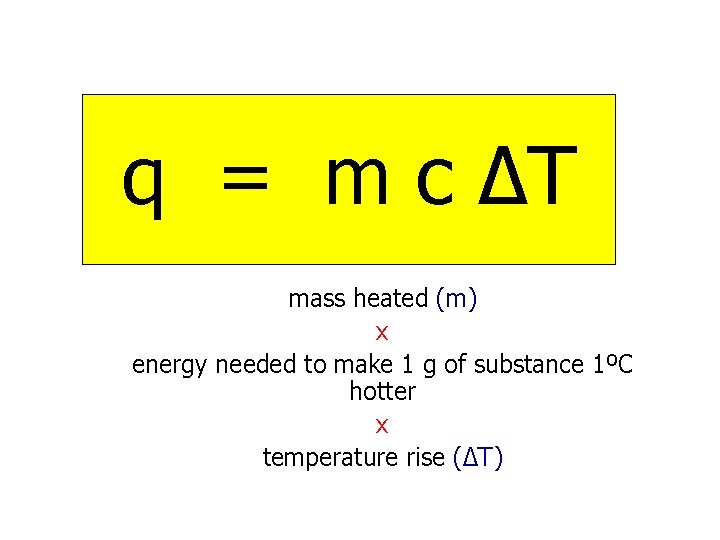
q = m c ∆T mass heated (m) x energy needed to make 1 g of substance 1ºC hotter x temperature rise (∆T)

Calculate the heat to take 255 g of water from 25. 2 C to 90. 5 C. Strategy Get the givens c = 4. 184 J/g∙°C, m = 255 g, ΔT = 90. 5°C – 25. 2°C = 65. 3°C. Formula q=mc ΔT Solution q = 255 g × 4. 184 J/g∙°C x 65. 3°C q= 69669. 876 J / 1000 J/k. J q= + 69. 7 k. J (3 figs) the heat is going INTO the water, so it is +ve for the water This is heat that was GIVEN to the water, but FROM where. A chemical must have given it’s heat away so it is –VE for the chemical – 96. 7 k. J NOW, molar heat

2 moles of ethanol burned Energy given out = 2734 k. J Energy given out by 1 mole = 2734 k. J 2 = 1367 k. J -1 – 1367 k. J mol ∆H =

q ∆H = moles in reaction

Experimental Determination of Enthalpy Changes by Calorimetry Calorimeter = a container used for measuring the temperature change of solution Determination of Enthalpy Change of Neutralization
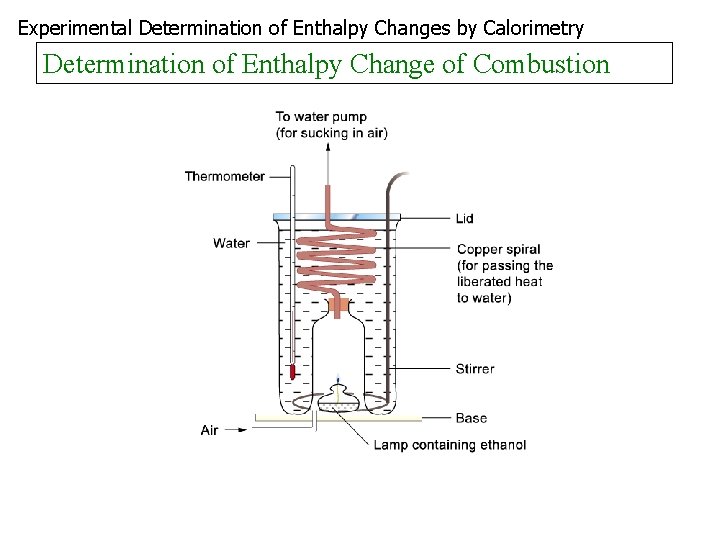
Experimental Determination of Enthalpy Changes by Calorimetry Determination of Enthalpy Change of Combustion

Cal Problems worked 4 min https: //www. youtube. com/watc h? v=Hs 5 x 0 -IU 2 F 4

