Caliber Deliver More Deliver Excellence Quality Focus in

Caliber| Deliver More Deliver Excellence Quality Focus in Pharma 4. 0 ERA INDIAN DRUG MANUFACTURERS' ASSOCIATION DATE : 24 th MAY 2019 MUMBAI

Caliber| Deliver More Deliver Excellence Clean & Correct Data? We will discuss Common Issues in Quality Management Meeting Quality Management Challenges Beyond Challenges & The Future

Caliber| Deliver More Deliver Excellence Clean & Correct Data THE BASICS

Caliber| Deliver More Deliver Excellence Good Laboratory Practices: GLP • GLPs rest on two foundations: 4 M Rule – The right resources • ALCOA – The right execution •
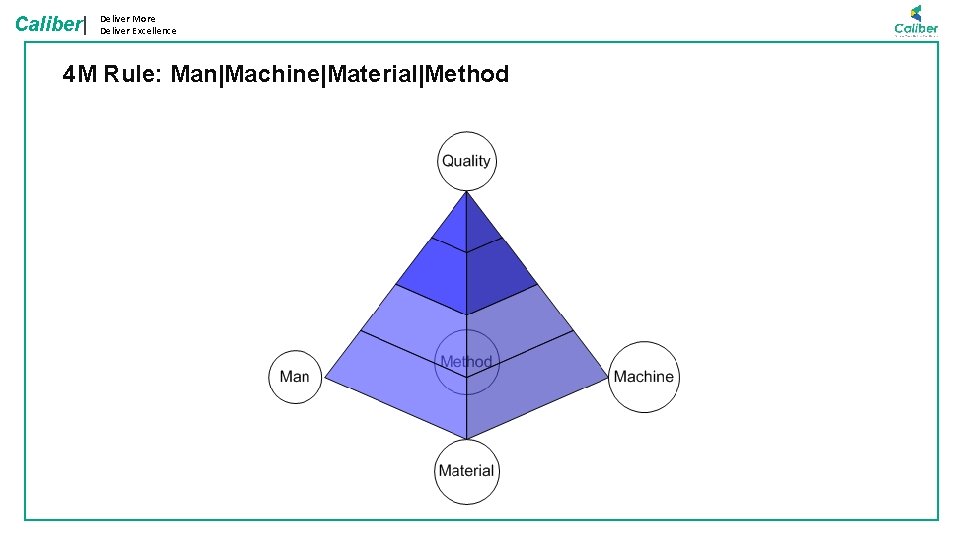
Caliber| Deliver More Deliver Excellence 4 M Rule: Man|Machine|Material|Method

Deliver More Deliver Excellence Caliber| • Instruments/ Machines are to be in fit condition for using in GMP environment • Calibrated instruments only to be used • Instrument with reported problem should not be used • Only suitable instrument should be used • 6% of issues are attributed Calibration records MATERIAL • Inadequate records and SOP Contributes to more than 10% of the deviations • SOP, STP, GTP Records version management • Training on the latest procedures • Using Right method and protocols for testing MACHINERY • Qualified person to carry out a specific Testing: • By category • By Product • By Test • Trained in all SOP, GTPs for operating in GLP area • One of the major areas of concern. • Training and Qualification issues (4% ) METHOD MAN Categorization of 483 s on lab management • All material used for Testing need to be Valid • Standards, Chemicals Volumetric solutions • Valid: • Available • Not Expired • Right standard/ solution • Close to 9% issues are due to Material

