Calculation of dissociative electron attachment cross sections Daniel






































- Slides: 45

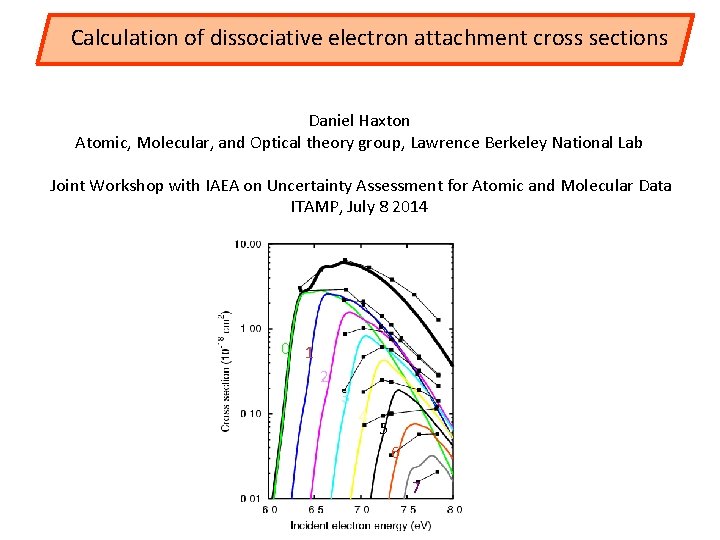
Calculation of dissociative electron attachment cross sections Daniel Haxton Atomic, Molecular, and Optical theory group, Lawrence Berkeley National Lab Joint Workshop with IAEA on Uncertainty Assessment for Atomic and Molecular Data ITAMP, July 8 2014 0 1 2 3 4 5 6 7

Calculation of dissociative electron attachment cross sections D. E. A. : AB + e- A- + B Dissociative Electron Attachment (DEA) is a basic physical process that may occur in plasmas, or in everyday materials bombarded by ionizing radiation. Reactive products: ions and radicals. CF + e- C* + FH 2 + e- H + HCHOOH + e- CHO 2 - + H DEA leads to damage in technological and biological systems.

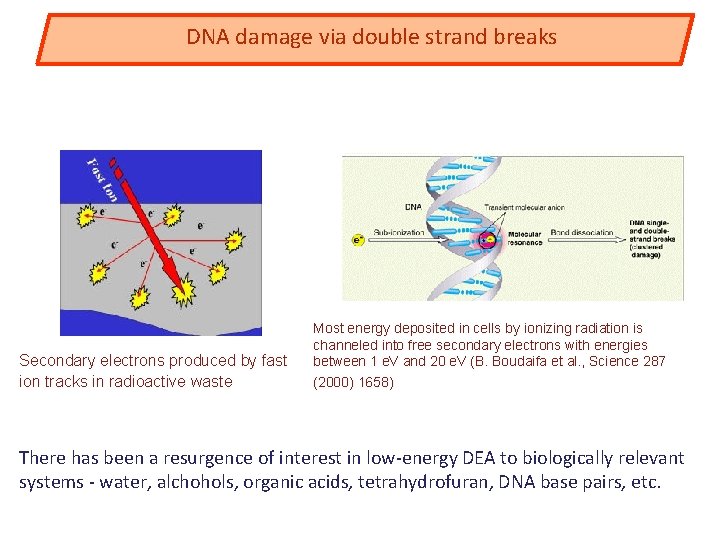
DNA damage via double strand breaks Secondary electrons produced by fast ion tracks in radioactive waste Most energy deposited in cells by ionizing radiation is channeled into free secondary electrons with energies between 1 e. V and 20 e. V (B. Boudaifa et al. , Science 287 (2000) 1658) There has been a resurgence of interest in low-energy DEA to biologically relevant systems - water, alchohols, organic acids, tetrahydrofuran, DNA base pairs, etc.

