Ca PO 4 PTH VIT D Calcium Phosphorus



























































- Slides: 81

Ca++, PO 4, PTH & VIT D Calcium, Phosphorus & Vitamin D In Chronic Renal Failure By Dr. Rick Hiller

Phosphorus Measurement and Balance • Normal concentration between 2. 5 and 4. 5 mg/dl. • 85% of total body stores are contained in bone (hydroxyapatite), 14% is intracellular, and 1% extracellular.

Phosphorus Measurement and Balance • 70% of the extracellular phosphorus is organic (phospholipids) and the remaining 30% is inorganic. • 15% of the inorganic is protein bound; the remaining is complexed with sodium, magnesium, or calcium or circulates as free monohydrogen or dihydrogen forms. • This freely circulating phosphorus is what is measured.

Phosphorus Measurement and Balance • 2/3 of ingested phosphorus is excreted in urine; the remaining in stool. • Foods high in phosphorus are also high in protein. • Three organs are involved in phosphate homeostasis: intestine, kidney, and bone. • Major hormones involved are Vit. D and PTH

Phosphorus Homeostasis • 60 -70% of dietary phosphorus is absorbed by the GI tract via: – Passive transport – Active transport stimulated by calcitriol and PTH • Antacids, phosphate binders, and calcium bind to phosphorus, decreasing the free amount available for absorption

Phosphorus Homeostasis • Inorganic phosphorus is freely filtered by the glomerulus. • 70 -80% is then reabsorbed in the proximal tubule. The remaining is reabsorbed in the distal tubule. • Phosphorus excretion can be increased primarily by increasing plasma phosphorus concentration and PTH.

Phosphorus Homeostasis • Phosphorus excretion can also be increased to a lesser degree by volume expansion, metabolic acidosis, glucocorticoids, and calcitonin. • This regulation occurs in the proximal tubule via the sodium-phosphate cotransporter.


Calcium Measurement and Balance • Normal Concentration between 8. 5 and 10. 5 mg/d. L • Serum levels are 0. 1 -0. 2% of extracellular calcium; this is only 1% of total body calcium • The remainder of total body calcium is stored in bone.

Calcium Measurement and Balance • Ionized calcium is physiologically active and is 40% of total serum calcium. • Non-ionized calcium is bound to albumin, citrate, bicarbonate, and phosphate • Ionized calcium can be corrected from total calcium by adding 0. 8 mg/d. L for every 1 mg decrease in serum albumin below 4 mg/d. L

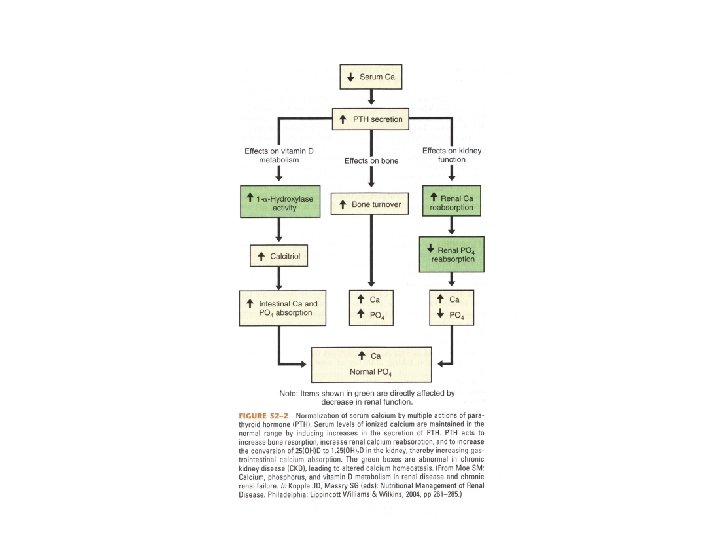
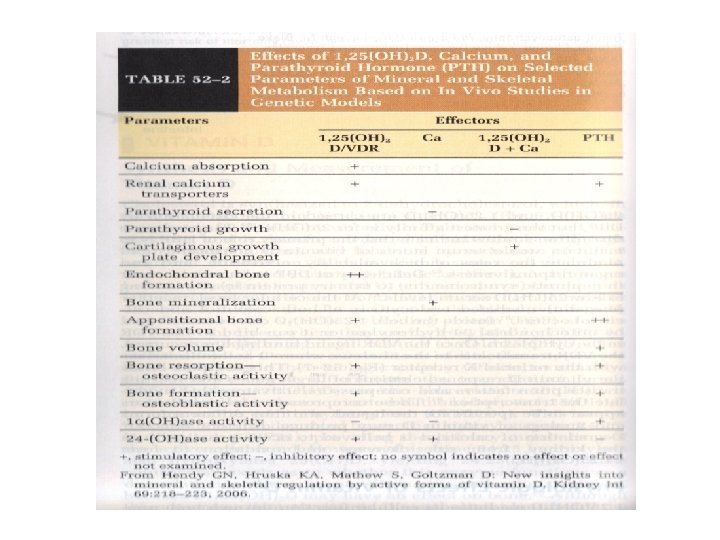
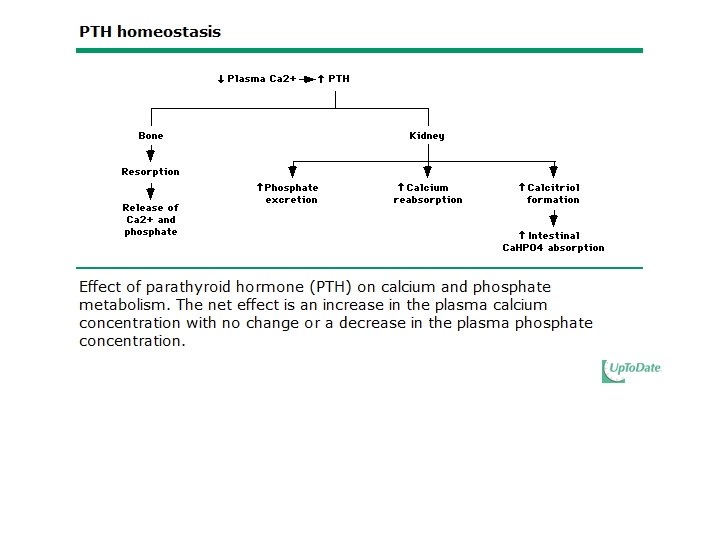
Calcium Measurement and Balance • PTH regulates serum ionized calcium by – Increasing bone resorption – Increasing renal calcium reabsorption – Increasing the conversion of 25(OH)D to 1, 25(OH)2 D in the kidney which increases the GI absorption of calcium