Caliber| Deliver More Deliver Excellence ALCOA • Attributable — Who acquired the data or performed an action and when? • Legible — Can you read the data and any entries? • Contemporaneous — Documented at the time of the activity. • Original — A written printout or observation or a certified copy thereof. • Accurate — No errors or editing without documented amendments.
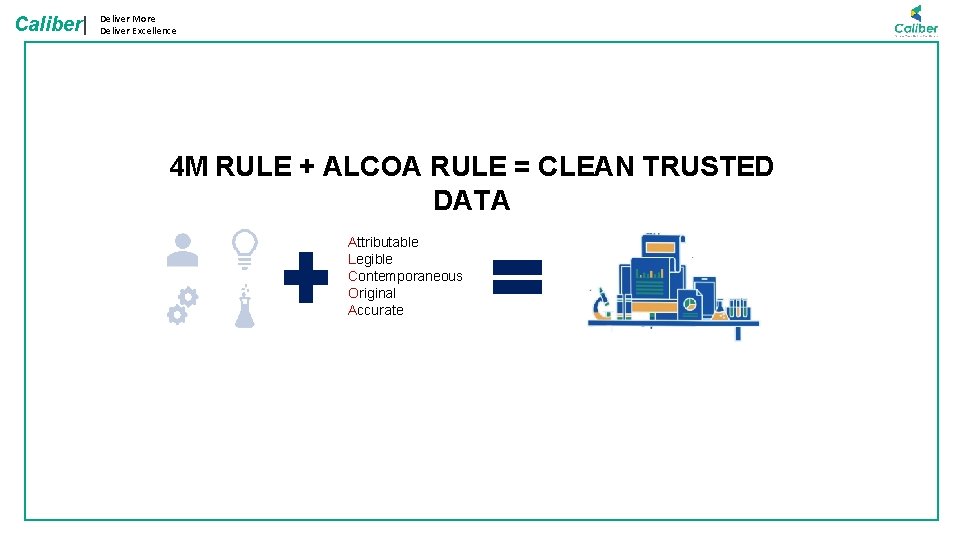
Caliber| Deliver More Deliver Excellence 4 M RULE + ALCOA RULE = CLEAN TRUSTED DATA Attributable Legible Contemporaneous Original Accurate

