Ca AntiEGFR Mabs Cetuximab Panitumumab AntiAngiogenesis Bevacizumab Aflibercept









Ca ΠΑΧΕΟΣ ΕΝΤΕΡΟΥ – ΟΡΘΟΥ Μεταστατική νόσος • Μονοκλωνικά αντισώματα – Anti-EGFR Mabs: • Cetuximab, Panitumumab – Anti-Angiogenesis: • Bevacizumab, Aflibercept, Ramucirumab
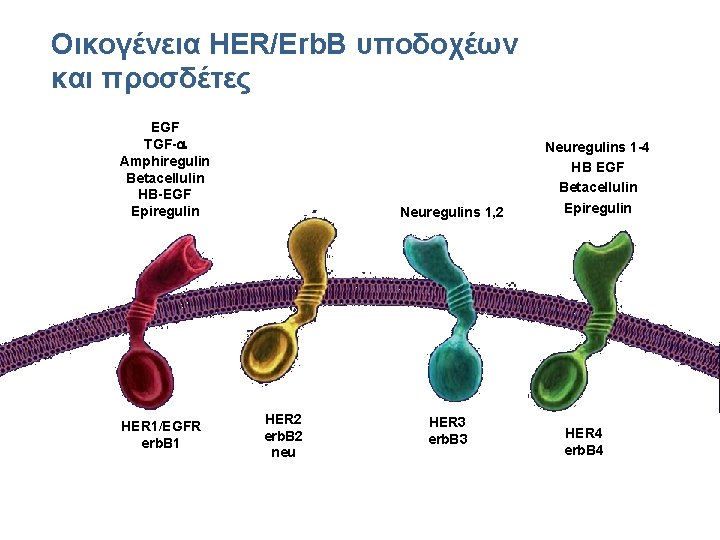
Οικογένεια HER/Erb. B υποδοχέων και προσδέτες Ligands: EGF TGF-a Amphiregulin Betacellulin HB-EGF Epiregulin HER 1/EGFR erb. B 1 Neuregulins 1, 2 HER 2 erb. B 2 neu HER 3 erb. B 3 Neuregulins 1 -4 HB EGF Betacellulin Epiregulin HER 4 erb. B 4
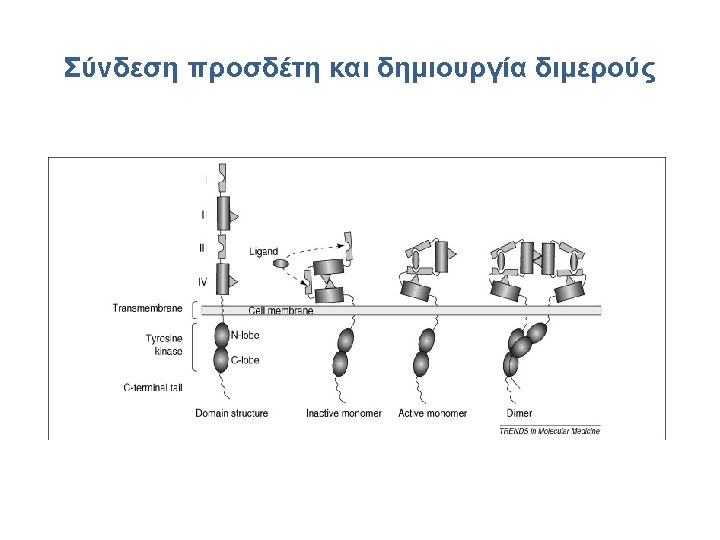
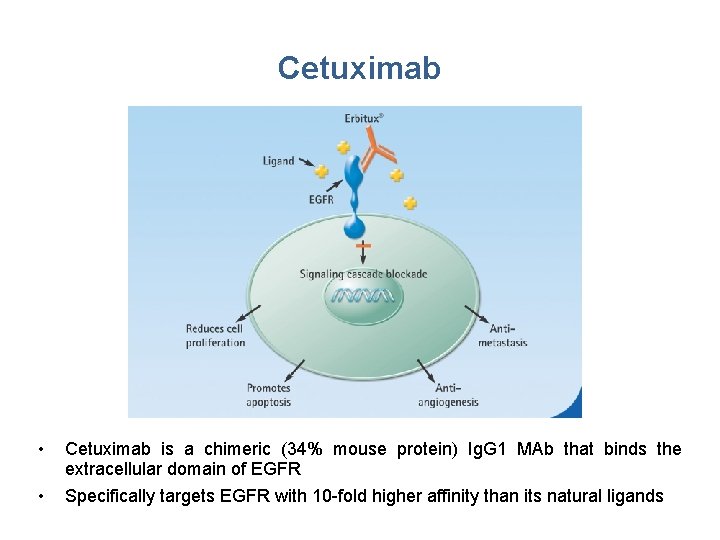
Cetuximab • Cetuximab is a chimeric (34% mouse protein) Ig. G 1 MAb that binds the extracellular domain of EGFR • Specifically targets EGFR with 10 -fold higher affinity than its natural ligands
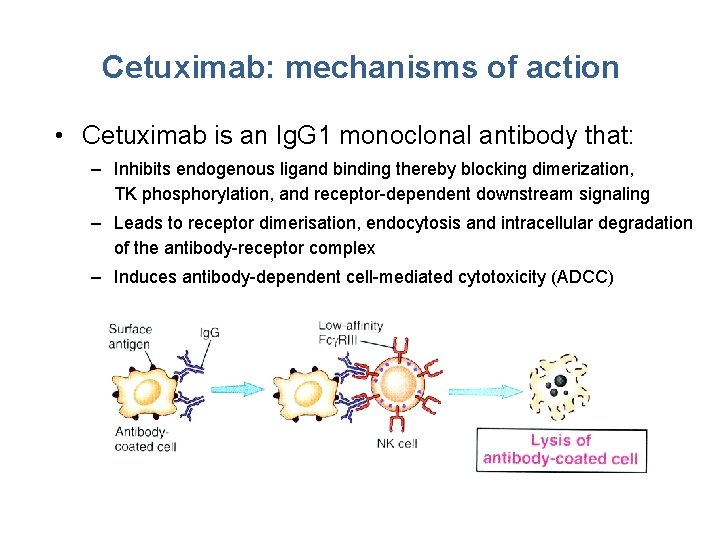