Calcium Measurement and Balance • Decreased PTH and Vit. D maintain protection against calcium overload by increasing renal excretion and reducing intestinal absorption.

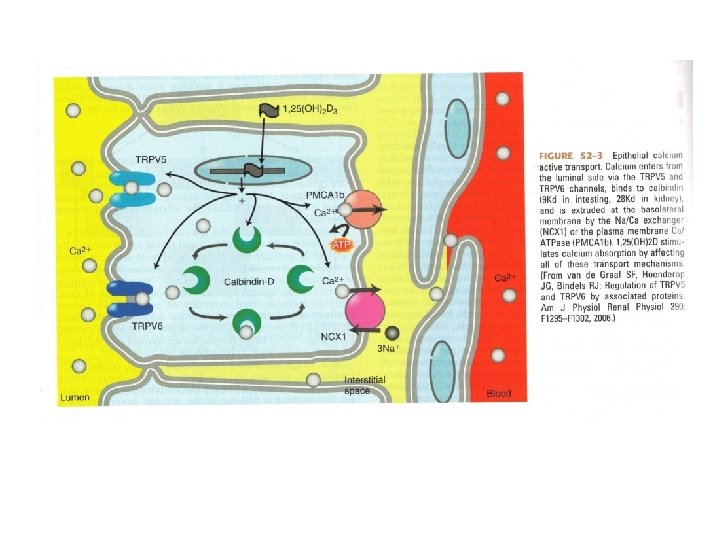
Calcium Homeostasis • Calcium absorption primarily occurs in the duodenum through Vit. D dependent and Vit. D independent pathways. • 60 -70% of calcium is reabsorbed passively in the proximal tubule, with another 10% reabsorbed in the thick ascending limb


Calcium-Sensing Receptor • Expressed in organs controlling calcium homeostasis: parathyroid gland, thyroid C cells, intestines, and kidneys. • Expression is regulated by 1, 25(OH)2 D

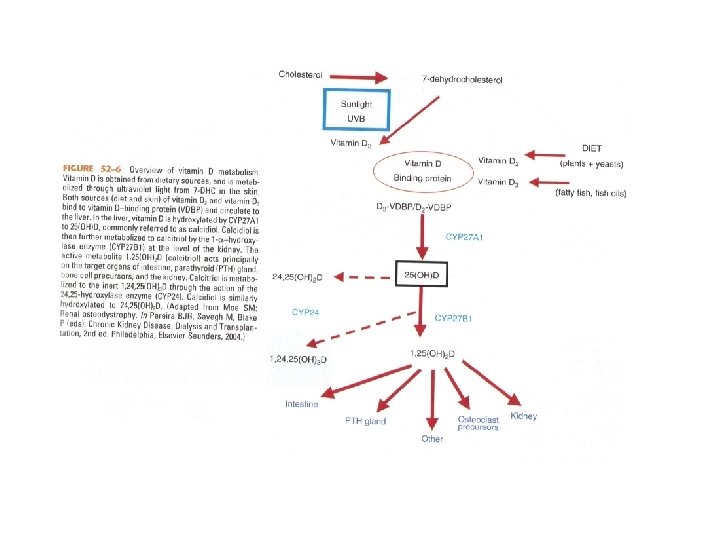
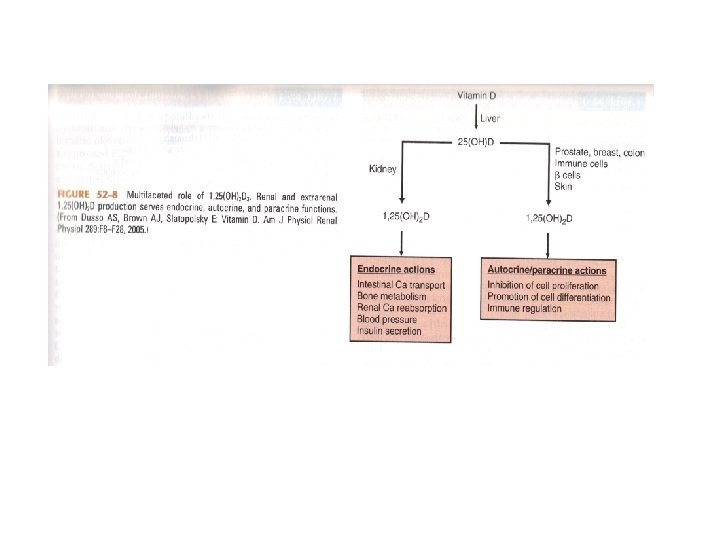
Synthesis and Measurement of Vitamin D • Vitamin D 3 is metabolized in the skin from 7 -dehydrocholesterol • Vitamin D 2 (ergocalciferol) is obtained in the diet from plant sources • Vitamin D 3 (cholecalciferol) is also obtained in the diet from animal sources

