Calculations from Chemical Equations Stoichiometry Module 3 Stoichiometry














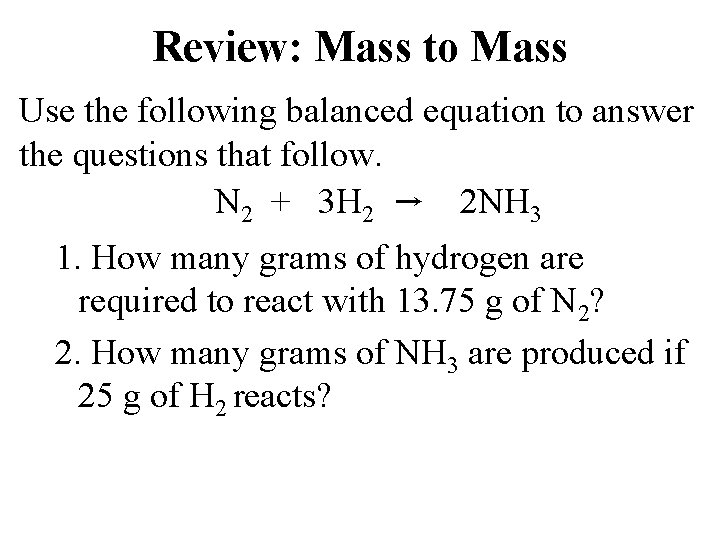
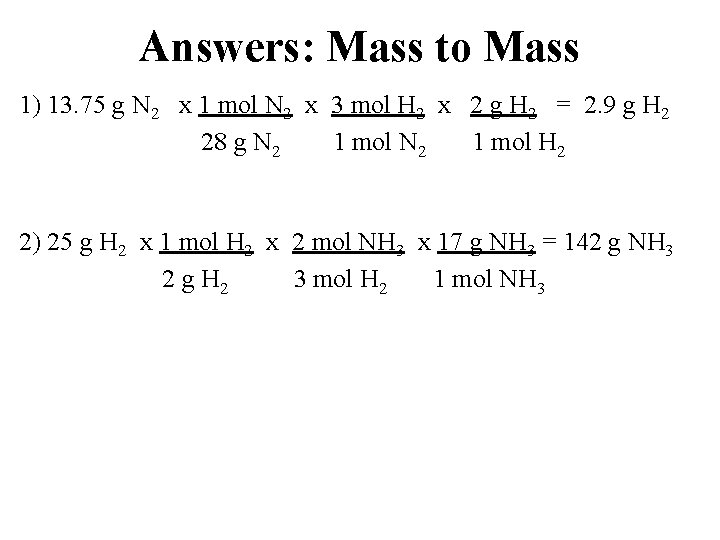





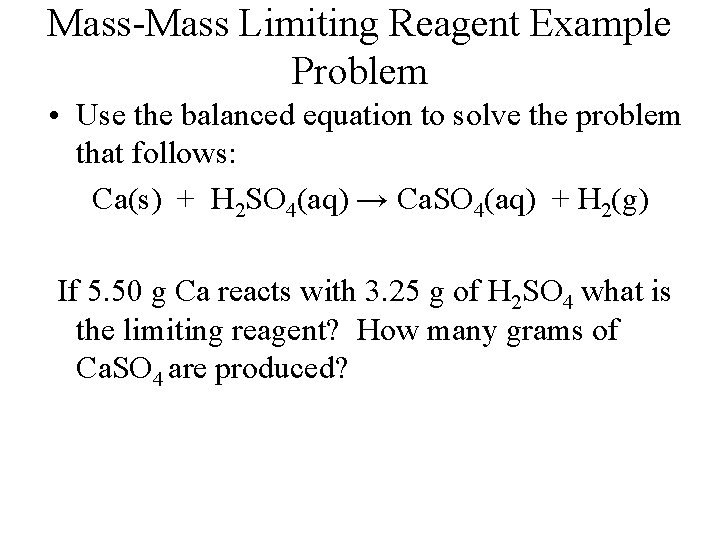





- Slides: 33

Calculations from Chemical Equations: Stoichiometry Module 3

Stoichiometry o The word stoichiometry derives from two Greek words: stoicheion = "element" metron = "measure" o Stoichiometry is the study of the quantitative relationships between substances in a chemical reaction. B A B B C B http: //www. explrelearning. com/index. cfm B