Caliber| Deliver More Deliver Excellence Common Issues in Quality Management 483, NON-CONFORMANCE, ERRORS, CHALLENGES
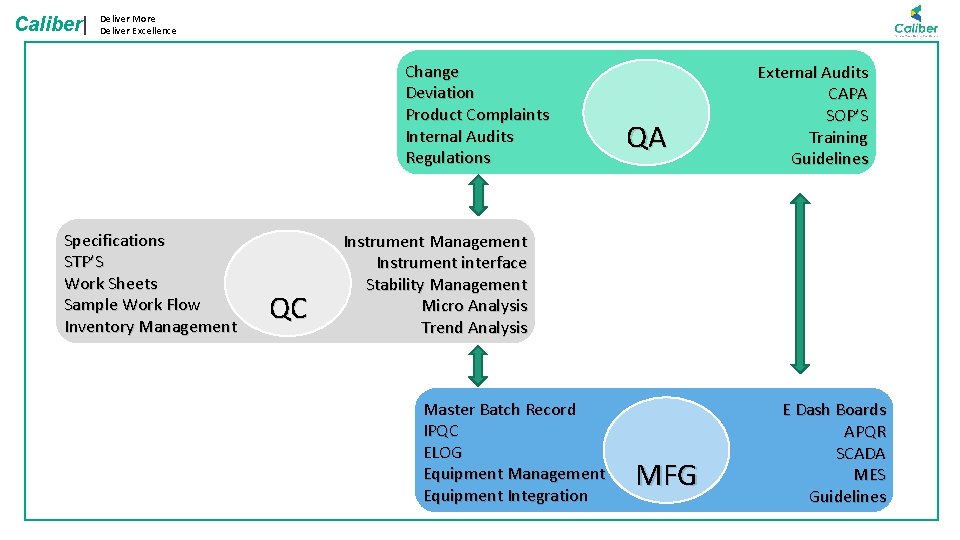
Caliber| Deliver More Deliver Excellence Change Deviation Product Complaints Internal Audits Regulations Specifications STP’S Work Sheets Sample Work Flow Inventory Management QC QA External Audits CAPA SOP’S Training Guidelines Instrument Management Instrument interface Stability Management Micro Analysis Trend Analysis Master Batch Record IPQC ELOG Equipment Management Equipment Integration MFG E Dash Boards APQR SCADA MES Guidelines
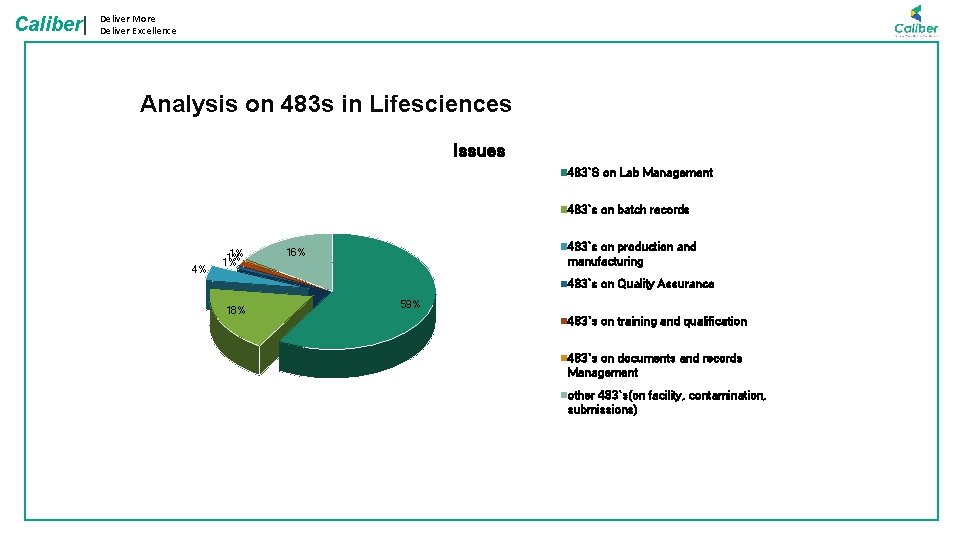
Caliber| Deliver More Deliver Excellence Analysis on 483 s in Lifesciences Issues 483`S on Lab Management 483`s on batch records 4% 1% 1% 1% 483`s on production and manufacturing 16% 483`s on Quality Assurance 18% 59% 483`s on training and qualification 483`s on documents and records Management other 483`s(on facility, contamination, submissions)
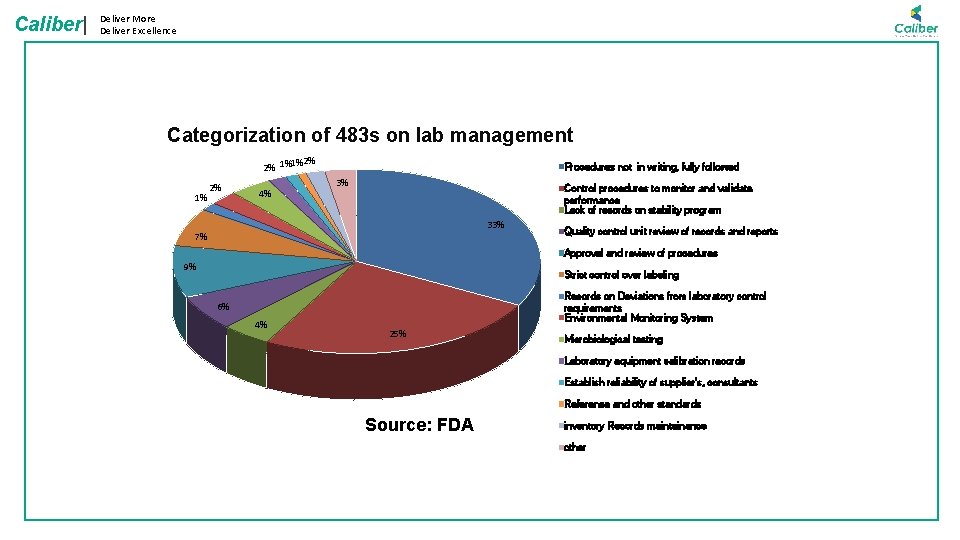
Caliber| Deliver More Deliver Excellence Categorization of 483 s on lab management 2% 2% 1%1% 1% 2% 4% Procedures not in writing, fully followed 3% Control procedures to monitor and validate performance Lack of records on stability program 33% 7% Quality control unit review of records and reports Approval and review of procedures 9% Strict control over labeling Records on Deviations from laboratory control requirements Environmental Monitoring System 6% 4% 25% Microbiological testing Laboratory equipment calibration records Establish reliability of supplier's, consultants Reference and other standards Source: FDA inventory Records maintainance other

Caliber| Deliver More Deliver Excellence Common Challenges QC QA Mfg • Re-writing errors and calculation errors • Repeated works- Low efficiency • Compliance risk – 483 s • Lack of information • Insufficient training • Improper review • Lack of controls • FDA & other agencies looking at self regulation - shifting responsibilities to QA • PAT, Process improvement, Manufacturing excellence • Relevant and current to derive meaningful conclusions • Self governance, 24 X 7 audit Readiness • Un-controlled Document Templates. • Tedious Review and Approval process • Issuance: Takes more time to write on all the pages • High rate of corrections • Executed BRM compilation