Synthesis and Measurement of Vitamin D • In the Liver, Vitamins D 2 and D 3 are hydroxylated to 25(OH)D (calcidiol) • Calcidiol then travels to the kidney where it is converted to 1, 25(OH)2 D


Physiologic Effects of Vitamin D • Facilitates the uptake of calcium in the intestinal and renal epithelium • Enhances the transport of calcium through and out of cells • Is important for normal bone turnover

Physiologic Effects of Vitamin D • Elevated serum PTH increases the hydroxylation of Vitamin D in the kidney • This causes a rise in serum calcium and feeds back to the parathyroid gland decreasing PTH secretion



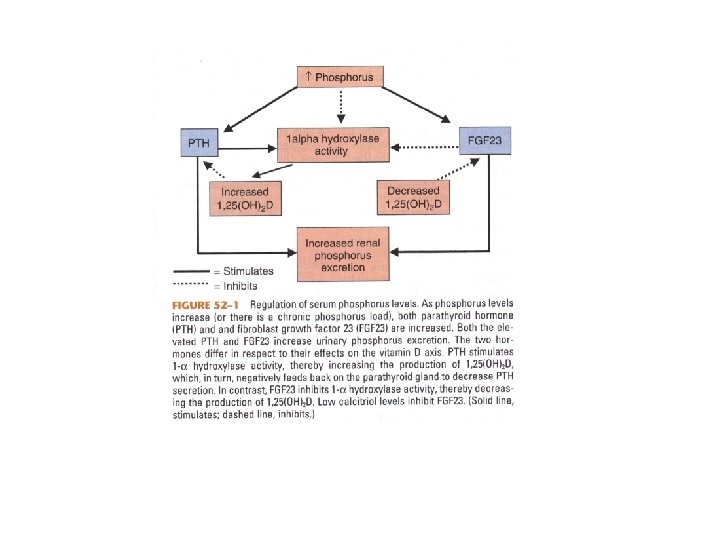
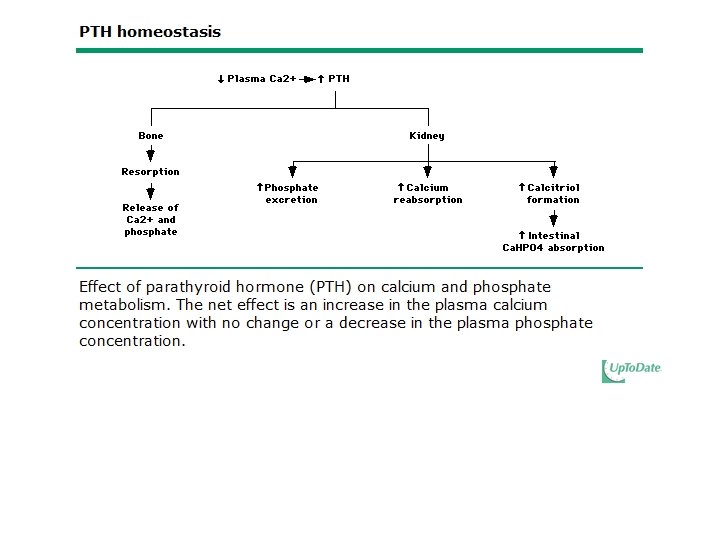
Regulation and Biologic Effects of Parathyroid Hormone • Primary function of PTH is to maintain calcium homeostasis by: – Increasing bone mineral dissolution – Increasing renal reabsorption of calcium and excretion of phosphorus – Increasing activity of renal 1 -α-hydroxylase – Enhancing GI absorption of calcium and phosphorus



Regulation of Parathyroid Hormone • Hypocalcemia is more important in stimulating PTH release • Normal or elevated Calcitriol is more important in inhibiting PTH release

