CALCULATING VANDERVAAL FORCES BETWEEN MACROSCOPIC FORCES CONTENT INTRODUCTION

CALCULATING VANDERVAAL FORCES BETWEEN MACROSCOPIC FORCES

CONTENT • INTRODUCTION • ATTRACTION BETWEEN TWO SEMI INFINITE BLOCKS • CALCULATION OF ENERGIES • MAGNITUDES OF HAMAKER CONSTANT • VANDER WAALS INTERACTION FOR OTHER GEOMETRIES

INTRODUCTION • Requires that all pairwise combinations of intermolecular attraction between the two bodies be summed. • Considering the case of blocks of material with planar surfaces. • Done by the Hamaker.
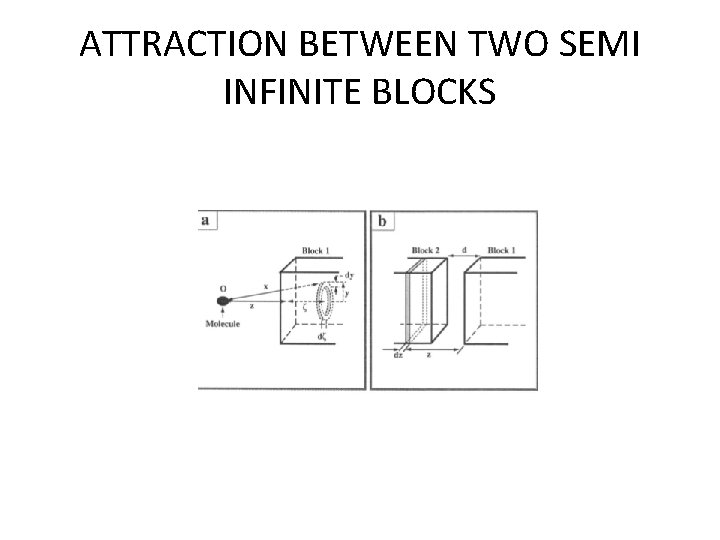
ATTRACTION BETWEEN TWO SEMI INFINITE BLOCKS

• From the diagram, considering • (a) interaction between a molecule and a block of material • (b) interaction between two blocks of material. • Material placed in O and distance Z from surface of the material and distance X be the ring shaped volume element.

• The volume of this ring is given by d. V = 2πζy dy ζ. Therefore the increment of interaction between the molecule and the block due to the molecules a distance x from the point 0 is given by d. Ф=-(ᵨNA/M)ᵦ 2πy. X 6 dy dζ eq 1 • We assume the ring is located a distance ζ inside the surface of the block; then X 2 =( Z+ ζ)2 + Y 2 eqn 2



• Therefore the integration of Equation (1)over the block gives • Ф = -(ᵨNA/M)ᵦπ/ 6 Z 3 Suppose point 0 is located inside a second block of material, recognize that all atoms in a slice of the second block a distance Z from the first block will be attracted toward the second with an energy given by above equation. • we position a volume element of thickness dz at this location in the second block, we realize that it contains •

(ᵨN/M) volumes per unit area. Then increment of the attraction per unit area due to the slice of the material. d. Ф = -(ᵨNA/M)2 ᵦπ/6 z-3 dz Integrated over values of z between the distance of closest approach d and infinity. The result of this integration gives the potential energy of attraction per unit area between two blocks of infinite extension,


• The hamaker constant has the energy units length 6 and the term parentheness and has the volume. • Though it has expressed in units per area of the facing surfaces. • Hence it depend upon the geometries of the bodies involved.

MAGNITUDES OF THE HAMAKER CONSTANTS Stipulations involved due to conventional methods. • Assume that the dispersion component is the dominant contributor to the attraction; therefore f 3 = (3/4)hν((α/47 rᵋ). • Recognize that (p. N, /M) is the reciprocal of the volume per molecule, and that (a/47 rᵋ, ) is typically about 10% the magnitude of atomic volume; therefore A = ( 3/4)n 2 h ν(0. 1)2. The quantity h ν is the same order of magnitude as the ionization potential.

VANDER WAAL INTERACTIONS FOR OTHER GEOMETRIES

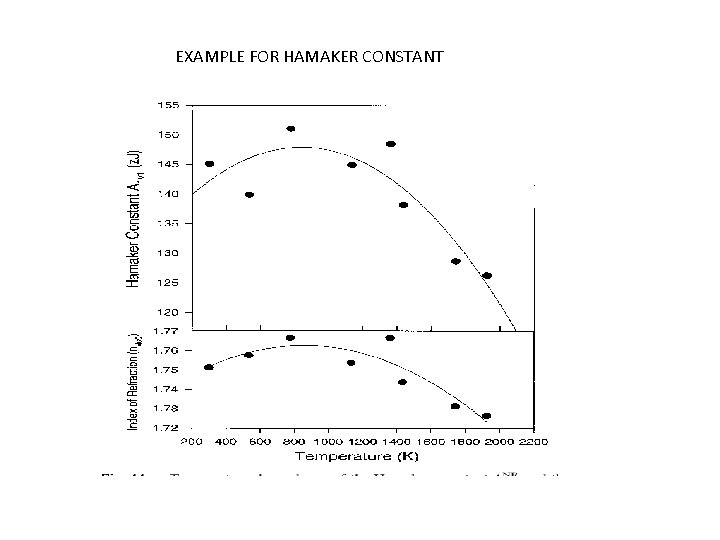
EXAMPLE FOR HAMAKER CONSTANT

REFERENCES • Principles of colloids and surface chemistry, HIEMENZ AND RAJAGOPALAN. , third edition. , Guided by , Dr. k. Geetha, Assistant Professor , Periyar Manniyammai Institute of science and Technology, Thanjavur.
- Slides: 17