Basics Steps in Commmon Stoichiometry Problems • 1. Convert starting substance to moles • 2. Convert moles of starting substance to moles of desired substance (use mole ratio) • 3. Convert moles of desired substance to desired unit (i. e. g, particles, L)

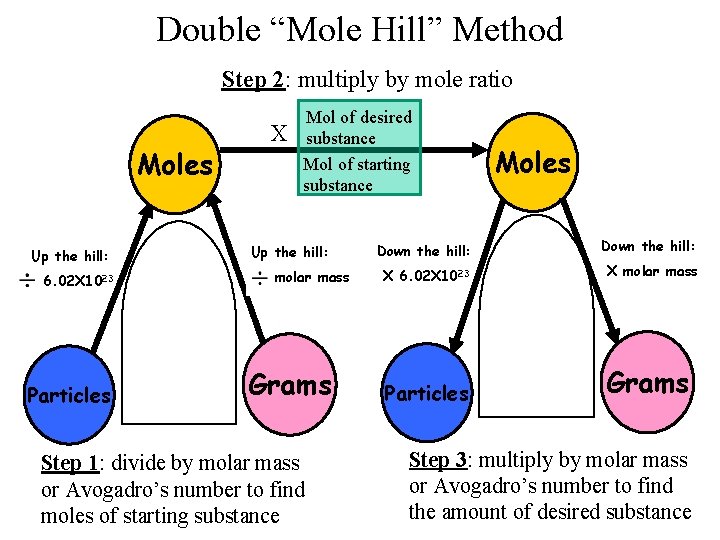
Double “Mole Hill” Method Step 2: multiply by mole ratio Moles Up the hill: 6. 02 X 1023 Particles X Mol of desired substance Mol of starting substance Up the hill: molar mass Grams Step 1: divide by molar mass or Avogadro’s number to find moles of starting substance Moles Down the hill: X 6. 02 X 1023 X molar mass Particles Grams Step 3: multiply by molar mass or Avogadro’s number to find the amount of desired substance

What is the Mole Ratio? • A ratio between the number of moles of two substances in a balanced equation. The mole ration is sometimes called the stoichiometric factor. • A mole ratio relates the amount in moles of two substances by using the coefficients of a balanced equation N 2 (g) + 3 H 2 (g) → 2 NH 3 (g) • Example 1: What is the mole ratio of nitrogen to ammonia? 1 mol N 2 2 mol NH 3 • Example 2: What is the mole ratio of hydrogen to nitrogen? 3 mol H 2 1 mol N 2

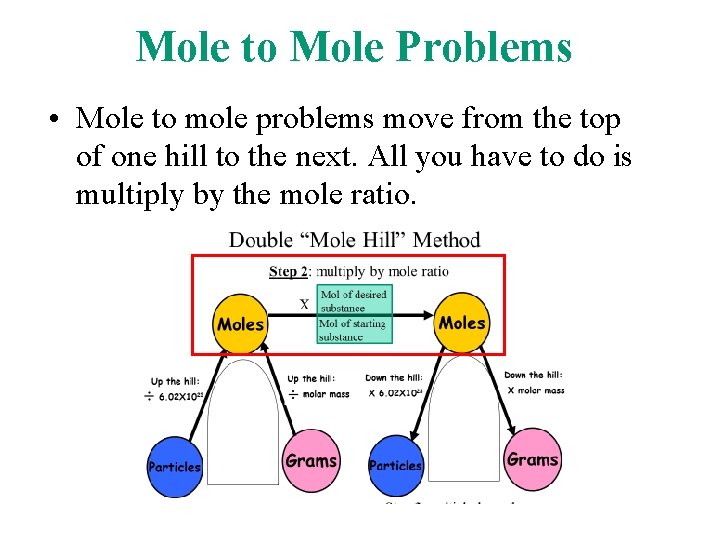
Mole to Mole Problems • In a “mole to mole” stoichiometry problem, you are given the moles of one substance and asked to find the number of moles of another substance, based on a chemical equation.