Cetuximab: mechanisms of action • Cetuximab is an Ig. G 1 monoclonal antibody that: – Inhibits endogenous ligand binding thereby blocking dimerization, TK phosphorylation, and receptor-dependent downstream signaling – Leads to receptor dimerisation, endocytosis and intracellular degradation of the antibody-receptor complex – Induces antibody-dependent cell-mediated cytotoxicity (ADCC)
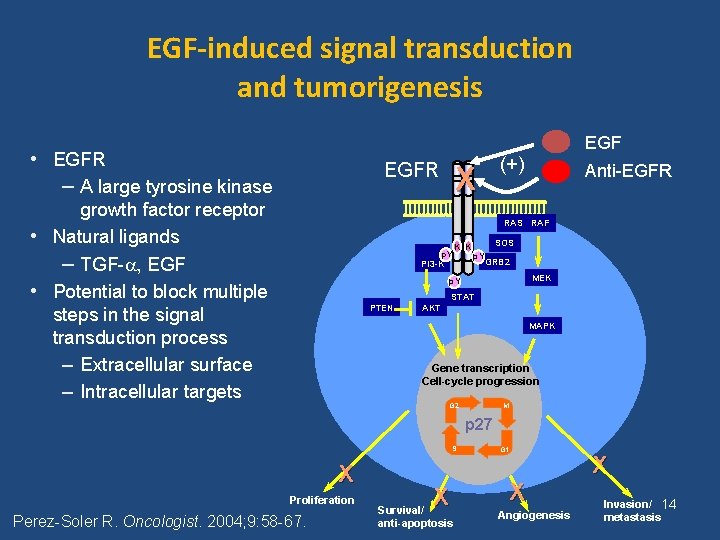
EGF-induced signal transduction and tumorigenesis EGF • EGFR – A large tyrosine kinase EGFR growth factor receptor • Natural ligands – TGF- , EGF • Potential to block multiple steps in the signal transduction process – Extracellular surface – Intracellular targets X (+) Anti-EGFR RAS RAF p. Y K K PI 3 -K SOS p. Y GRB 2 MEK p. Y STAT PTEN AKT MAPK Gene transcription Cell-cycle progression G 2 M p 27 S X Proliferation Perez-Soler R. Oncologist. 2004; 9: 58 -67. X Survival/ anti-apoptosis G 1 X Angiogenesis X Invasion/ 14 metastasis