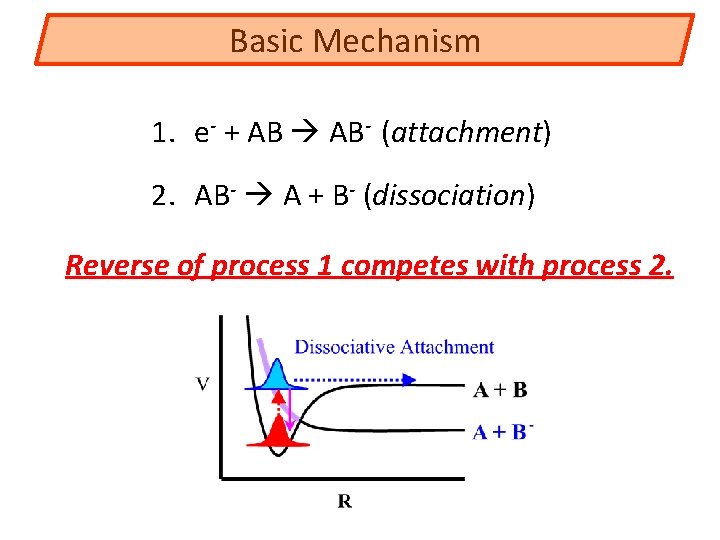
Basic Mechanism 1. e- + AB AB- (attachment) 2. AB- A + B- (dissociation) Reverse of process 1 competes with process 2.

Basic Mechanism Resonant processes include DEA Nonresonant processes

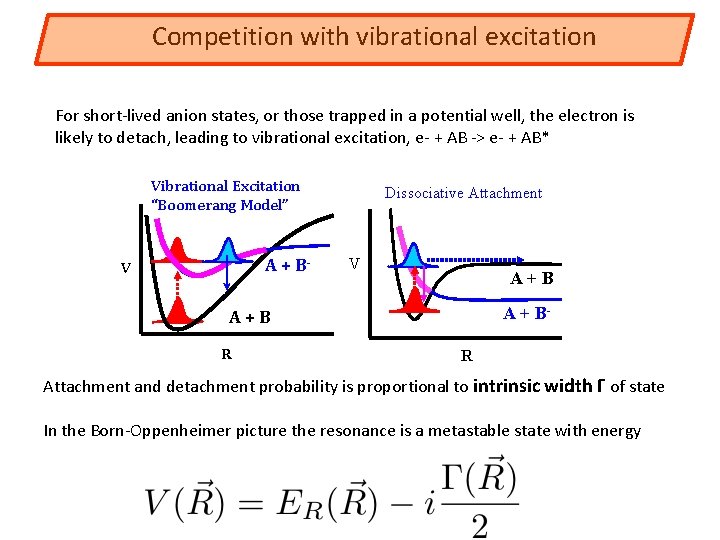
Competition with vibrational excitation For short-lived anion states, or those trapped in a potential well, the electron is likely to detach, leading to vibrational excitation, e- + AB -> e- + AB* Vibrational Excitation “Boomerang Model” A + B- V Dissociative Attachment V A+B A + B- A+B R R Attachment and detachment probability is proportional to intrinsic width Γ of state In the Born-Oppenheimer picture the resonance is a metastable state with energy

Summary - Basics Dissociative electron attachment is described by TWO STEPS Big picture: calculating FIRST STEP (attachment) is relatively easy. If second step (dissociation) goes 100% (survival probability is 100%), then calculating second step is not necessary to get total cross section. Survival probability (and branching ratios) associated with second step may be VERY DIFFICULT to calculate requiring major effort, if the polyatomic nuclear dynamics is complicated. So if the molecule takes a time tdiss to dissociate, the cross section depends on the width as

Summary - Basics So uncertainty in dissociative electron attachment (DEA) cross section depends upon survival probability Survival probability given roughly by ratio of DEA to vibrational excitation So prior knowledge of this ratio (from experiment or theory) should affect uncertainty in DEA cross section. ( Isotope effect is also due to survival probability )