Excess powdered zinc was added to 100 ml of 0. 2 mol/L copper (II) sulphate solution. A temperature rise of 10 o. C was recorded. Find the MOLAR enthalpy change for the reaction. H = m x c x T H = 100 g x 4. 18 KJ kg-1 K-1 x 10 o. C 1000 = 4. 180 KJ This is for the no of moles of Cu. SO 4 used in the experiment No of moles of Cu. SO 4 = 0. 2 x 100 = 0. 02 moles 1000 H = 4. 180 = 209 KJ mol-1 0. 02 The reaction is exothermic so we need to put in a negative sign Hr = - 209 KJ mol-1 of Cu. SO 4

Combustion To find the heat of combustion of a substance a known mass of the substance is burned, the heat released transferred to water and the enthalpy change found as before In an experiment to find the heat of combustion of ethanol the following results were obtained Initial mass of lamp + ethanol = 65. 20 g Final mass of lamp = 64. 28 g Final temperature of water Initial temperature of water = = 47. 1 o. C 28. 5 o. C Mass of the water = 300 g What are the products of complete combustion of ethanol? What mass of ethanol was burnt? How many moles is this? What quantity of heat was transferred to the water? Find Hc of ethanol Identify any sources of error Is ethanol a good fuel?

C 2 H 5 OH + 3 O 2 2 CO 2 + 3 H 2 O q=mc∆t H = 300 x 4. 18 KJ kg-1 K-1 x 18. 6 K = 23. 3 KJ 1000 H lost = h gained (so 23. 3 KJ gained by calorimeter was lost in burning) Mass of ethanol used = 0. 92 g = 0. 92 g = 0. 020 mol 46 g/mol H=mc∆t / n Hc = - 23. 3 = -1160 KJ/mol 0. 020

Errors Heat lost to surroundings (air, can thermometer) Errors in measuring temperature change (unavoidable error in reading thermometer) Errors in measuring masses (unavoidable error in reading balance) The enthalpy change of combustion is high ethanol is a good fuel

Chemistry in Action: Fuel Values of Foods and Other Substances C 6 H 12 O 6 (s) + 6 O 2 (g) 6 CO 2 (g) + 6 H 2 O (l) H = -2820 k. J/mol Per gram Sugar = 2999 KJ / 180 g = 15. 7 KJ/gram 1 cal = 4. 184 J 1 Cal = 1000 cal = 4184 J =4. 184 KJ If 1. 0 gram sugar is = 15. 7 KJ Or 15. 7 KJ / 4. 184 KJ/Cal = 3. 75 Cal So sugar burned is approx 4 Cal/g Here 26 g x 4 Cal/g = 104 Cal from sugar

Foods and Fuels Cheetos burned v Cereal https: //www. youtube. com/watch? v=UDgea. AM d. YIY Foods • 1 nutritional Calorie, 1 Cal = 1000 cal = 1 kcal. • Energy in us comes from carbohydrates and fats TED X (3 min, Calories) https: //www. youtube. com/watch? v=VEQa. H 4 Lru. Uo • Intestines: carbohydrates converted into glucose: C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O, H = -2816 k. J • Fats break down as follows: 2 C 57 H 110 O 6 + 163 O 2 114 CO 2 + 110 H 2 O, H = -75, 520 k. J Fats contain more energy; are not water soluble, so are good for energy storage.

1) In an experiment, 1. 00 g of propanone (CH 3 COCH 3) was completely burned in air. The heat evolved raised the temperature of 150 g of water from 18. 8 C to 64. 3 C. Use this data to calculate the enthalpy of combustion of propanone (the specific heat capacity of water is 4. 18 J g 1 K-1). q = mc∆T m = 150 c = 4. 18 ∆T = 45. 5 q = 150 x 4. 18 x 45. 5 = 28530 J ∆H = q / moles of propane = mass / Mr = 1. 00 / 58. 0 = 0. 01724 ∆H = – 28. 53 / 0. 01724 = -1650 k. J mol-1 (3 sig fig)

2) In an experiment, 1. 00 g of hexane (C 6 H 14) was completely burned in air. The heat evolved raised the temperature of 200 g of water from 293. 5 K to 345. 1 K. Use this data to calculate the enthalpy of combustion of hexane (the specific heat capacity of water is 4. 18 J g-1 K-1). q = mc∆T m = 200 c = 4. 18 ∆T = 51. 6 q = 200 x 4. 18 x 51. 6 = 43140 J ∆H = q / moles of propane = mass / Mr = 1. 00 / 86. 0 = 0. 01163 ∆H = – 43. 14 / 0. 01163 = -3710 k. J mol-1 (3 sig fig)