Caliber| Deliver More Deliver Excellence Meeting Quality Management Challenges TOOLS

Caliber| Deliver More Deliver Excellence Tools to Address & Avoid 483 s Caliber. LIMS – QC Caliber. BRM - Mfg Caliber. QAMS - QA Caliber. DMS - QA Nichelon 5 CMS - QA ü ROBUST ü RELIABLE ü SECURE
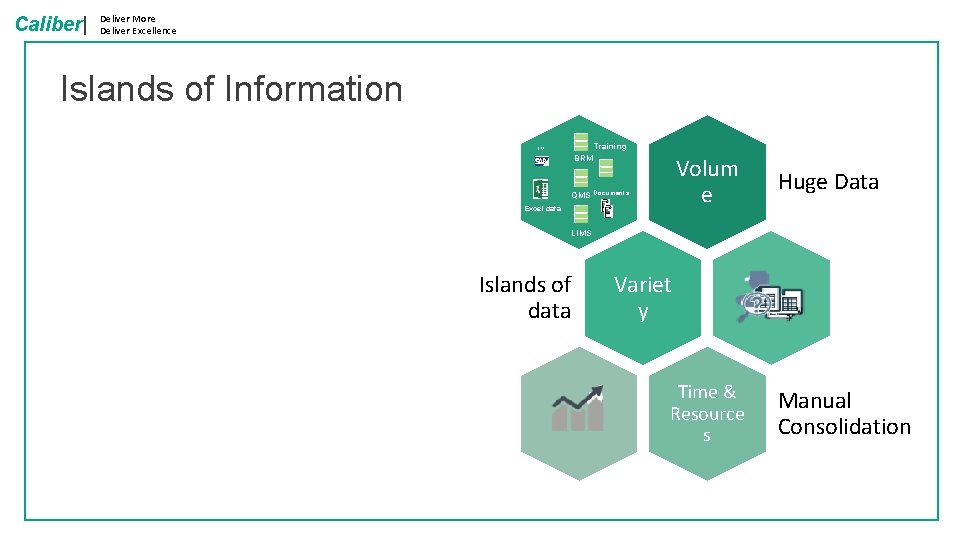
Caliber| Deliver More Deliver Excellence Islands of Information Training ERP BRM Volum e Huge Data Time & Resource s Manual Consolidation QMS Documents Excel data LIMS Islands of data Variet y
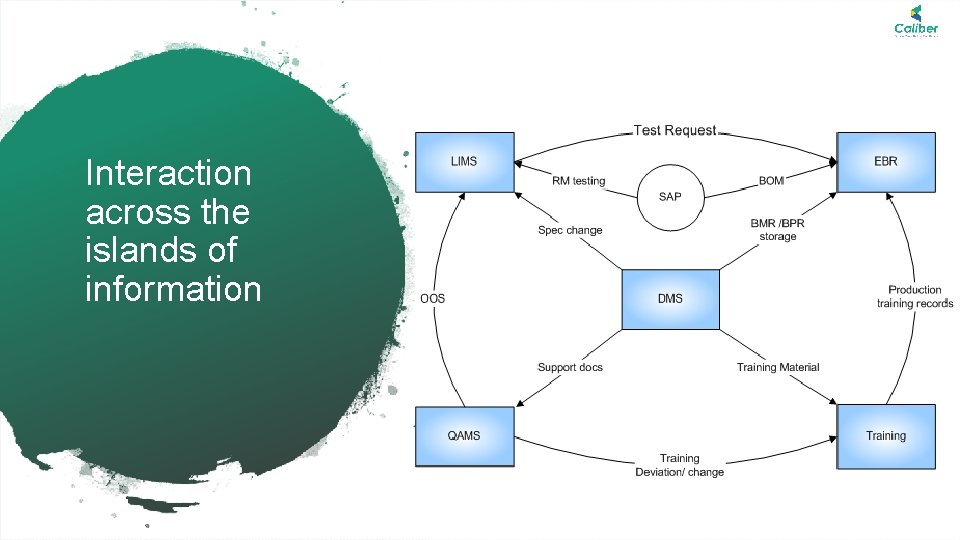
Caliber| Deliver More Deliver Excellence Interaction across the islands of information