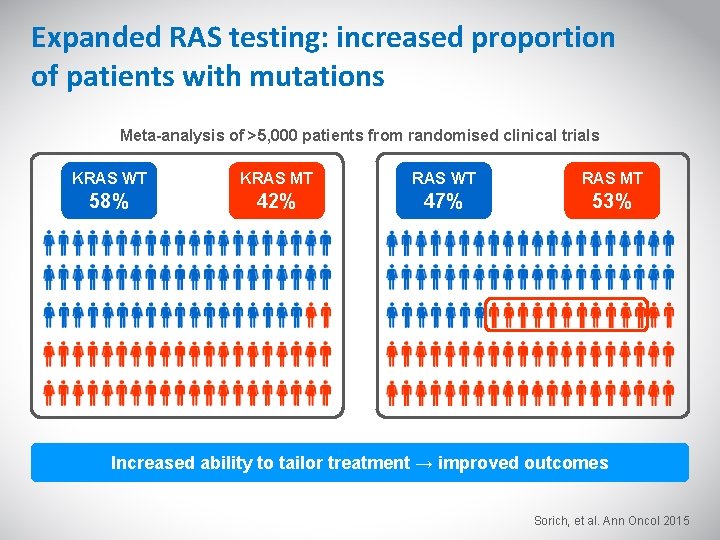
Expanded RAS testing: increased proportion of patients with mutations Meta-analysis of >5, 000 patients from randomised clinical trials KRAS WT KRAS MT RAS WT RAS MT 58% 42% 47% 53% Increased ability to tailor treatment → improved outcomes Sorich, et al. Ann Oncol 2015
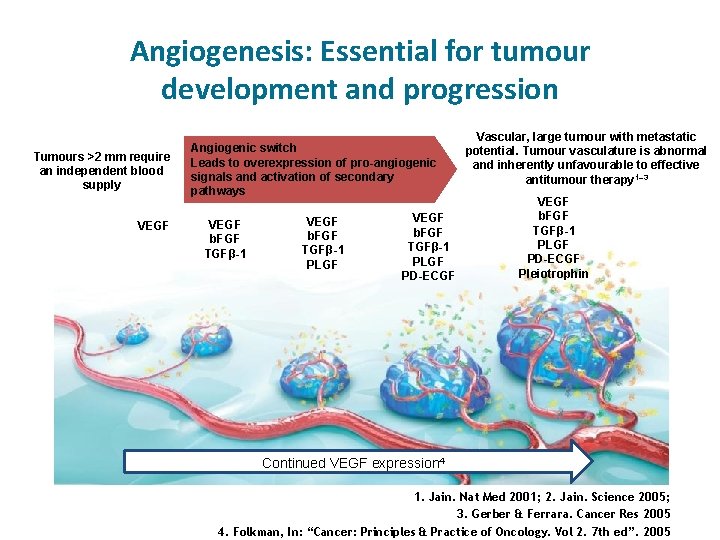
Angiogenesis: Essential for tumour development and progression Tumours >2 mm require an independent blood supply VEGF Angiogenic switch Leads to overexpression of pro-angiogenic signals and activation of secondary pathways VEGF b. FGF TGFβ-1 PLGF PD-ECGF Vascular, large tumour with metastatic potential. Tumour vasculature is abnormal and inherently unfavourable to effective antitumour therapy 1– 3 VEGF b. FGF TGFβ-1 PLGF PD-ECGF Pleiotrophin Continued VEGF expression 4 1. Jain. Nat Med 2001; 2. Jain. Science 2005; 3. Gerber & Ferrara. Cancer Res 2005 4. Folkman, In: “Cancer: Principles & Practice of Oncology. Vol 2. 7 th ed”. 2005

