Calorimetry Burning of a Match Potential energy System

Calorimetry
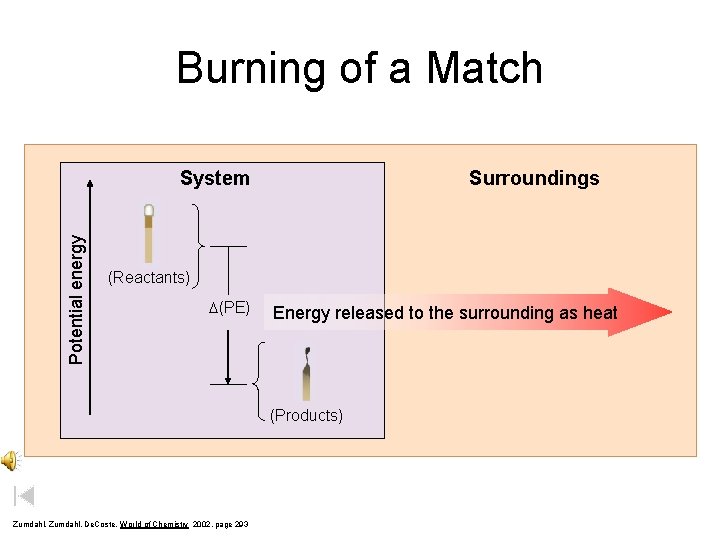
Burning of a Match Potential energy System Surroundings (Reactants) D(PE) Energy released to the surrounding as heat (Products) Zumdahl, De. Coste, World of Chemistry 2002, page 293
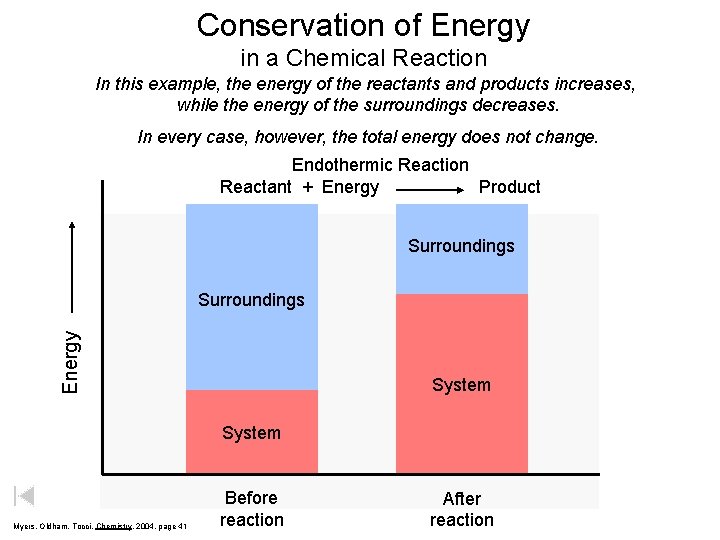
Conservation of Energy in a Chemical Reaction In this example, the energy of the reactants and products increases, while the energy of the surroundings decreases. In every case, however, the total energy does not change. Endothermic Reaction Reactant + Energy Product Surroundings Energy Surroundings System Myers, Oldham, Tocci, Chemistry, 2004, page 41 Before reaction After reaction
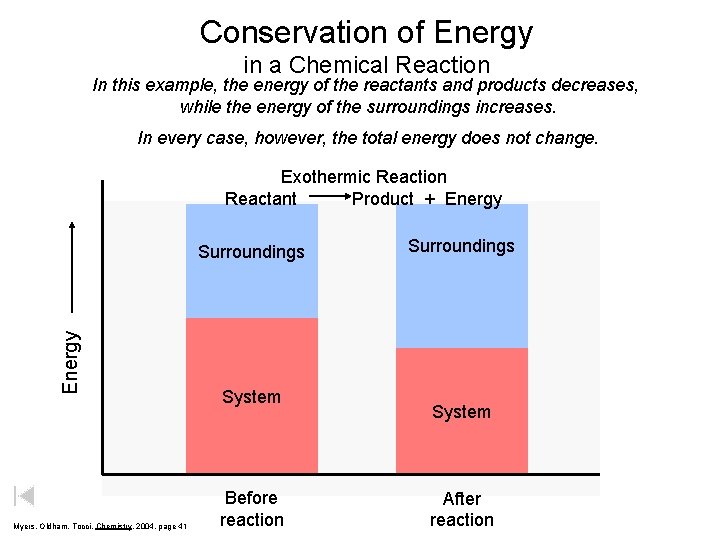
Conservation of Energy in a Chemical Reaction In this example, the energy of the reactants and products decreases, while the energy of the surroundings increases. In every case, however, the total energy does not change. Exothermic Reaction Reactant Product + Energy Surroundings Myers, Oldham, Tocci, Chemistry, 2004, page 41 System Before reaction Surroundings System After reaction