Caliber| Deliver More Deliver Excellence Beyond Challenges & The Future THE LAB OF THE FUTURE

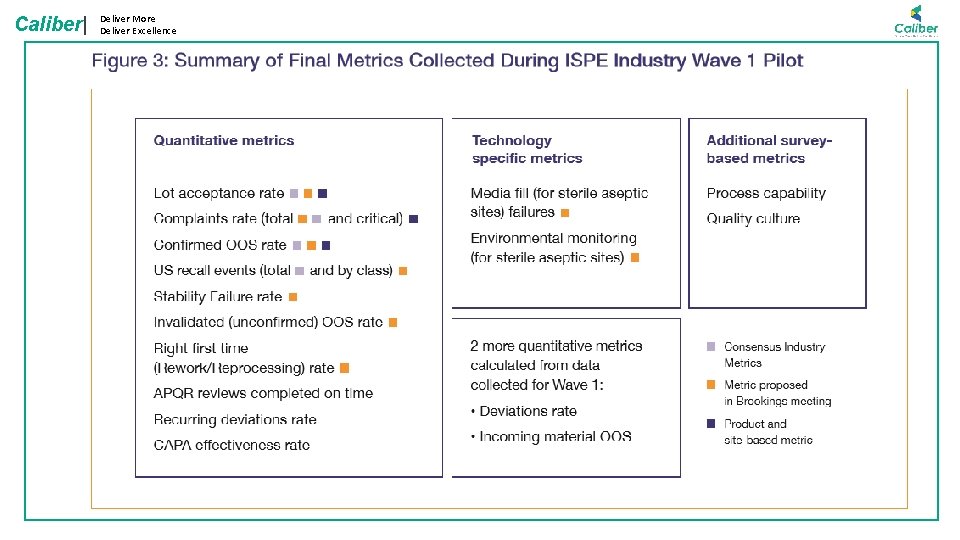
Caliber| Deliver More Deliver Excellence Quality Metrics
Caliber| Deliver More Deliver Excellence