Critical role of VEGF throughout angiogenesis • Activation may occur at various stages of tumour development 1– 4 • Angiogenesis is mediated primarily via the interaction of VEGF-A with VEGFR-2 1– 3 – VEGF binding to capillary endothelial cell receptors generates a downstream signal cascade, promoting angiogenesis Angiogenesis correlates with disease progression in many tumours 1– 4 VEGF receptor Facilitates survival of existing endothelial cells Contributes to vascular abnormalities 1, 2, 6, 7, 9 Stimulates new vessel growth Increases vessel permeability 1. Ferrara. Endocr Rev 2004; 2. Hicklin & Ellis. JCO 2005 3. Ferrara Nat Rev Drug Discov 2004; 4. Bergers & Benjamin. Nat Rev Cancer 2003
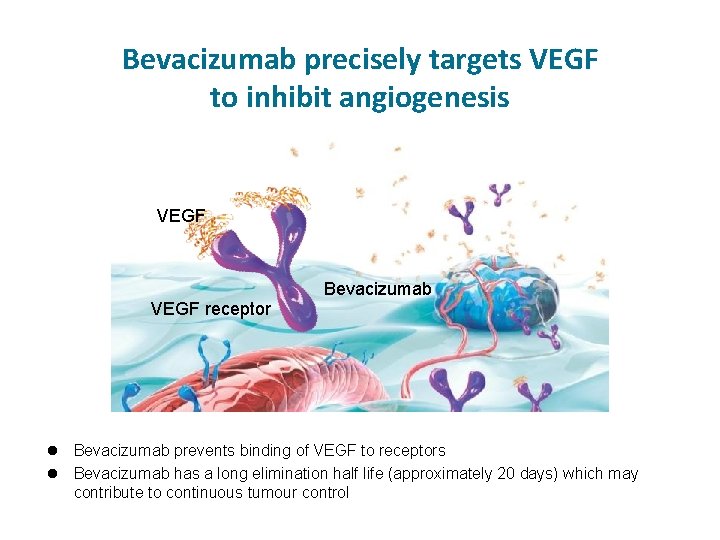