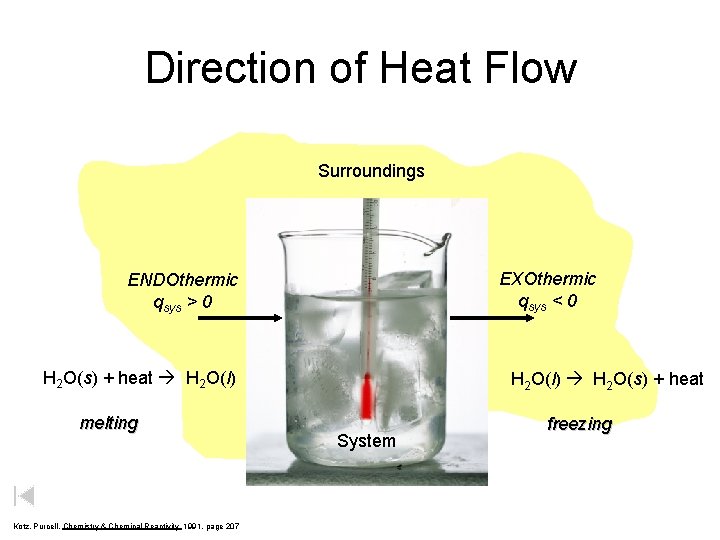
Direction of Heat Flow Surroundings ENDOthermic qsys > 0 System H 2 O(s) + heat H 2 O(l) melting Kotz, Purcell, Chemistry & Chemical Reactivity 1991, page 207 EXOthermic qsys < 0 H 2 O(l) H 2 O(s) + heat System freezing
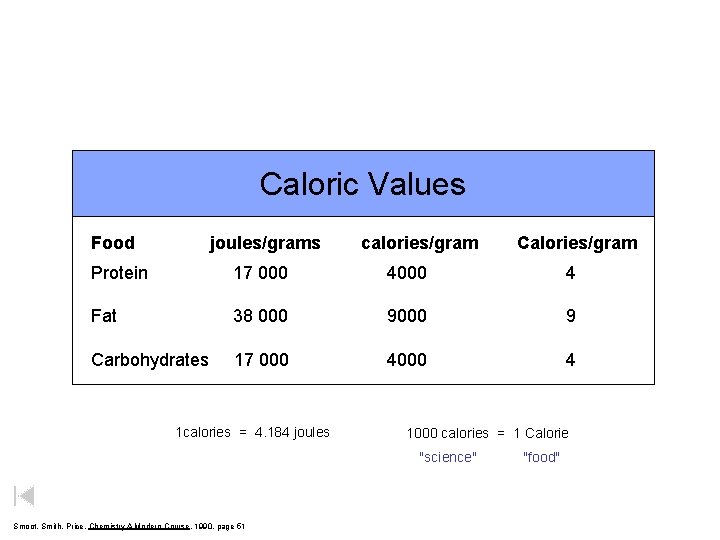
Caloric Values Food joules/grams calories/gram Calories/gram Protein 17 000 4 Fat 38 000 9 Carbohydrates 17 000 4 1 calories = 4. 184 joules 1000 calories = 1 Calorie "science" Smoot, Smith, Price, Chemistry A Modern Course, 1990, page 51 "food"
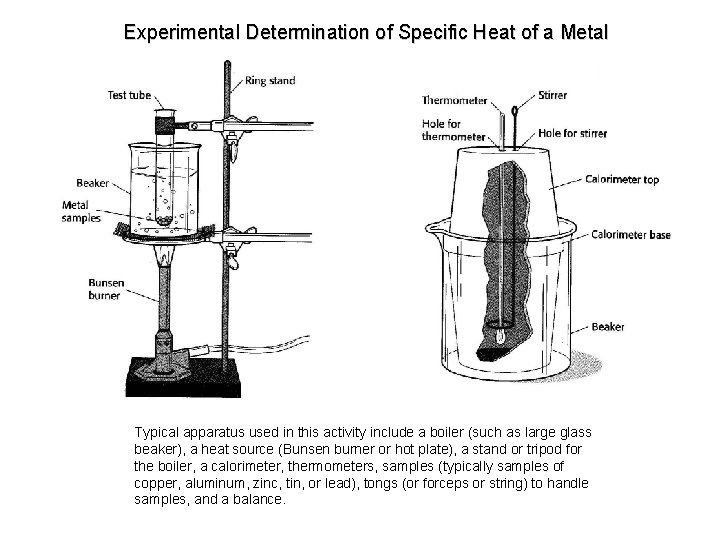
Experimental Determination of Specific Heat of a Metal Typical apparatus used in this activity include a boiler (such as large glass beaker), a heat source (Bunsen burner or hot plate), a stand or tripod for the boiler, a calorimeter, thermometers, samples (typically samples of copper, aluminum, zinc, tin, or lead), tongs (or forceps or string) to handle samples, and a balance.
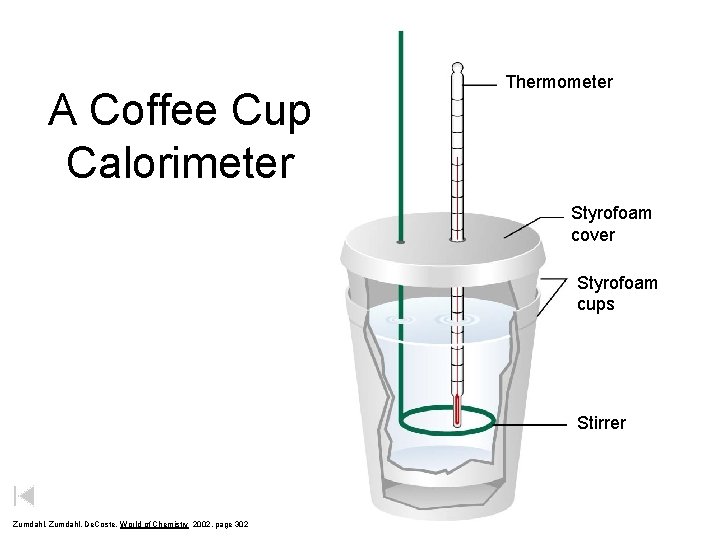
A Coffee Cup Calorimeter Thermometer Styrofoam cover Styrofoam cups Stirrer Zumdahl, De. Coste, World of Chemistry 2002, page 302

A Bomb Calorimeter