Mole to Mole Problems • Mole to mole problems move from the top of one hill to the next. All you have to do is multiply by the mole ratio.

Example 1 Q: How many moles of NH 3 can be produced if 4. 5 moles of H 2 reacts? N 2(g) + 3 H 2(g) → 2 NH 3(g) • Multiply starting moles by mole ratio with moles of desired substance on top of the ratio and moles of given substance on bottom 4. 5 mol H 2 X 2 mol NH 3 = 3. 0 mol NH 3 3 mol H 2

Example 2 Q: How many moles of Mg(OH)2 are required to react with 2. 5 moles of HCl? 2 HCl + Mg(OH)2 → Mg. Cl 2 + 2 H 2 O Multiply starting moles by mole ratio with moles of desired substance on top of the ratio and moles of given substance on bottom 2. 5 mol HCl X 1 mol Mg(OH)2 = 1. 25 mol Mg(OH)2 2 mol HCl

Review: Mole to Mole Solve the following problems using the balanced equation: Ni(NO 3)2 + 2 Na. OH → Ni(OH)2 + 2 Na. NO 3 • 1. How many moles of sodium nitrate can be produced from 7. 5 moles of nickel (II) nitrate? • 2. How many moles of sodium hydroxide are required to produce with 23. 0 moles of sodium nitrate?

Answers: Mole to Mole 1) 7. 5 mol Ni(NO 3)2 X 2 mol Na. NO 3 = 15 mol Na. NO 3 1 mol Ni(NO 3)2 2) 23 mol Na. NO 3 X 2 mol Na. OH = 23 mol Na. OH 2 mol Na. NO 3

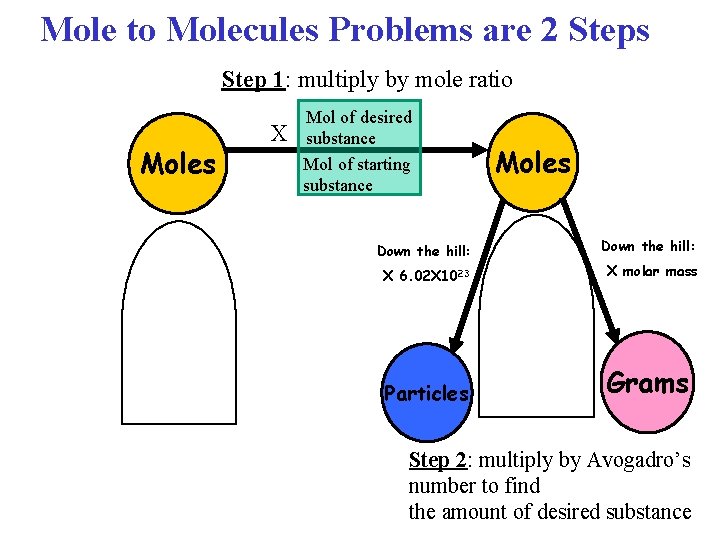
Mole to Molecules Problems • In a “mole to molecules” stoichiometry problem, you are given the moles of one substance and asked to find the number of molecules of another substance, based on a chemical equation.

