Calculations in Determination of Iron In Food Lab

Calculations in Determination of Iron In Food Lab Travis Pyrzynski That’s in Food?

Data Gathered In Lab n n n Mass of the sample. 4 absorbances for known concentrations. Mass of unknown solution. Homer is a big fan of Beer’s Law.
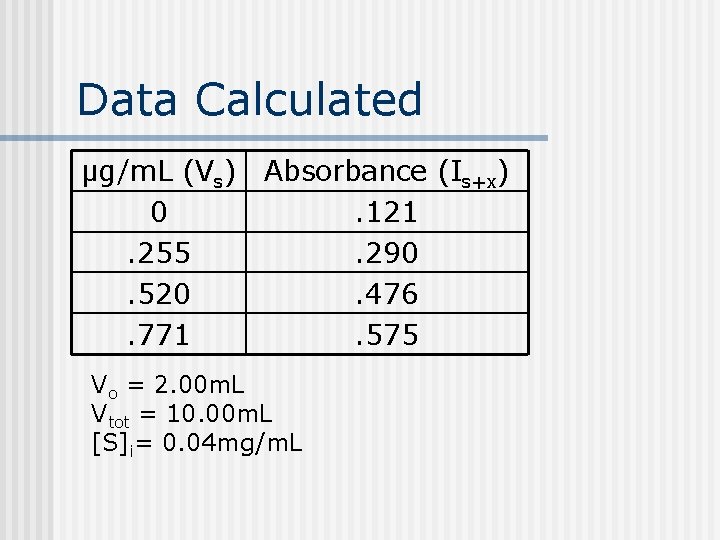
Data Calculated μg/m. L (Vs) Absorbance (Is+x) 0. 121. 255. 290. 520. 476. 771. 575 Vo = 2. 00 m. L Vtot = 10. 00 m. L [S]i= 0. 04 mg/m. L

Calculating a Line n n Here the Method of Least Squares was used to set up a line y=mx+b. Data needed to use Method: x, y, x 2, Σx, Σy, Σx 2.

Equations n We need to use the standard addition equation to find the concentration of the unknown. This is the basic equation. Solving for the initial Concentrations. Rearranging the equation allows us to get a straight line. Where is the y value and is the x value.
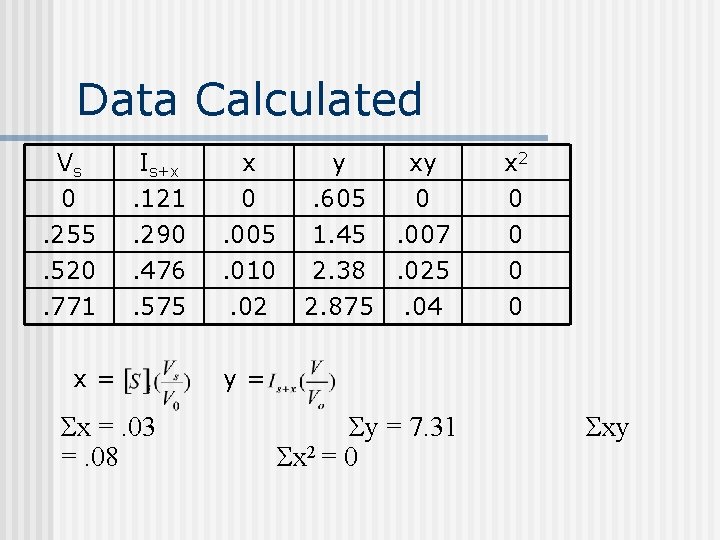
Data Calculated Vs 0. 255. 520 Is+x. 121. 290. 476 x 0. 005. 010 y. 605 1. 45 2. 38 xy 0. 007. 025 x 2 0 0 0 . 771 . 575 . 02 2. 875 . 04 0 x= Σx =. 03 =. 08 y= Σy = 7. 31 Σx 2 = 0 Σxy

What to Do Now that we have calculated these we use to get a straight line. n The slope m = n The intercept b = n. D = n n is the number of points n

Solving di n n di = yi – (m*xi+b) d 1 =. 605 –(0 +. 666) = -. 0611 di -. 0611. 0177. 151 -. . 105 (di)2 3. 73*10 -3 3. 12*10 -4 2. 28*10 -2 1. 16*10 -2

Standard Deviations n Standard Deviation of Vertical Deviations n Standard Deviation of y-intercept n Standard Deviation of Slope

Sample D = (Σx 2)n- (Σx) =. 00053 n m = {(Σxy)n – (Σx)(Σy)}/D = 150 n b ={Σ(x 2)(Σy) – (Σx)(Σxy)}/D =. 666 n Putting them together!!! y=150 x+. 666

What Does That Do For Us? !? ! n n Now that we have an equation for the standards, we are able to calculate the concentration of iron in food. Setting the equation equal to zero will give us the concentration of the unknown. 0=150 x+. 666 x = 4. 43*10 -3 mg/m. L Multiply by the volume of the solution (4. 43*10 -3 mg/m. L)*(10 m. L) = 4. 43*10 -2 μg

Now What? ! Whatchu expect me do wif dat?

To Continue…. Now we have the grams of iron in food. n Next we calculate the percent of iron in food. n (4. 43*10 -2 /5. 7064)*100 =. 776%

Again Now we calculate the uncertainty in the wt/wt%. n Using equation 5. 17 from the book n =. 203 μg

Uncertainty in wt/wt% n n Solving for the uncertainty The percent uncertainty now becomes 2. 24*10 -4 +/- 1. 68%

And that’s it! Dat’s Wha’ I’m Talkin’ Bout!

But Wait…. There’s More Standard Deviation for 200 Alex…. n. What does the n represent in the equations for standard deviation? The degrees of freedom, which is the number of independent pieces of information available. n That is why when you find an average, you are left with n-1 degrees of freedom. I prefer to give the answer before the question.
- Slides: 17