Calibration Temperature Percent Error What is Calibration calibration

Calibration, Temperature & Percent Error

What is Calibration? calibration of any measuring device is: distance between two closest lines • NOT distance between two closest #’s • NOT all same instruments calibrated the same

not all graduated cylinders calibrated same way

Large graduated cylinder: ? capacity ? calibration 1000 ml 10 ml

? calibration 1 ml 67. 4 ml

? calibration 1 ml 13. 0 ml 12. 9 ml ? 13. 1 ml ?

6. 68 ml ? calibration 0. 2 ml

Can you figure the calibration of any piece of lab equipment?

meter stick: two closest lines are 1 mm (millimeter) apart

? calibration 25 ml

quadruple beam balance

What’s the calibration? 183. 599 g 183. 600 g 0. 01 g 183. 601 g

buret ? calibration 0. 1 ml 21. 35 ml note: unlike graduated cylinder, numbers go down, so you read backwards from way read graduated cylinder

Temperature Scales

scientifically speaking … the definition of temperature: temperature is measure of average kinetic energy of particles in system
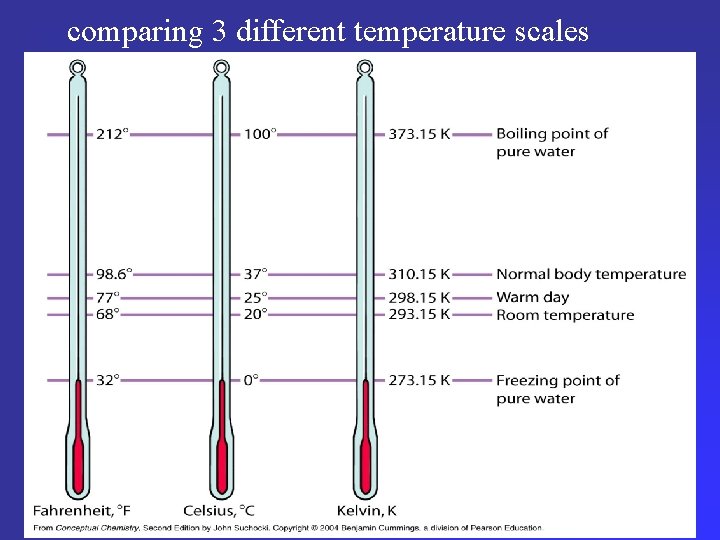
comparing 3 different temperature scales comparing 3 different scales
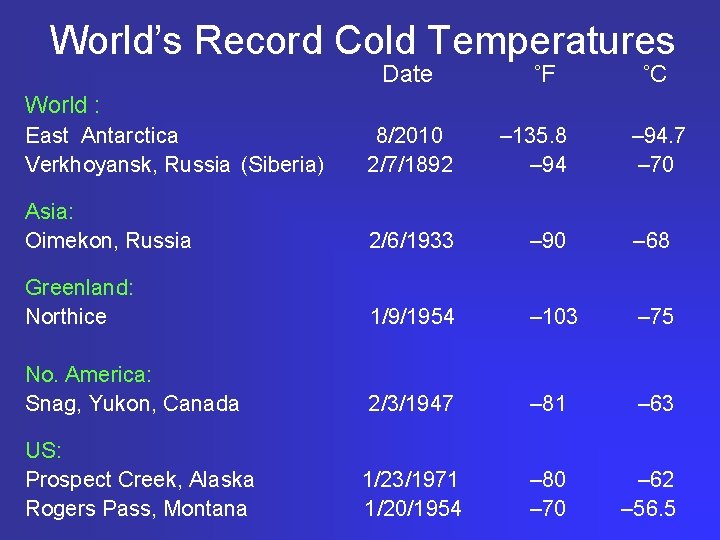
World’s Record Cold Temperatures Date ˚F ˚C World : East Antarctica Verkhoyansk, Russia (Siberia) 8/2010 2/7/1892 – 135. 8 – 94 Asia: Oimekon, Russia 2/6/1933 – 90 – 68 Greenland: Northice 1/9/1954 – 103 – 75 No. America: Snag, Yukon, Canada 2/3/1947 – 81 – 63 1/23/1971 1/20/1954 – 80 – 70 – 62 – 56. 5 US: Prospect Creek, Alaska Rogers Pass, Montana – 94. 7 – 70

Conversion formulas To convert between celsius & Kelvin scales: K = °C + 273 (more precisely 273. 15) °C = K – 273 [F = (9/5 °C) + 32]

errors in science experiments are inevitable and need to be dealt with …

Percent Error ratio of error to accepted value % error = error accepted value x 100