Mole to Molecules Problems are 2 Steps Step 1: multiply by mole ratio Moles X Mol of desired substance Mol of starting substance Moles Down the hill: X 6. 02 X 1023 X molar mass Particles Grams Step 2: multiply by Avogadro’s number to find the amount of desired substance

Example 1 Q: How many molecules of CO 2 can be produced if 11. 0 moles of O 2 reacts? O 2(g) + 2 CO(g) → 2 CO 2(g) • Multiply starting moles by mole ratio with moles of desired substance on top of the ratio and moles of given substance on bottom, then multiply by Avogadro’s number. 11. 0 mol O 2 X 2 mol CO 2 X 6. 02 x 1023 CO 2 molecules = 1 mol O 2 1 mol CO 2 = 1. 32 x 1025 CO 2 molecules

Example 2 Q: How many molecules of water are produced when 0. 85 moles of HCl reacts? 2 HCl + Mg(OH)2 → Mg. Cl 2 + 2 H 2 O • Multiply starting moles by mole ratio with moles of desired substance on top of the ratio and moles of given substance on bottom, then multiply by Avogadro’s number. 0. 85 mol HCl X 2 mol H 2 O X 6. 02 x 1023 H 2 O molecules = 2 mol HCl 1 mol H 2 O = 5. 1 x 1023 H 2 O molecules

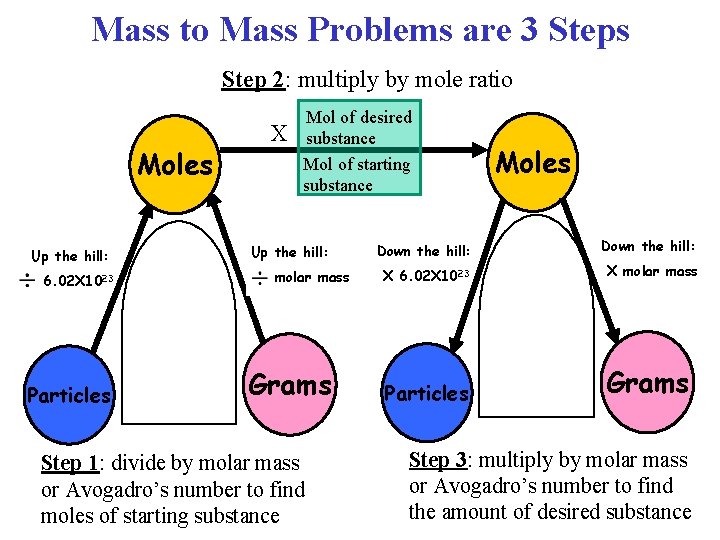
Mass to Mass Problems • In a “mass to mass” stoichiometry problem, you are given the mass in grams of one substance and asked to find the mass in grams of another substance, based on a chemical equation.

Mass to Mass Problems are 3 Steps Step 2: multiply by mole ratio Moles Up the hill: 6. 02 X 1023 Particles X Mol of desired substance Mol of starting substance Up the hill: molar mass Grams Step 1: divide by molar mass or Avogadro’s number to find moles of starting substance Moles Down the hill: X 6. 02 X 1023 X molar mass Particles Grams Step 3: multiply by molar mass or Avogadro’s number to find the amount of desired substance

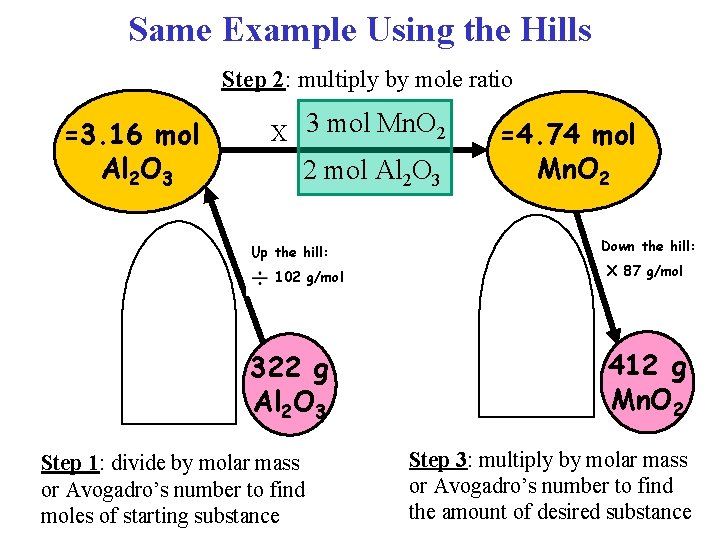
Example Q: How many grams of Mn. O 2 are required to produce 322 g of Al 2 O 3? 3 Mn. O 2 + 4 Al → 2 Al 2 O 3 + 3 Mn 322 g Al 2 O 3 x 1 mol Al 2 O 3 x 3 mol Mn. O 2 x 87 g Mn. O 2 102 g Al 2 O 3 2 mol Al 2 O 3 1 mol Mn. O 2 Step 1: Divide by molar mass = 412 g Mn. O 2 Step 2: Multiply by mole ratio Step 3: Multiply by molar mass