3) In an experiment, 1. 56 g of propan-1 -ol (CH 3 CH 2 OH) was completely burned in air. The heat evolved raised the temperature of 0. 250 dm 3 of water from 292. 1 K to 339. 4 K. Use this data to calculate the enthalpy of combustion of propan-1 -ol (the specific heat capacity of water is 4. 18 J g -1 K-1). q = mc∆T m = 250 c = 4. 18 ∆T = 47. 3 q = 250 x 4. 18 x 47. 3 = 49430 J ∆H = q / moles of propane = mass / Mr = 1. 56 / 60. 0 = 0. 02600 ∆H = – 49. 43 / 0. 02600 = -1900 k. J mol-1 (3 sig fig)

4) 25 cm 3 of 2. 0 mol dm-3 nitric acid was added to 25 cm 3 of 2. 0 mol dm-3 potassium hydroxide solution. The temperature rose by 13. 7 C. Calculate the enthalpy of neutralisation for this reaction. Assume that the density of the solution is 1. 00 g cm-3, the specific heat capacity of the solution is 4. 18 J g-1 K-1. q = mc∆T m = 50 c = 4. 18 ∆T = 13. 7 q = 50 x 4. 18 x 13. 7 = 2863 J ∆H = q / mol Mol HNO 3 = conc x vol = 2. 0 x 25/1000 = 0. 050 Mol KOH = conc x vol = 2. 0 x 25/1000 = 0. 050 HNO 3 + KOH → Na. NO 3 + H 2 O ∆H = – 2. 863 / 0. 050 = -57. 3 k. J mol-1 (3 sig fig)

5) 50 cm 3 of 0. 10 mol dm-3 silver nitrate solution was put in a calorimeter and 0. 2 g of zinc powder added. The temperature of the solution rose by 4. 3 C. Work out which reagent was in excess and then calculate the enthalpy change for the reaction (per mole Limiting reagent. Assume that the density of the solution is 1. 00 g cm-3, the specific heat capacity of the solution is 4. 18 J g-1 K-1. Ignore the heat capacity of the metals. q = mc∆T m = 50 c = 4. 18 ∆T = 4. 3 q = 50 x 4. 18 x 4. 3 = 898. 7 J ∆H = q / mol Mol Ag. NO 3 = conc x vol = 0. 1 x 50/1000 = 0. 0050 Mol Zn = mass / Mr = 0. 2 / 65. 4 = 0. 00306 XS 2 Ag. NO 3 + Zn → Zn. NO 3 + 2 Ag ∆H = – 0. 8987 k. J / 0. 0050 Ag. NO 3 = -180 k. J mol-1 (3 sig fig)

EXTRA PROBLEMS

1) In an experiment, 0. 600 g of propane (C 3 H 8) was completely burned in air. The heat evolved raised the temperature of 100 g of water by 64. 9 C. Use this data to calculate the enthalpy of combustion of propane (the specific heat capacity of water is 4. 18 J g-1 K-1). q = mc∆T m = 100 c = 4. 18 ∆T = 64. 9 q = 100 x 4. 18 x 64. 9 = 27130 J ∆H = q / moles of propane = mass / Mr = 0. 600 / 44. 0 = 0. 01364 ∆H = – 27. 13 / 0. 01364 = -1990 k. J mol-1 (3 sig fig)

2) 50 cm 3 of 1. 0 mol dm-3 hydrochloric acid was added to 50 cm 3 of 1. 0 mol dm-3 sodium hydroxide solution. The temperature rose by 6. 8 C. Calculate the enthalpy of neutralisation for this reaction. Assume that the density of the solution is 1. 00 g cm-3, the specific heat capacity of the solution is 4. 18 J g-1 K-1. q = mc∆T m = 100 c = 4. 18 ∆T = 6. 8 q = 100 x 4. 18 x 6. 8 = 2842 J ∆H = q / mol Mol HCl = conc x vol = 1. 0 x 50/1000 = 0. 050 Mol Na. OH = conc x vol = 1. 0 x 50/1000 = 0. 050 HCl + Na. OH → Na. Cl + H 2 O ∆H = – 2. 842 / 0. 050 = -57 k. J mol-1 (2 sig fig)