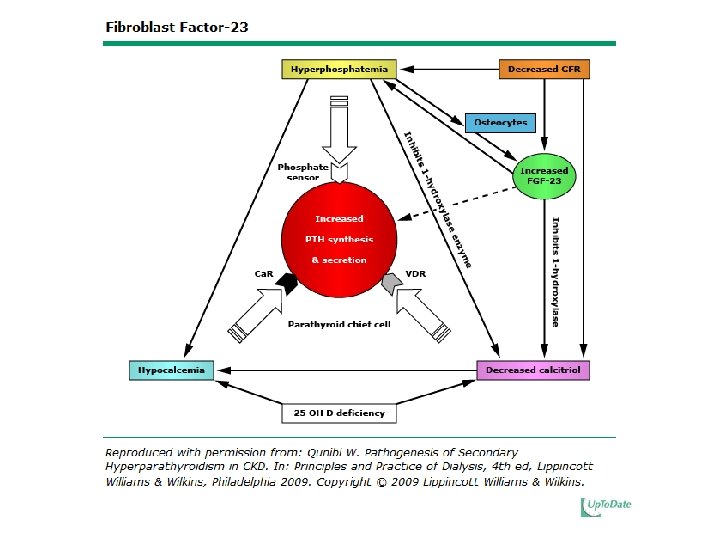
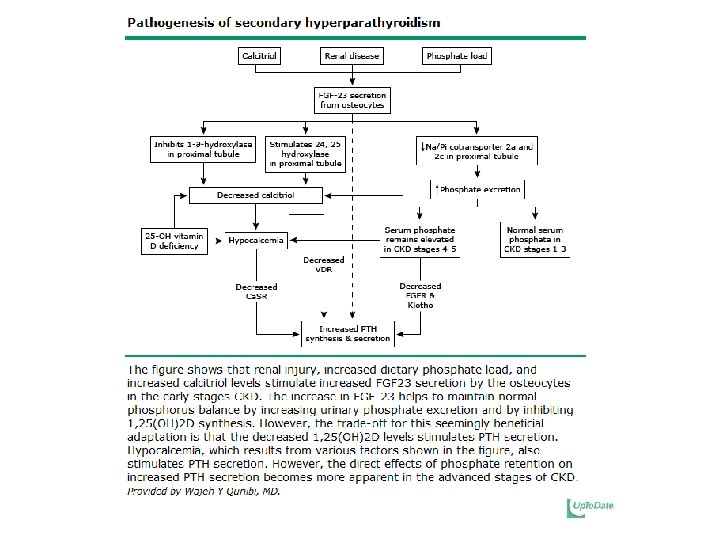
Regulation of Parathyroid Hormone • Increased PTH in Secondary Hyperparathyroidism is due to: – Loss of renal mass – Low 1, 25(OH)2 D – Hyperphosphatemia – Hypocalcemia – Elevated FGF-23


Measurement of PTH • Plasma PTH levels provide: – a noninvasive way to initially diagnose renal bone disease – Allow for monitoring of the disorder – Provide a surrogate measure of bone turnover in patients with CKD


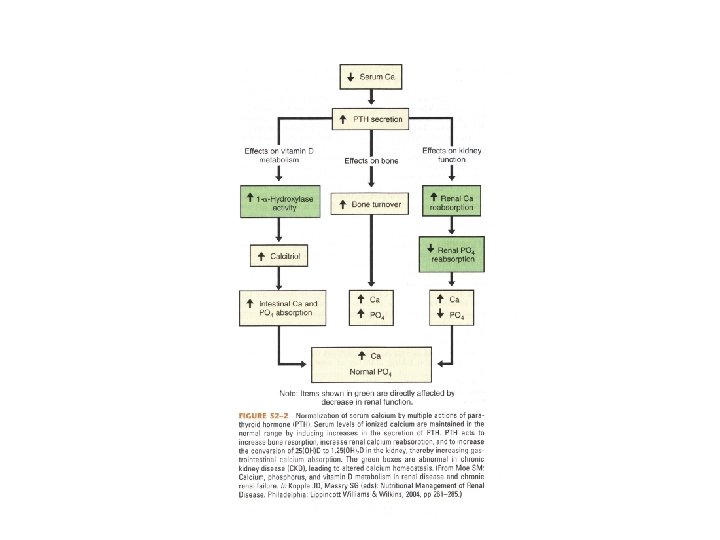
Effects of CKD • Chronic Renal Failure disrupts homeostasis by: – Decreasing excretion of phosphate – Diminishing the hydroxylation of 25(OH)D to calcitriol – Decreasing serum calcium • Leads to Secondary Hyperparathyroidism


Secondary HPT • Initially, the hypersecretion of PTH is appropriate to normalize plasma Ca 2+ and phosphate concentrations. • Chronically, it becomes maladaptive, reducing the fraction of filtered phosphate that is reabsorbed from 80 -95% to 15%

Secondary HPT • Secondary HPT begins when the GFR declines to <60 ml/min/1. 73 m 2 • Serum Ca 2+ and PO 4 levels remain normal until GFR declines to 20 ml/min/1. 73 m 2 • Low levels of calcitriol occur much earlier, possibly even before elevations in i. PTH.

Secondary HPT • Secondary HPT tries to correct: – hypocalcemia by increasing bone resorption – Calcitriol deficiency by stimulating 1 hydroxylation of calcidiol (25 -hydroxyvitamin D) in the proximal tubule

Hypocalcemia • Total Serum Calcium usually decreases during CKD due to: – Phosphate retention – Decreased calcitriol level – Resistance to the calcemic actions of PTH on bone

