Calculating Number of Entities Calculating Number of Entities

Calculating Number of Entities

Calculating Number of Entities (N) from Mass We are not able to count the number of atoms in reactions; we must count them indirectly by measuring out a certain mass of a substance and calculating the number of moles. In order to estimate the number of entities in a large sample we need to take a small sample and find out the mass of a small number of entities.

Calculating Number of Entities and Avogadro’s number • There are times when need to know exactly how many atoms are in something. This is especially true when dealing with gases as they change in pressure with temperature. • To find the number of entities we use the formula

• N = n. NA • Number of entities = (# of mols) x(Avogadro's #)

Example • Find the number of atoms in 0. 004 moles of water. • Find the number of molecules in 30. 0 g of Cl 2 (g)

Determining the number of particles in a mole

Avogadro’s Number as Conversion Factor 6. 02 x 1023 particles 1 mole or 1 mole 6. 02 x 1023 particles Note that a particle could be an atom OR a molecule!
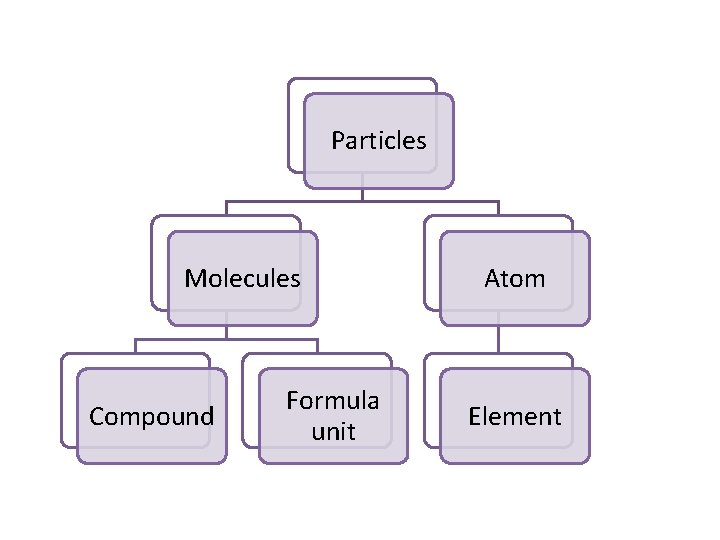
Particles Molecules Compound Formula unit Atom Element

Equation to find number of particles! x 1023 number of moles 6. 02 x particles = number of 1 mole Example: How many particles of sucrose are in 3. 50 moles?

Equation to find number of moles! number of particles x number of moles 1 mole = 6. 02 x 1023 particles Example: How many moles are contained in 4. 50 x 1024 atoms of zinc?

Learning Check 1. Number of atoms in 0. 500 mole of Al 2. Number of moles of S in 1. 8 x 1024 S atoms

Molar Mass

Molar Mass The Mass of 1 mole (in grams) Molar mass is equal to the atomic mass (look on the periodic table) 1 mole of C atoms= 12. 01 g 1 mole of Au atoms = 196. 97 g 1 mole of Cu atoms = 63. 55 g Always round to the hundredths place.

Mole to Mass Conversion Where will you find the number of grams per mole? Number of moles x number of grams = mass 1 mole The Periodic Table Example: Calculate the mass in grams of 0. 0450 moles of chromium. 0. 0450 moles Cr x 52. 0 g Cr = 2. 34 g Cr 1 mol Cr Look at your Periodic Table

Mass to Mole Conversion Mass x Where will you find the number of grams per mole? 1 mole = number of moles number of grams The Periodic Table Example: How many moles of calcium are there in 525 grams of calcium? 525 g Ca x 1 mol Ca = 13. 1 mol Ca 40. 1 g Ca Look at your Periodic Table

Mass to Atoms Conversion Two steps: 1. Use the mass to moles conversion 2. Now calculate the number of atoms using the moles to particles equation.

Example: How many atoms of gold are in a pure gold nugget having a mass of 25. 0 g?

Atoms/Molecules and Grams How many atoms of Cu are present in 35. 4 g of Cu? 35. 4 g Cu 1 mol Cu 1023 atoms Cu 63. 5 g Cu mol Cu = 3. 4 X 1023 atoms Cu 6. 02 X 1
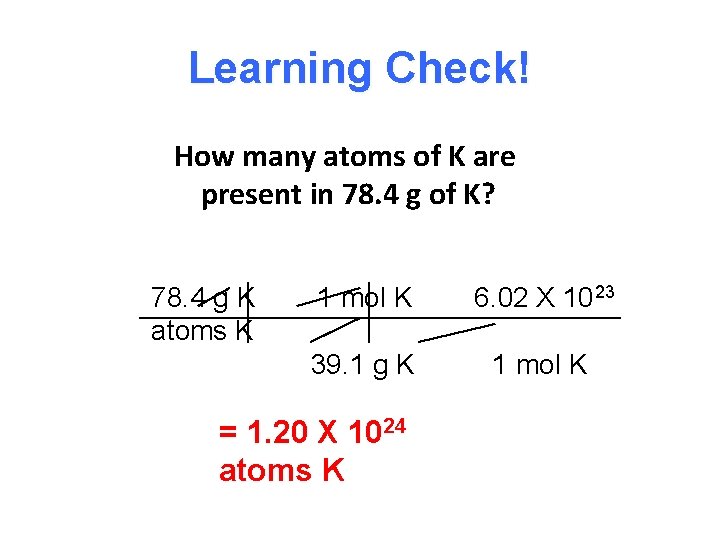
Learning Check! How many atoms of K are present in 78. 4 g of K? 78. 4 g K atoms K 1 mol K 6. 02 X 1023 39. 1 g K 1 mol K = 1. 20 X 1024 atoms K
- Slides: 19