3) 100 cm 3 of 0. 200 mol dm-3 copper sulphate solution was put in a calorimeter and 2. 00 g of magnesium powder added. The temperature of the solution rose by 25. 1 C. Work out which reagent was in excess and then calculate the enthalpy change for the reaction. Assume that the density of the solution is 1. 00 g cm-3, the specific heat capacity of the solution is 4. 18 J g-1 K-1. Ignore the heat capacity of the metals. q = mc∆T m = 100 c = 4. 18 ∆T = 25. 1 q = 100 x 4. 18 x 25. 1 = 10490 J ∆H = q / mol Mol Cu. SO 4 = conc x vol = 0. 200 x 100/1000 = 0. 020 Mol Mg = mass / Mr = 2. 00 / 24. 3 = 0. 0823 XS Cu. SO 4 + Mg → Mg. SO 4 + Cu ∆H = – 10. 49 / 0. 020 = -525 k. J mol-1 (3 sig fig)

4) 50 cm 3 of 2. 0 mol dm-3 hydrochloric acid was added to 50 cm 3 of 2. 0 mol dm-3 ammonia solution. The temperature rose by 12. 4 C. Calculate the enthalpy of neutralisation for this reaction. Assume that the density of the solution is 1. 00 g cm-3, the specific heat capacity of the solution is 4. 18 J g-1 K-1. q = mc∆T m = 100 c = 4. 18 ∆T = 12. 4 q = 100 x 4. 18 x 12. 4 = 5183 J ∆H = q / mol Mol HCl = conc x vol = 2. 0 x 50/1000 = 0. 100 Mol NH 3 = conc x vol = 2. 0 x 50/1000 = 0. 100 HCl + NH 3 → NH 4 Cl ∆H = – 5. 183 / 0. 100 = -51. 8 k. J mol-1 (3 sig fig)

5) 50 cm 3 of 1. 0 mol dm-3 nitric acid was added to 20 cm 3 of 1. 0 mol dm-3 barium hydroxide solution. The temperature rose by 7. 9 C. Calculate the enthalpy of neutralisation for this reaction (per mole of nitric acid reacting). Assume that the density of the solution is 1. 00 g cm-3, the specific heat capacity of the solution is 4. 18 J g-1 K-1. q = mc∆T m = 70 c = 4. 18 ∆T = 7. 9 q = 70 x 4. 18 x 7. 9 = 2312 J ∆H = q / mol Mol HNO 3 = conc x vol = 1. 0 x 50/1000 = 0. 050 XS Mol Ba(OH)2 = conc x vol = 1. 0 x 20/1000 = 0. 020 2 HNO 3 + Ba(OH)2 → Ba(NO 3)2 + 2 H 2 O ∆H = – 2. 312 / 0. 040 = -57. 8 k. J mol-1 (3 sig fig)

6) 25 cm 3 of 1. 00 mol dm-3 copper sulphate solution was put in a calorimeter and 6. 0 g of zinc powder added. The temperature of the solution rose by 50. 6 C. Work out which reagent was in excess and then calculate the enthalpy change for the reaction. Assume that the density of the solution is 1. 00 g cm-3, the specific heat capacity of the solution is 4. 18 J g-1 K-1. Ignore the heat capacity of the metals. q = mc∆T m = 25 c = 4. 18 ∆T = 50. 6 q = 25 x 4. 18 x 50. 6 = 5288 J ∆H = q / mol Mol Cu. SO 4 = conc x vol = 1. 0 x 25/1000 = 0. 025 Mol Zn = mass / Mr = 6. 0 / 65. 4 = 0. 0917 XS Cu. SO 4 + Zn → Zn. SO 4 + Cu ∆H = – 5. 288 / 0. 025 = -212 k. J mol-1 (3 sig fig)