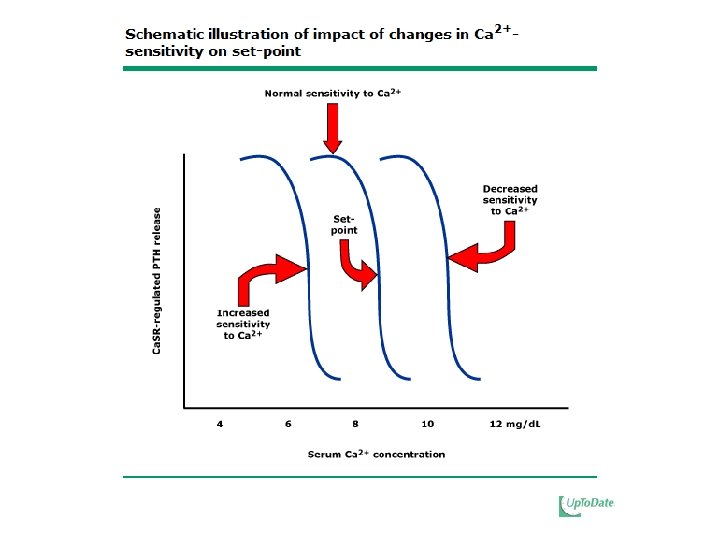
Hypocalcemia • Potent stimulus to the release of PTH – Increases m. RNA levels via posttranscription – Stimulates proliferation of parathyroid cells • Plays a predominant role via Ca. SR: • Major therapeutic target for suppressing parathyroid gland function




Decreased Vitamin D • Decreases calcium and phosphorus absorption in the GI tract. • Directly increases PTH production due to the absence of the normal suppressive effect of calcitriol • Indirectly increases secretion of PTH via the GI mediated hypocalcemic stimulus

Decreased Vitamin D • Administering calcitriol to normalize plasma levels can prevent or reverse secondary HPT • Calcitriol deficiency may change the set point between PTH and plasma free calcium


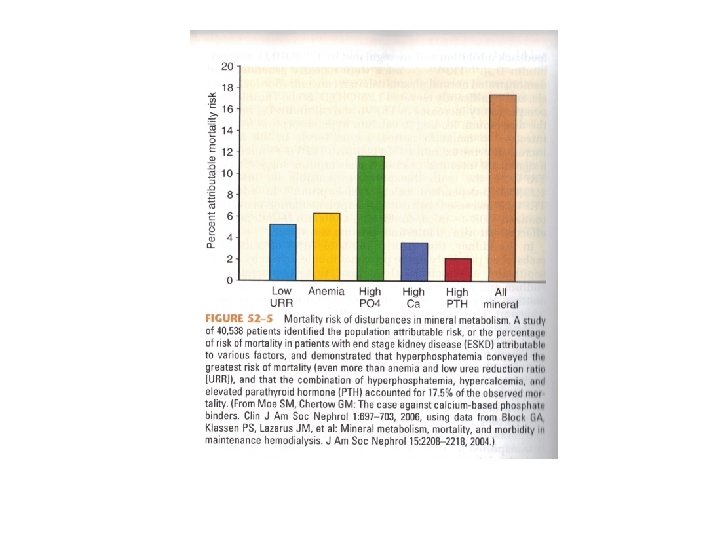
Mechanisms by which Phosphate Retention may lead to HPT • Diminishes the renal production of calcitriol • Directly increases PTH gene expression • Hyperphosphatemia, hypocalcemia, and elevated PTH account for ~17. 5% of observed, explainable mortality risk in HD patients with the major cause of death being cardiovascular events


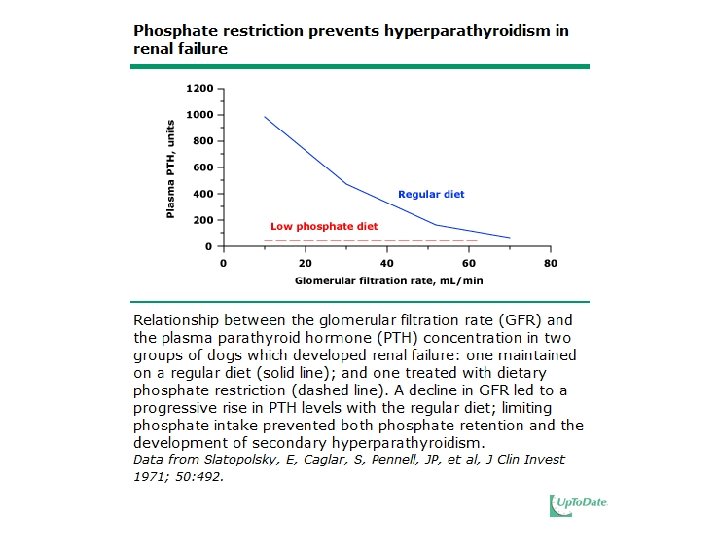
Secondary HPT • If phosphate retention is prevented, then secondary hyperparathyroidism does not occur.