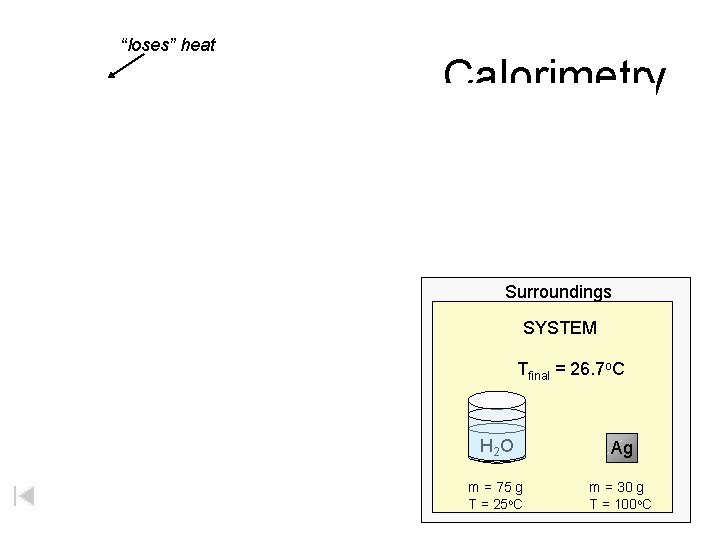
“loses” heat Calorimetry Surroundings SYSTEM Tfinal = 26. 7 o. C H 2 O Ag m = 75 g T = 25 o. C m = 30 g T = 100 o. C

Calorimetry Surroundings SYSTEM H 2 O Ag m = 75 g T = 25 o. C m = 30 g T = 100 o. C
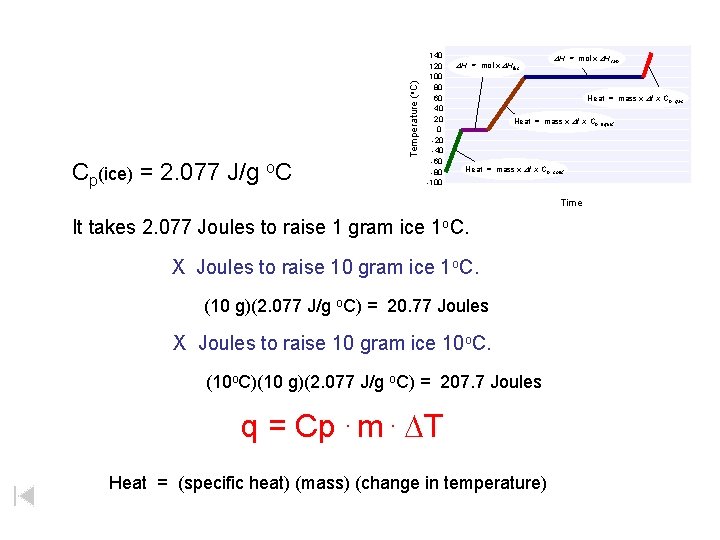
Temperature (o. C) Cp(ice) = 2. 077 J/g o. C 140 120 100 80 60 40 20 0 -20 -40 -60 -80 -100 DH = mol x DHfus DH = mol x DHvap Heat = mass x Dt x Cp, gas Heat = mass x Dt x Cp, liquid Heat = mass x Dt x Cp, solid Time It takes 2. 077 Joules to raise 1 gram ice 1 o. C. X Joules to raise 10 gram ice 1 o. C. (10 g)(2. 077 J/g o. C) = 20. 77 Joules X Joules to raise 10 gram ice 10 o. C. (10 o. C)(10 g)(2. 077 J/g o. C) = 207. 7 Joules q = Cp. m. DT Heat = (specific heat) (mass) (change in temperature)

Temperature (o. C) q = Cp. m. DT 140 120 100 80 60 40 20 0 -20 -40 -60 -80 -100 DH = mol x DHfus Heat = mass x Dt x Cp, gas Heat = mass x Dt x Cp, liquid Heat = mass x Dt x Cp, solid Heat = (specific heat) (mass) (change in temperature) Given Ti = -30 o. C Tf = -20 o. C q = 207. 7 Joules DH = mol x DHvap Time