7) 3. 53 g of sodium hydrogencarbonate was added to 30. 0 cm 3 of 2. 0 mol dm-3 hydrochloric acid. The temperature fell by 10. 3 K. Work out which reagent was in excess and then calculate the enthalpy change for the reaction. Assume that the density of the solution is 1. 00 g cm-3, the specific heat capacity of the solution is 4. 18 J g-1 K-1. q = mc∆T m = 30 c = 4. 18 ∆T = 10. 3 q = 30 x 4. 18 x 10. 3 = 1292 J ∆H = q / mol Mol HCl = conc x vol = 2. 0 x 30/1000 = 0. 060 XS Mol Na. HCO 3 = mass / Mr = 3. 53 / 84. 0 = 0. 0420 HCl + Na. HCO 3 → Na. Cl + H 2 O + CO 2 ∆H = 1. 292 / 0. 0420 = +30. 8 k. J mol-1 (3 sig fig)

8) A calorimeter was calibrated by burning 2. 00 g of methanol (CH 3 OH) whose enthalpy of combustion is -715 k. J mol-1. The temperature of the calorimeter rose from 19. 6 C to 52. 4 C. The same calorimeter was used to measure the enthalpy of combustion of propan-2 -ol. 1. 50 g of propan-2 -ol CH 3 CH(OH)CH 3 raised the temperature by from 19. 8 C to 56. 2 C. Calculate the heat capacity of the calorimeter and then the enthalpy of combustion of propan-2 -ol. ∆H = q / mol q = ∆H x mol Mol CH 3 OH = mass / Mr = 2. 00 / 32. 0 = 0. 0625 q = ∆H x mol = 715 x 0. 0625 = 44. 69 k. J q = mc∆T mc = q/∆T = 44. 69 / 32. 8 = 1. 363 k. J K-1

9) A calorimeter was calibrated by burning 2. 00 g of methanol (CH 3 OH) whose enthalpy of combustion is -715 k. J mol-1. The temperature of the calorimeter rose from 19. 6 C to 52. 4 C. The same calorimeter was used to measure the enthalpy of combustion of propan-2 -ol. 1. 50 g of propan-2 -ol CH 3 CH(OH)CH 3 raised the temperature by from 19. 8 C to 56. 2 C. Calculate the heat capacity of the calorimeter and then the enthalpy of combustion of propan-2 -ol. q = mc∆T mc = 1. 363 ∆T = 36. 4 q = 1. 363 x 36. 4 = 49. 61 k. J ∆H = q / mol Mol C 3 H 7 OH = mass / Mr = 1. 50 / 60. 0 = 0. 0250 ∆H = – 49. 61 / 0. 0250 = -1984 k. J mol-1 (3 sig fig)

PRACTICE enthalpy change of solution ΔHS Enthalpy change of solution ΔHS – is when 1 mole of substance is dissolved 1. Place a clean, dry polystyrene cup into an outer plastic cup to increase insulation and stability. 2. Use the measuring cylinder to transfer 25 cm 3 of water into the polystyrene cup. 3. Place a thermometer into the cup through a cardboard lid and record this temperature as an initial temperature 4. Weight 1 g NH 4 Cl of and put it into polystyrene cup. 5. Measure the temperature of the mixture in the cup and record the greatest temperature change that you observed as a final temperature; 6. Rinse the plastic cup with water and shake to dry. 7. Repeat same steps with Na. OH (keep this solution for next experiment) 8. Calculate the enthalpy change for both using the equation q = mcΔT NH 4 Cl Mass (g) Volume of water is measured (cm 3) Initial temperature (K) Final temperature (K) ΔH ( k. Jmol-1 ) Na. OH

PRACTICE enthalpy change of neutralisation ΔHN The standard enthalpy change of neutralisation is the enthalpy change when solutions of an acid an alkali react together under standard conditions to produce 1 mole of water. 1. Place a thermometer into the cup with Na. OH through a cardboard lid and record this temperature as an initial temperature; 2. Use the measuring cylinder to transfer 25 cm 3 of HCl into the polystyrene cup. 3. Measure the temperature of the mixture in the cup immediately record the greatest temperature change that you observed as a final temperature; 4. Calculate the enthalpy change for reaction using the equation q = mcΔT
- Slides: 42