Same Example Using the Hills Step 2: multiply by mole ratio =3. 16 mol Al 2 O 3 X 3 mol Mn. O 2 2 mol Al 2 O 3 Up the hill: 102 g/mol 322 g Al 2 O 3 Step 1: divide by molar mass or Avogadro’s number to find moles of starting substance =4. 74 mol Mn. O 2 Down the hill: X 87 g/mol 412 g Mn. O 2 Step 3: multiply by molar mass or Avogadro’s number to find the amount of desired substance
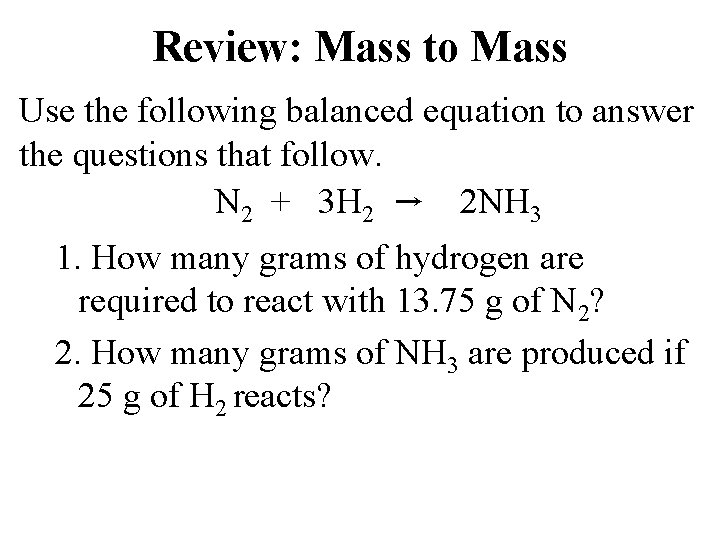
Review: Mass to Mass Use the following balanced equation to answer the questions that follow. N 2 + 3 H 2 → 2 NH 3 1. How many grams of hydrogen are required to react with 13. 75 g of N 2? 2. How many grams of NH 3 are produced if 25 g of H 2 reacts?
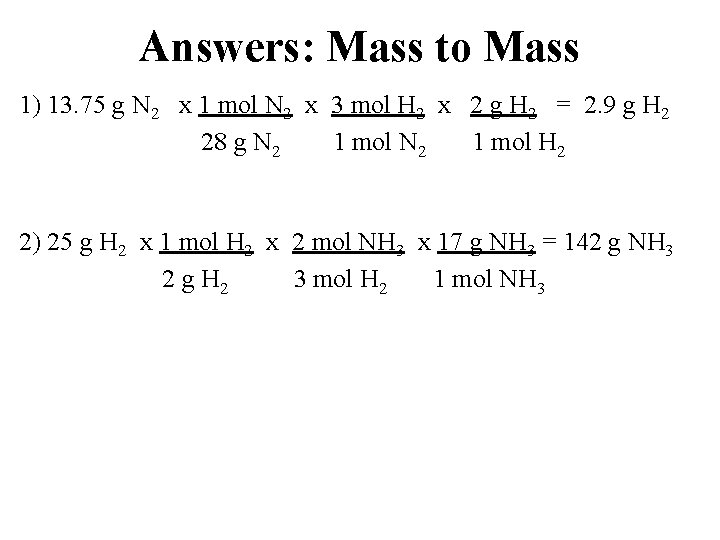
Answers: Mass to Mass 1) 13. 75 g N 2 x 1 mol N 2 x 3 mol H 2 x 2 g H 2 = 2. 9 g H 2 28 g N 2 1 mol H 2 2) 25 g H 2 x 1 mol H 2 x 2 mol NH 3 x 17 g NH 3 = 142 g NH 3 2 g H 2 3 mol H 2 1 mol NH 3