240 g of water (initially at 20 o. C) are mixed with an unknown mass of iron (initially at 500 o. C). When thermal equilibrium is reached, the system has a temperature of 42 o. C. Find the mass of the iron. Fe T = 500 o. C mass = ? grams T = 20 o. C mass = 240 g - LOSE heat = GAIN heat - [(Cp, Fe) (mass) (DT)] = (Cp, H O) (mass) (DT) 2 - [(0. 4495 J/go. C) (X g) (42 o. C - 500 o. C)] = (4. 184 J/go. C) (240 g) (42 o. C - 20 o. C)] Drop Units: - [(0. 4495) (X) (-458)] = (4. 184) (240 g) (22) 205. 9 X = 22091 X = 107. 3 g Fe Calorimetry Problems 2 question #5

A 97 g sample of gold at 785 o. C is dropped into 323 g of water, which has an initial temperature of 15 o. C. If gold has a specific heat of 0. 129 J/go. C, what is the final temperature of the mixture? Assume that the gold experiences no change in state of matter. Au T = 785 o. C mass = 97 g T = 15 o. C mass = 323 g - LOSE heat = GAIN heat - [(Cp, Au) (mass) (DT)] = (Cp, H O) (mass) (DT) 2 Drop Units: - [(0. 129 J/go. C) (97 g) (Tf - 785 o. C)] = (4. 184 J/go. C) (323 g) (Tf - 15 o. C)] - [(12. 5) (Tf - 785 o. C)] = (1. 35 x 103) (Tf - 15 o. C)] -12. 5 Tf + 9. 82 x 103 = 1. 35 x 103 Tf - 2. 02 x 104 3 x 104 = 1. 36 x 103 Tf Tf = 22. 1 o. C Calorimetry Problems 2 question #8