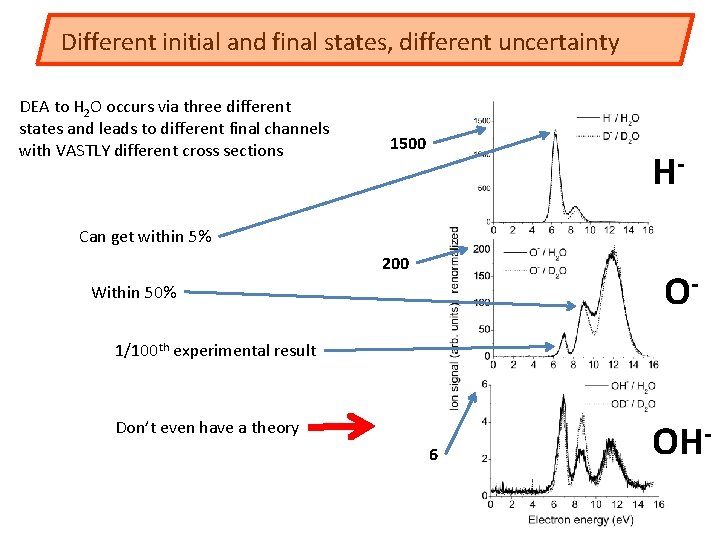
Different initial and final states, different uncertainty DEA to H 2 O occurs via three different states and leads to different final channels with VASTLY different cross sections 1500 H- Can get within 5% 200 O- Within 50% 1/100 th experimental result Don’t even have a theory 6 OH-

Angular distributions Combination of experiment and theory allows us to determine that the molecule dissociates into the three-body channel via scissoring backwards 10

Angular distributions Our interest currently is in angular distributions because they can tell us about dynamics. Combination of experiment and theory allows us to determine that the molecule dissociates into the three-body channel via scissoring backwards 11

Angular distributions Our interest currently is in angular distributions because they can tell us about dynamics. Combination of experiment and theory allows us to determine that the molecule dissociates into the three-body channel via scissoring backwards 12

Angular distributions Our interest currently is in angular distributions because they can tell us about dynamics. Combination of experiment and theory allows us to determine that the molecule dissociates into the three-body channel via scissoring backwards 13

Angular distributions Acetylene AXIAL RECOIL 30 DEGREES MORE ELABORATE TREATMENT 14 Calculations / experiment indicate breakup at ~30 degrees H-C-C bond angle consistent with Orel and Chorou PRA 77 042709

Complex Kohn Method for Electron-Molecule Scattering Complex Kohn Electron-Molecule Scattering Code: Developed 1987 -1995 T. N. Rescigno, A. E. Orel, B. Lengsfield, C. W. Mc. Curdy Lawrence Livermore National Lab, Lawrence Berkeley National Lab The “Kohn Suite” consists of scattering codes coupled to MESA, a flexible electronic structure code from Los Alamos written in the 1980 s and no longer maintained. Continuum Functions Quantum Chemistry Complex Kohn Variational Method: Stationary principle for the T-Matrix (scattering amplitude), Walter Kohn

Complex Kohn Method for Electron-Molecule Scattering 3 parts of wave function for Kohn method in usual implementation. Similar capabilities as UK R-matrix. Only in particular situations are there significant differences in Kohn or R-matrix capabilities.

Complex Kohn Method for Electron-Molecule Scattering Limitations of Present Capabilities Ø Small size of Systems – Small Polyatomics 6 -10 atoms maximum but only limited target response for more than ≈5 atoms Ø Highly Correlated Target States only for smaller systems – strongly target states ≈ 5, 000 - 10, 000 configurations Ø Energies < ≈ 50 e. V and low asymptotic angular momentumset for inner region of continuum functions Poor Computational efficiency – Recently removed the limit of 160 orbitals, but serial calculations with legacy code require weeks of computation – No parallel versions of either structure or scattering codes. Ø Ø NEW IMPLEMENTATION HAS BEEN PLANNED (Rescigno, Mc. Curdy, Lucchese)

Complex Kohn Method for Electron-Molecule Scattering But the future looks promising for calculating total widths (lifetimes). Advancements in Kohn suite – Mc. Curdy Rescigno Lucchese Electronic structure methods for metastable states (Sci. DAC project) It’s the survival probability that’s the problem.

Dissociative Attachment to CO 2 e- - CO 2 DEA and vibrational excitation have been studied since the 1970 s Ø 4 e. V 2Πu shape resonance produces O- and vib excitation Ø 8. 2 e. V 2Πg Feshbach resonance produces O- Ø Ø 13 e. V Feshbach resonance produces O- Ø Three DEA peaks identified by Sanche in CO 2 films at 8. 2 e. V 11. 2 e. V and 15 e. V in 2004 Schulz measured O 2 - from an 11. 2 e. V resonance in 1970 s Chantry (1972) and Fayard (1976)