Calculation of Error measuredaccepted value – accepted value x 100 value absolute value: always positive
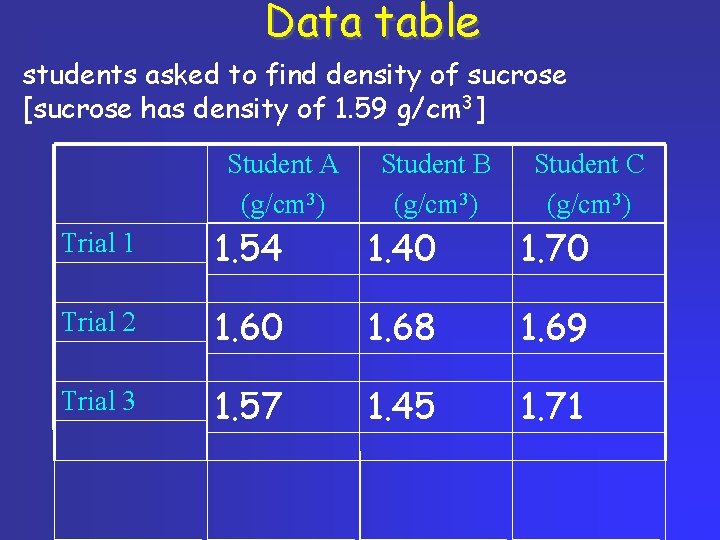
Data table students asked to find density of sucrose [sucrose has density of 1. 59 g/cm 3] Student A (g/cm 3) Student B (g/cm 3) Student C (g/cm 3) Trial 1 1. 54 1. 40 1. 70 Trial 2 1. 60 1. 68 1. 69 Trial 3 1. 57 1. 45 1. 71
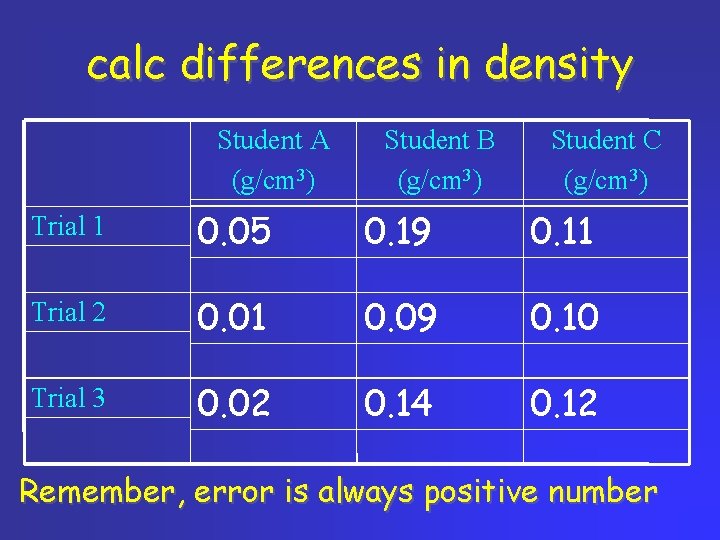
calc differences in density Student A (g/cm 3) Student B (g/cm 3) Student C (g/cm 3) Trial 1 0. 05 0. 19 0. 11 Trial 2 0. 01 0. 09 0. 10 Trial 3 0. 02 0. 14 0. 12 Remember, error is always positive number
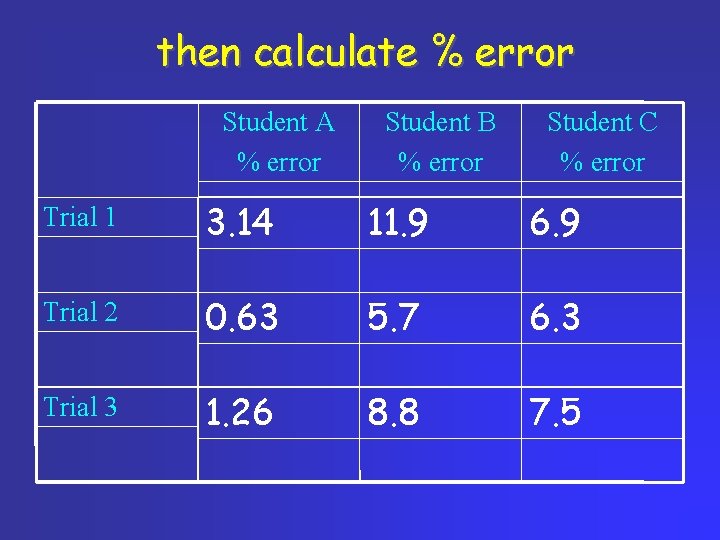
then calculate % error Student A % error Student B % error Student C % error Trial 1 3. 14 11. 9 6. 9 Trial 2 0. 63 5. 7 6. 3 Trial 3 1. 26 8. 8 7. 5

What can do with error? • easy to compare data from one student/group to another • easy to compare data from one trial to another • can map change from one lab to next or from start of year to end
- Slides: 25