Caliber| Deliver More Deliver Excellence Reporting: Levels Process Capability Metrics APQR & Other special report (MIS) Data Warehouse: Inter-department Analytics & correlation Department Analytics and Reports Data Source: LIMS, QAMS, EBR, SAP, etc
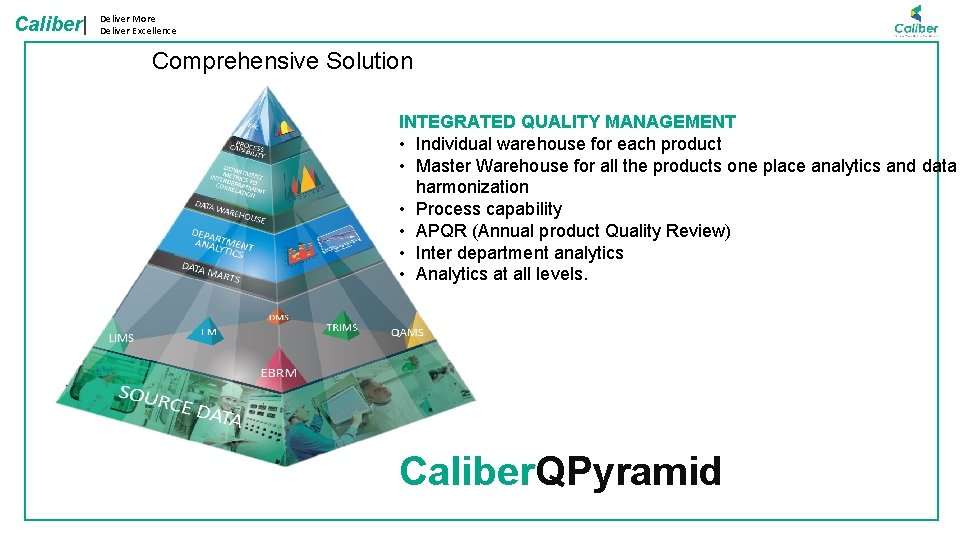
Caliber| Deliver More Deliver Excellence Comprehensive Solution INTEGRATED QUALITY MANAGEMENT • Individual warehouse for each product • Master Warehouse for all the products one place analytics and data harmonization • Process capability • APQR (Annual product Quality Review) • Inter department analytics • Analytics at all levels. Caliber. QPyramid
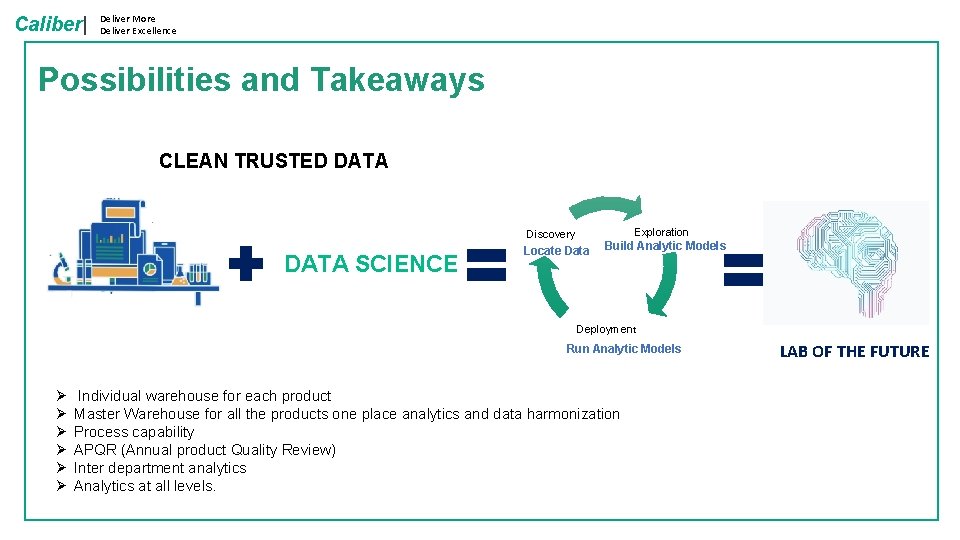
Caliber| Deliver More Deliver Excellence Possibilities and Takeaways CLEAN TRUSTED DATA Exploration Discovery DATA SCIENCE Locate Data Build Analytic Models Deployment Run Analytic Models Ø Ø Ø Individual warehouse for each product Master Warehouse for all the products one place analytics and data harmonization Process capability APQR (Annual product Quality Review) Inter department analytics Analytics at all levels. LAB OF THE FUTURE

Caliber| Deliver More Deliver Excellence ML & AI – LAB OF THE FUTURE Futuristic Solutions from Caliber Foundation for ML and AI is strong data availability Machine Learning Capacity Planning Inventory checking based on the samples logged Machine failure detection MTBF Leading analytics

Caliber| Deliver More Deliver Excellence ML & AI – LAB OF THE FUTURE Futuristic Solutions from Caliber Foundation for ML and AI is strong data availability AI - Artificial Intelligence Routing samples to the analyst who has lesser workload Looking at the inventory and ordering the material Suggesting alternative machine in case of failure informing delays and effects to the stakeholders

Caliber| Deliver More Deliver Excellence Analytics leading to ML
Caliber| Deliver More Deliver Excellence

Caliber| Deliver More Deliver Excellence ABOUT US 2001 Founded, Products for Pharma 80+ Logos 10 Countries 70 K + Users