Limiting Reagents • The limiting reagent is the reactant that limits how much product can be made in a chemical reaction. – It gets completely used up in the reaction • The other reactant is called the excess reagent. – Some of it is left over after the reaction

Limiting Reagent Simulation • Go to following website: • http: //phet. colorado. edu/en/simulation/reactantsproducts-and-leftovers • Click ‘Run Now!’ button, and Click ‘OK’ button • Create the following sandwich equation: – 2 pieces of bread + 3 pieces of cheese – Which ingredient is the limiting reagent if you have the following amounts of ingredient in your refrigerator: • A. 7 pieces of bread and 6 pieces of cheese • B. 4 pieces of bread and 7 pieces of cheese

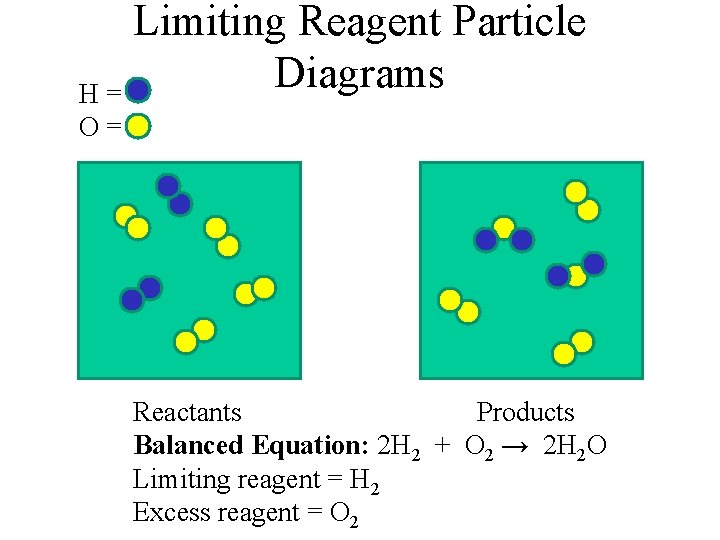
Limiting Reagent Particle Diagrams H= O= Reactants Products Balanced Equation: 2 H 2 + O 2 → 2 H 2 O Limiting reagent = H 2 Excess reagent = O 2

A Mole-Mole Limiting Reagent Problem • Use the balanced equation to solve the problem that follows: 3 Ca(s) + 2 P(s) → Ca 3 P 2(s) If 12. 7 mol Ca reacts with 9. 54 mol of P what is the limiting reagent? How many moles of Ca 3 P 2 are produced?

Mole-Mole Limiting Reagent Problems • Step 1: Set up 2 mole-mole problems 12. 7 mol Ca x 1 mol Ca 3 P 2 = 4. 23 mol Ca 3 P 2 3 mol Ca 9. 54 mol P x 1 mol Ca 3 P 2 = 4. 77 mol Ca 3 P 2 2 mol P • Step 2: Choose the reactant that makes less product. The limiting reagent is Ca. • Step 3: Choose the smaller amount of product. 4. 23 mol of Ca 3 P 2 can be produced.