Dissociative Attachment to CO 2 DEA is minor channel; mostly vibrational excitation. 1. 5 x 10 -15 cm 2 total cross section 1. 5 x 10 -16 cm 2 vibrational excitation Mc. Curdy Isaacs Meyer Rescigno PRA 67, 042708 (2003) DEA cross section: 1. 5 x 10 -19 cm 2

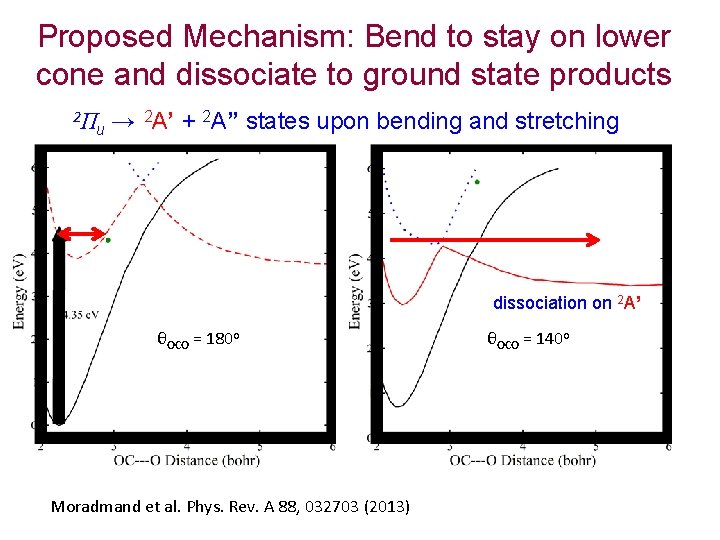
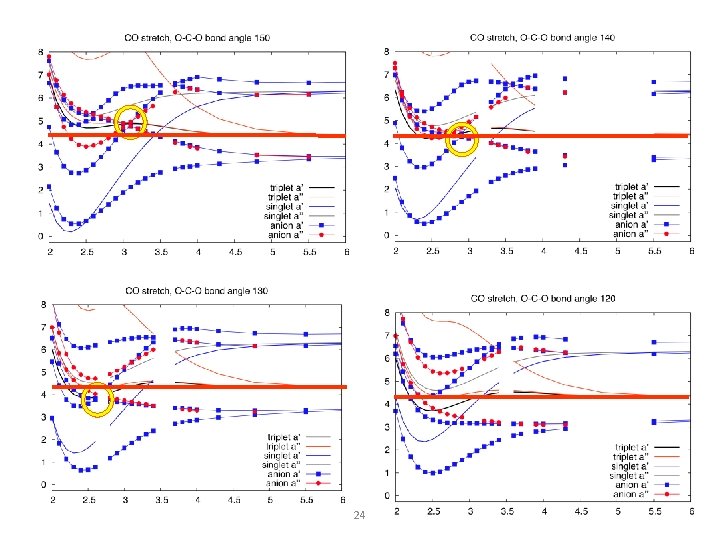
Dissociative Attachment to CO 2 Width of (one component of the) resonance is very large when molecule is bent. STRONG effect of lifetime on final breakup channel. Mc. Curdy Isaacs Meyer Rescigno PRA 67, 042708 (2003)

Dissociative Attachment to CO 2 Feshbach resonance conical intersection CO 2 - shape resonance CO 2 ground state (CAS + single and doubles CI on both neutral and anion states) Dashed = neutral, solid colored = anion 3 components of O- 2 P make a 2Π resonance and a 2Σ virtual state

Proposed Mechanism: Bend to stay on lower cone and dissociate to ground state products 2Π u → 2 A’ + 2 A” states upon bending and stretching dissociation on 2 A’ θOCO = 180 o Moradmand et al. Phys. Rev. A 88, 032703 (2013) θOCO = 140 o

24

NO 2

Dissociative Recombination of NO 2+ + e. NO 2+ ground neutral NO 2 excited states Calculation done blind, no experiment now or then Step 1: Identify candidate states! Attachment at zero electron energy. Work done with Chris Greene at JILA, University of Colorado Boulder

Dissociative Recombination of NO 2+ + e. Candidates for direct DR

Dissociative Recombination of NO 2+ + e. Simple estimate of cross section as function of energy

Dissociative Recombination of NO 2+ + e-

Dissociative Recombination of NO 2+ + e. Put the pieces together

Dissociative Recombination of NO 2+ + e. The result Highly sensitive to position of resonant states in this case.