Caliber| Deliver More Deliver Excellence OUR JOURNEY * India’s largest Quality Management Products company (Gartner acknowledged) * Focused on Pharma Quality Management. * * Implemented first paperless laboratory in India * ISO 9001 2015 certified Team of 350 70000+ users 170 installations
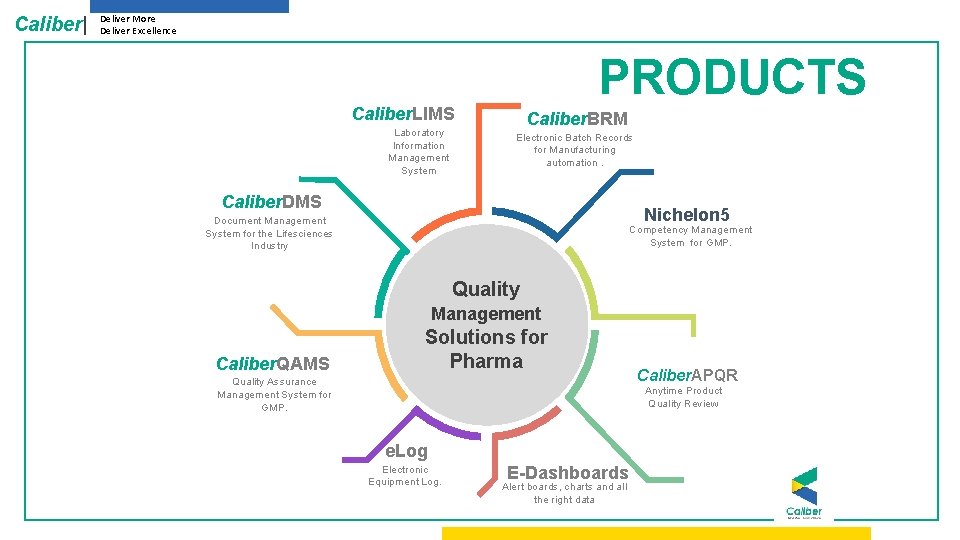
Caliber| Deliver More Deliver Excellence PRODUCTS Caliber. LIMS Laboratory Information Management System Caliber. BRM Electronic Batch Records for Manufacturing automation. Caliber. DMS Nichelon 5 Document Management System for the Lifesciences Industry Competency Management System for GMP. Quality Management Caliber. QAMS Solutions for Pharma Quality Assurance Management System for GMP. Caliber. APQR Anytime Product Quality Review e. Log Electronic Equipment Log. E-Dashboards Alert boards, charts and all the right data

Caliber| Deliver More Deliver Excellence OFFICES Caliber India Caliber U. S. A Design & Development Product Management Version upgrade Sales & Marketing HQ Support to NA & Canada, Mexico Alliances & Partnerships Support for India/Asia Pac Support Europe as backup Support for Europe Team of 350

Caliber| Deliver More Deliver Excellence CALIBER CAMPUS • Campus with 45, 000 Sq. ft • 350 team • Resilient Redundant Servers • Secure & Safe environment • Zero water wastage campus

Caliber| Deliver More Deliver Excellence f o e m o S OUR CUSTOMERS

Caliber| Deliver More Deliver Excellence Caliber INNOVATIONS Guiding the path for the industry

Caliber| Deliver More Deliver Excellence 100% PAPERLESS LABORATORY • Helping pharma companies achieve a 100% paperless laboratory – fighting the paper monster! • Seamless integration with SAP for Lots – auto login and result posting. • Integration with various types of Instruments including seamless integration with CDS (Empower 3) • On-line entry of the observations in Tablets and Terminals • Integration with DMS for generation of Sample wise Dossiers • Integration with QMS in case of Out Of Spec (OOS) & OOT

Caliber| Deliver More Deliver Excellence IQM First in the industry Integrated Quality Management Competency Management (Nichelon) Electronic Batch Records Quality Assurance (QAMS) (BRM) Document Management (DMS) Anytime PQR LIMS (APQR) EDashboards • The concept of IQM is becoming popular among the pharma players. • We have 6 customers with complete package and 6 customers with 4 products as integrated Quality Management suite. • We are adding to the top and bottom lines • Caliber is a leader in providing such innovative solutions for Pharma

Caliber| Deliver More Deliver Excellence QUALITY METRICS Caliber with multiple products for Pharma, has built a comprehensive solution for Quality Metrics: • • Individual warehouse for each product Master Warehouse for all the products one place analytics and data harmonization Process capability APQR (Annual product Quality Review) Inter department analytics Analytics at all levels.

Caliber| Deliver More Deliver Excellence ANYTIME PQR • Award Winning Product - APQR • Individual warehouse for each product • Master Warehouse for all the products one place analytics and data harmonization • Inter department analytics • Analytics at all levels.

Caliber| Deliver More Deliver Excellence ALERT BOARDS Caliber e. Dash. Board gives holistic insights on your Quality objectives of manufacturing by making information readily available as per the need to support important decisions. Caliber dynamic dashboards enable you to create a customized, detailed, factbased view of your actionable items and performance to facilitate decisionmaking.

Caliber| Deliver More Deliver Excellence We are Caliber Deliver More. Deliver Excellence.

Caliber| Deliver More Deliver Excellence Discussion Sumanth Chinta Head-PRESALES www. caliberuniversal. com
- Slides: 41