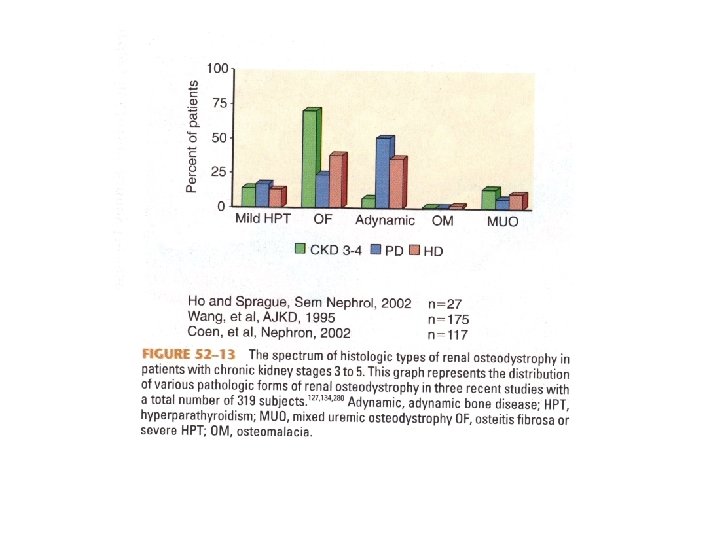
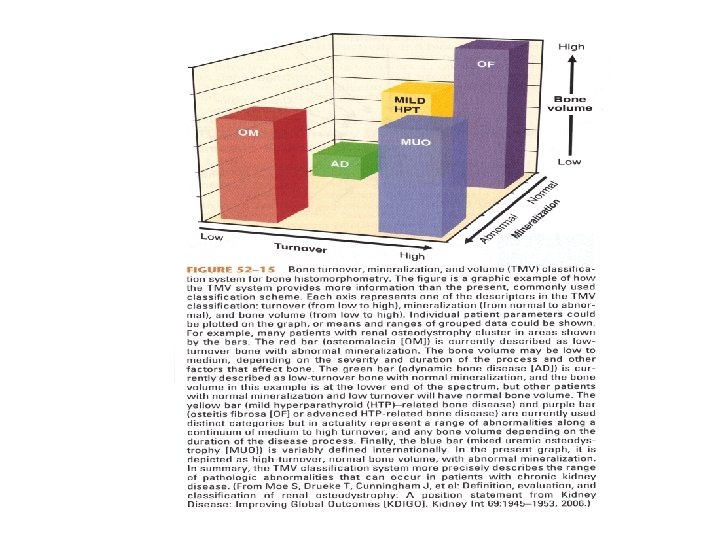
If Secondary HPT is not corrected • Renal Osteodystrophy – Osteitis fibrosa cystica – predominantly hyperparathyroid bone disease – Adynamic bone disease – diminished bone formation and resorption – Osteomalacia – defective mineralization in association with low osteoclast and osteoblast activities – Mixed uremic osteodystrophy – hyperparathyroid bone disease with a superimposed mineralization defect • Metastatic calcification


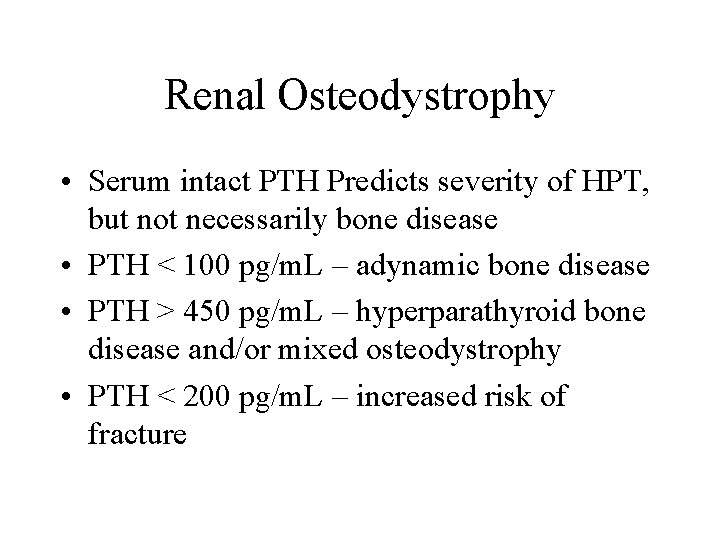
Renal Osteodystrophy • Serum intact PTH Predicts severity of HPT, but not necessarily bone disease • PTH < 100 pg/m. L – adynamic bone disease • PTH > 450 pg/m. L – hyperparathyroid bone disease and/or mixed osteodystrophy • PTH < 200 pg/m. L – increased risk of fracture

Renal Osteodystrophy • Low serum bone-specific alkaline phosphatase (<= 7 ng/m. L) and a low serum PTH suggests a low remodeling disorder • Elevated alkaline phosphatase (>= 20 ng/m. L) alone or with increased serum PTH (>200 pg/m. L) suggests high turnover bone disease.


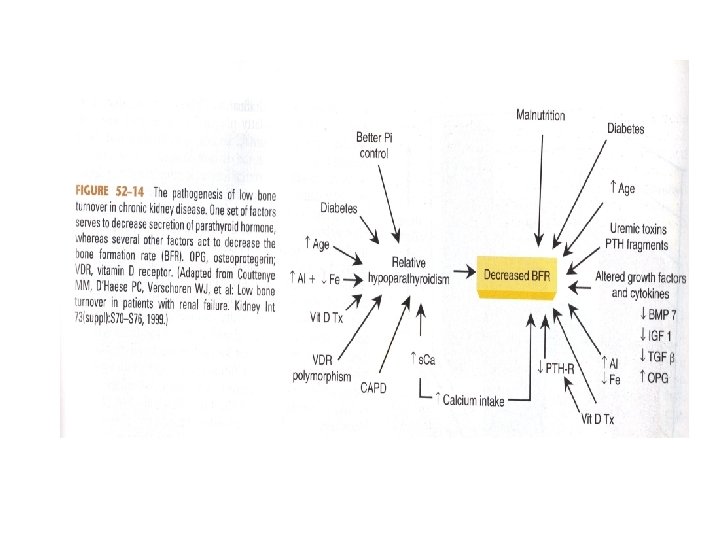
Low Bone Turnover • Most patients are asymptomatic • Increased risk of fracture due to impaired remodeling • Increased risk of vascular calcification due to inability of bone to buffer an acute calcium load