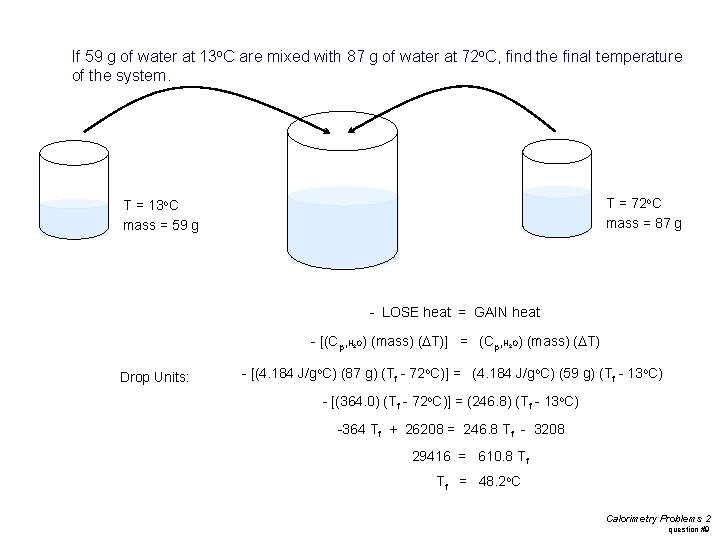
If 59 g of water at 13 o. C are mixed with 87 g of water at 72 o. C, find the final temperature of the system. T = 72 o. C mass = 87 g T = 13 o. C mass = 59 g - LOSE heat = GAIN heat - [(Cp, H O) (mass) (DT)] = (Cp, H O) (mass) (DT) 2 Drop Units: 2 - [(4. 184 J/go. C) (87 g) (Tf - 72 o. C)] = (4. 184 J/go. C) (59 g) (Tf - 13 o. C) - [(364. 0) (Tf - 72 o. C)] = (246. 8) (Tf - 13 o. C) -364 Tf + 26208 = 246. 8 Tf - 3208 29416 = 610. 8 Tf Tf = 48. 2 o. C Calorimetry Problems 2 question #9

A 322 g sample of lead (specific heat = 0. 138 J/go. C) is placed into 264 g of water at 25 o. C. If the system's final temperature is 46 o. C, what was the initial temperature of the lead? Pb T = ? o. C mass = 322 g Ti = 25 o. C mass = 264 g Tf = 46 o. C Pb - LOSE heat = GAIN heat - [(Cp, Pb) (mass) (DT)] = (Cp, H O) (mass) (DT) 2 Drop Units: - [(0. 138 J/go. C) (322 g) (46 o. C - Ti)] = (4. 184 J/go. C) (264 g) (46 o. C- 25 o. C)] - [(44. 44) (46 o. C - Ti)] = (1104. 6) (21 o. C)] - 2044 + 44. 44 Ti = 23197 44. 44 Ti = 25241 Ti = 568 o. C Calorimetry Problems 2 question #12
- Slides: 17