Mass-Mass Limiting Reagent Problems • Step 1: Convert both starting substances to moles by dividing by molar mass. • Step 2: Use mole ratios to find moles of desired substance. Whichever reactant gives you a smaller answer here is the limiting reactant. Go on to step 3 with this reactant only • Step 3: Convert moles of desired substance to grams by multiplying by molar mass.
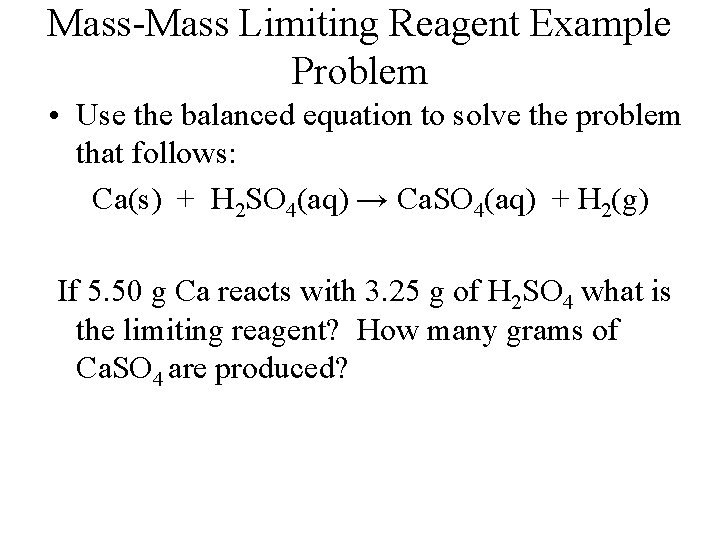
Mass-Mass Limiting Reagent Example Problem • Use the balanced equation to solve the problem that follows: Ca(s) + H 2 SO 4(aq) → Ca. SO 4(aq) + H 2(g) If 5. 50 g Ca reacts with 3. 25 g of H 2 SO 4 what is the limiting reagent? How many grams of Ca. SO 4 are produced?

Answer 5. 50 g Ca x 1 mol Ca. SO 4 = 0. 137226 mol Ca. SO 4 40. 08 g Ca 1 mol Ca 3. 25 g H 2 SO 4 x 1 mol Ca. SO 4 = 0. 033139 mol Ca. SO 4 98. 072 g H 2 SO 4 1 mol H 2 SO 4 is the limiting reagent. To determine how much Ca. SO 4 is produced convert 0. 033139 mol Ca. SO 4 to grams. 0. 033139 mol Ca. SO 4 x 136 g Ca. SO 4 = 4. 51 g Ca. SO 4 1 mol Ca. SO 4

Percent Yield • Percent yield tells you how close your lab yield is to the amount predicted by stoichiometry • You want percent yield to be as close to 100% as possible, but errors and happen and reaction conditions are difficult to control

Percent Yield • % Yield = actual yield x 100 theoretical yield • Actual yield: actual amount of product produced in experiment, will be given in the problem. • Theoretical yield: maximum amount of product that could be produced from a specific amount of reactant – You calculate this, it’s the smaller amount of product you get from doing the mass to mass problem if you have the masses of two reactants

Percent Yield Example: Determine the percent yield for the reaction between 46. 1 g of Ba and 13. 4 g of O 2 if 49. 8 g of Ba. O is produced. 2 Ba(s) + O 2(g) → 2 Ba. O(s) Actual yield = 49. 8 g Ba. O Theoretical yield: you must determine the limiting reagent and solve a mass-mass problem from the this reactant

Answer 46. 1 g Ba x 1 mol Ba x 2 mol Ba. O = 0. 3357 mol Ba. O 137. 33 g Ba 2 mol Ba 13. 4 g O 2 x 1 mol O 2 x 2 mol Ba. O = 0. 8376 mol Ba. O 31. 998 g O 2 1 mol O 2 Ba is the limiting reagent. Convert 0. 3357 mol Ba. O to grams to get theoretical yield. 0. 3357 mol Ba. O x 153. 329 g Ba. O = 51. 5 g Ba. O 1 mol Ba. O % Yield = 49. 8 g Ba. O x 100 = 96. 7 % 51. 5 g Ba. O