Metabolic Acidosis and Bone Mineral Disease • Stimulates physiochemical mineral dissolution buffering excess hydrogen ions • Leads to a gradual decline in bone calcium stores • Stimulates cell-mediated bone resorption via stimulating osteoclastic activity • Alkali therapy can slow progression of uremic bone disease

New Classification of Bone Disease • Developed to help clarify the interpretation of bone biopsy results • Provide a clinically relevant description of underlying bone pathology • Helps define pathophysiology and guide treatment



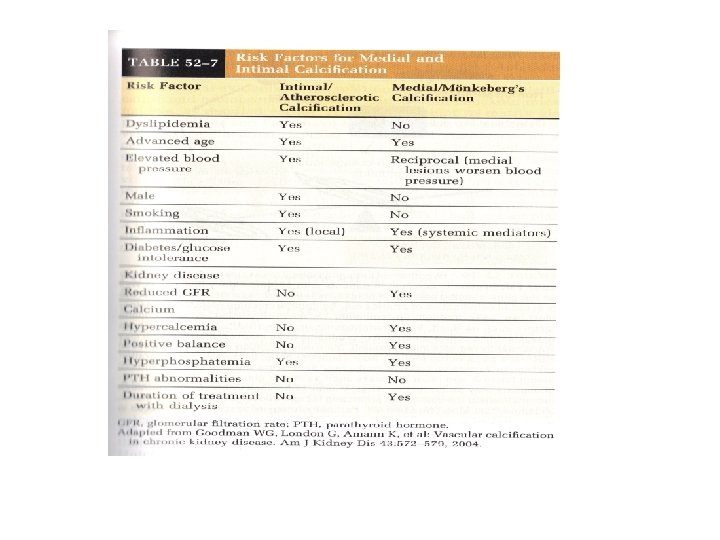
Vascular Calcification • Cardiovascular disease remains the leading cause of morbidity and mortality in CKD • Disorders of Mineral Metabolism – Accelerated atherosclerosis – Arterial calcification – Increased risk of adverse cardiovascular outcomes and death

Extraosseous Calcification • Calcium phosphate precipitation into joints, arteries, soft tissues, and viscera • Calciphylaxis • When the fraction of reabsorbed filtered phosphate declines to 15%, PTH cannot increase phosphate excretion but does continue to release calcium phosphate from bone

Phosphorus and Calcium in CKD • Hyperphosphatemia brings with it a very high population attributable risk of death • Combination of hyperphosphatemia, hypercalcemia, and elevated PTH accounted for 17. 5% of observed, explainable mortality in HD patients


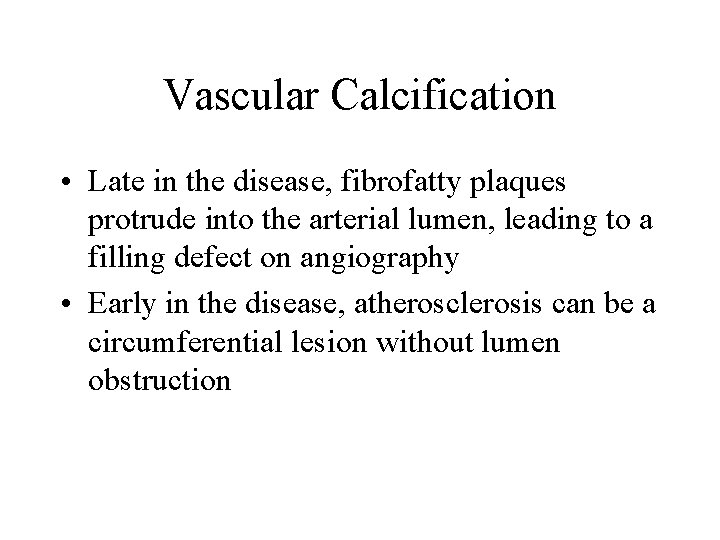
Vascular Calcification • Late in the disease, fibrofatty plaques protrude into the arterial lumen, leading to a filling defect on angiography • Early in the disease, atherosclerosis can be a circumferential lesion without lumen obstruction