Dissociative Attachment to H 2 O 3 resonance states, with multiple products from 2 B each O- production 1 2 B 2 A 2 B - H production 2 1 2 A 1 C. E. Melton, J. Chem. Phys. , 57, 4218 (1972) H 2 O + e- H 2 O- (2 B 1, 2 A 1, 2 B 2) { 1 H- + OH (2 ) H + OH 2 + O-

Dissociative Attachment to H 2 O Calculations have Revealed Different Dynamics of the Resonances in H 2 O 1. H- is produced from the 2 B 1 resonance directly 1. O- production from 2 B 2 resonance comes from passage through conical intersection to 2 A 1 surface. 2. O- production from 2 A 1 comes from three body breakup O+H+H.

Dissociative Attachment to H 2 O A Complete ab initio Treatment of Polyatomic Dissociative Attachment 1. 1. Electron scattering: Calculate the energy and width of the resonance for fixed nuclei – Complex Kohn calculations produce – CI calculations with ~ 900, 000 configurations produce – Fitting of complete resonance potential surface to dissociation Nuclear dynamics in the local complex potential model on the anion surface – – Multiconfiguration Time-Dependent Hartree (MCTDH) Flux correlation function (energy resolved projected flux) calculation of DA cross sections

Dissociative Attachment to H 2 O Complex Potential Energy Surfaces V(r 1, r 2, ) = ER - i /2 r 1 r 2 = h/ is lifetime

Dissociative Attachment to H 2 O Local complex potential model Dynamics on complex potential energy surface. In general this theory is sufficient for DEA. Derivation: given L 2 approximation to resonant state, φ, define effective Hamiltonian for that state. Feshbach partitioning:

Dissociative Attachment to H 2 O Local complex potential model: HOWEVER are many systems requiring more elaborate (nonlocal) treatment of effective operator – Horacek, Houfek, Domcke, others, e. g. Electron scattering in HCl: An improved nonlocal resonance model Phys. Rev. A 81, 042702 (2010) J. Fedor, C. Winstead, V. Mc. Koy, M. Čížek, K. Houfek, P. Kolorenč, and J. Horáček

Complete 2 B 1 (2 A’’) Potential Surface = 00 150 - O + H 2 700 - OH +H 350 104. 50 1250 - OH +H 1500 1800 r 1 O H 38 r 2 H

Dissociative Attachment to H 2 O Triatomic rovibrational dynamics calculated with Multiconfiguration Time-Dependent Hartree Method Adaptive method capable of handling multidimensional vibrational dynamics E. g. malonaldehyde 24 atoms H. D. Meyer et al, University of Heidelberg Cross section from energy resolved projected flux. Significant but manageable expense involved in computing a double Fourier transform.

Cross Sections for OH vibrational states compared with experiment 5. 99 vs 6. 5 10 -18 cm 2 0 1 2 3 4 5 6 7 D. S. Belic, M. Landau and R. I. Hall, Journal of Physics B 14, pp. 175 -90 (1981) 40 Calc. Shifted by in incident energy by +0. 34 e. V

Dissociative Attachment to H 2 O

Dissociative Attachment to H 2 O H- from 2 A 1 (middle peak) ~1 x 10 -18 cm 2 but overlaps 2 B 1 ~5 x 10 -19 cm 2 2 A 1

Dissociative Attachment to H 2 O Total O- production all states We got lucky with 2 B 2 Very happy with this level of agreement for 2 A 1 Very little O- from 2 B 1. . . even with Renner-Teller coupling to 2 A 1. . . subtleties of PES?

Conclusion IF we assume that DEA is driven by the direct, resonant process THEN the source of major uncertainty is the survival probability i. e. uncertainty in DEA is a function of ratio of vibrational excitation to DEA, and i. e. uncertainty in DEA is function of isotope effect, so as long as these are known a priori, from experiment or theory, even with low accuracy, the model should give higher uncertainty in theoretical result. Equivalently if the width is known to be large. Or if the width is known to be large in certain geometries and there is a decent chance of sampling those geometries. ALSO the precise energetics MAY give additional sensitivity to error Atomic, molecular, and optical theory group at LBNL CW Mc. Curdy TN Rescigno CY Lin J Jones X Li CS Trevisan AE Orel B Abeln Z Walters

Complex Kohn Method for Electron-Molecule Scattering