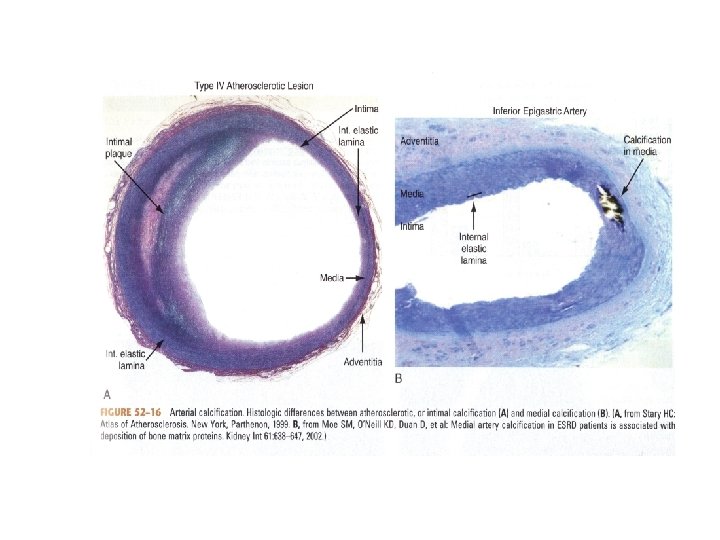
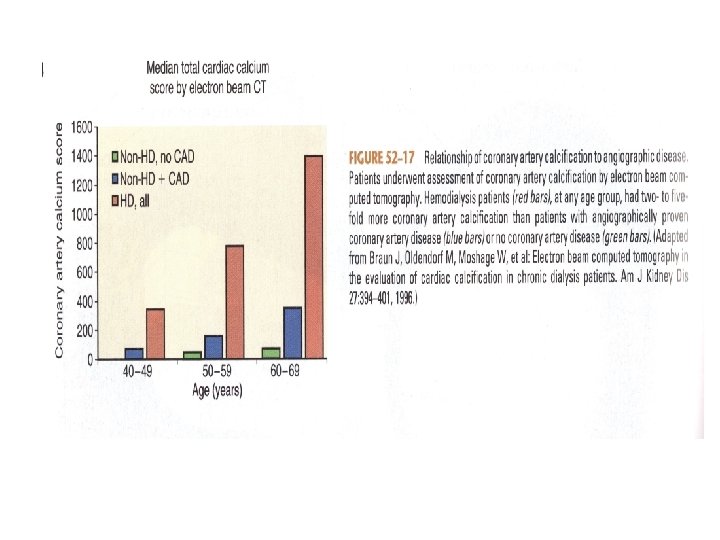
Vascular Calcification • Dialysis Patients have calcification scores that are two-to five fold greater than agematched individuals with normal kidney function and angiographically proven CAD • Dialysis patients have increased arterial calcification (intimal disease and medial layer thickening) in coronary, renal, and iliac arteries.



Post-Renal Transplant Bone Disease • Kidney Transplantation returns patients to CKD and to CKD-MBD. • Disorders of mineral metabolism occur post transplant and include: – Effects of medications – Persistence of underlying disorders – Development of hyperphosphaturia with hypophosphatemia


TREATMENT Secondary Hyperparathyroidism Treatment Options

• Dietary Restriction of Phosphorus • Phosphate Binders (calcium or non-calcium containing) • Vitamin D Analogues • Calcimimetics • Parathyroidectomy

Dietary Phosphate Restriction • 800 – 1, 000 mg per day • Reverses abnormalities of mineral metabolism – Increases plasma calcitriol – Diminishes PTH levels – Improves Ca 2+ intestinal absorption


Phosphate Binders • Limit the absorption of dietary phosphate • Calcium Salts • Non-calcium containing (sevelamer and lanthanum carbonate) • Calcium containing binders should be limited to <1500 mg of elemental calcium per day to keep total calcium intake <2000 mg per day

Phosphate Binders • Vitamin D will increase the intestinal absorption of calcium: calcium containing binders should be reduced accordingly • Patients with low turnover bone disease will deposit excess calcium in extraskeletal sites because their bones cannot take up the calcium.

Vitamin D • • • Ergocalciferol Limit dose of active Vitamin D analogues: Paricalcitol Doxercalciferol Calcitriol Dose limited by hypercalcemia and hyperphosphatemia

VITAMIN D ANALOGUES • Reduce dose of active Vitamin D as PTH levels diminish. • Adjust dose every 4 -8 weeks • Discontinue calcitriol during hypercalcemia • Contraindicated with PTH levels less than 150 pg/ml

Calcimimetics • • • Increase the sensitivity of the Ca. SR Decrease PTH gene expression Increase Vitamin D receptor expression Can reduce plasma PTH by more than 50% Cinacalcet (Sensipar) Limited by hypocalcemia

Treatment Goals in Dialysis Patients • Intact PTH between 150 -300 pg/m. L • Serum Phosphate between 3. 5 -5. 5 mg/d. L • Serum levels of total corrected Calcium between 8. 4 -9. 5 mg/d. L

Treatment Strategy • Reduce Serum Phosphate to normal range • Limit Excessive Calcium Loading • Use Calcimimetic for elevated PTH with Ca>9. 5 • Avoid active Vitamin D analogues and if used, reduce dose as treatment progresses • Prevent progression of parathyroid disease • Maintain bone health and prevent fractures

References • • • Brenner, Barry M. Brenner & Rector’s The Kidney. 8 th Edition. Saunders Elsevier 2008. Pp. 1784 -1809. Rose, Burton D. and Theodore W. Post. Chapter 6 F: Hormonal Regulation of Calcium and Phosphate Balance. Up To Date 2010. Pp. 1 -10. Rose, Burton D. and Theodore W. Post. Chapter 6 G: Calcium and Phosphate Metabolism in Renal Failure. Up To Date 2010. Pp. 1 -8. Qunibi, Wajeh Y. and William L. Henrich. Pathogenesis of Renal Osteodystrophy. Up To Date 2010. Pp. 1 -15. Quarles, Darryl L. Bone Biopsy and the Diagnosis of Renal Osteodystrophy. Up To Date 2010. Pp. 1 -17. Quarles, Darryl L. and Robert E. Cronin. Management of Secondary Hyperparathyroidism and Mineral Metabolism Abnormalities in Dialysis Patients. Up To Date 2010. Pp. 1 -21.