Bevacizumab precisely targets VEGF to inhibit angiogenesis VEGF receptor Bevacizumab prevents binding of VEGF to receptors l Bevacizumab has a long elimination half life (approximately 20 days) which may contribute to continuous tumour control l
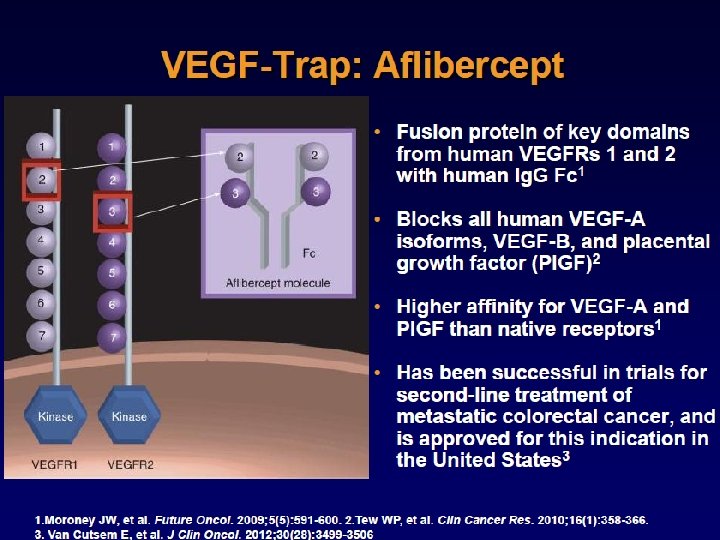
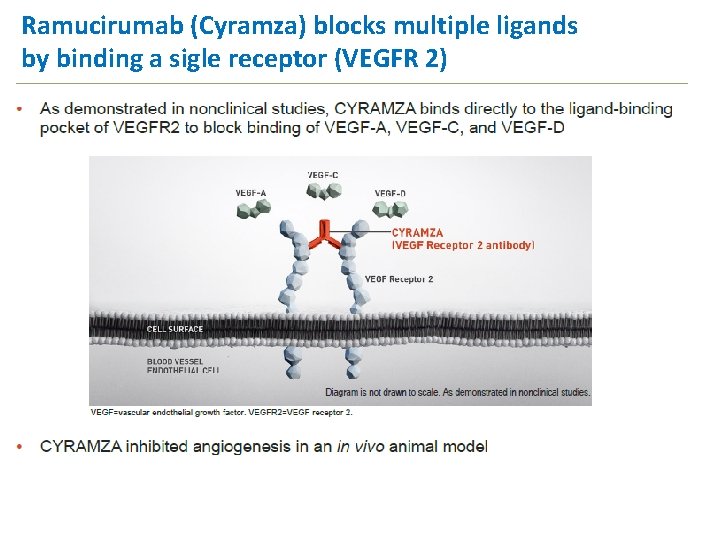
Ramucirumab (Cyramza) blocks multiple ligands by binding a sigle receptor (VEGFR 2) 20
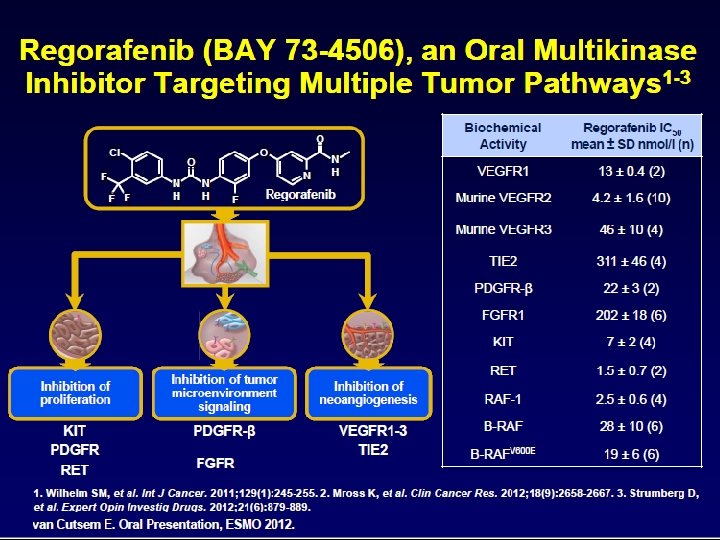
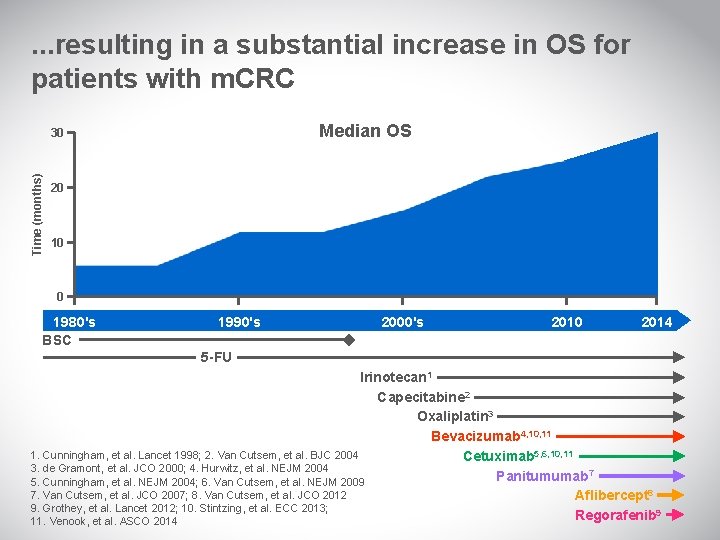
. . . resulting in a substantial increase in OS for patients with m. CRC Median OS Time (months) 30 20 10 0 1980's BSC 1990's 2000's 2010 2014 5 -FU Irinotecan 1 Capecitabine 2 Oxaliplatin 3 Bevacizumab 4, 10, 11 1. Cunningham, et al. Lancet 1998; 2. Van Cutsem, et al. BJC 2004 Cetuximab 5, 6, 10, 11 3. de Gramont, et al. JCO 2000; 4. Hurwitz, et al. NEJM 2004 Panitumumab 7 5. Cunningham, et al. NEJM 2004; 6. Van Cutsem, et al. NEJM 2009 7. Van Cutsem, et al. JCO 2007; 8. Van Cutsem, et al. JCO 2012 Aflibercept 8 9. Grothey, et al. Lancet 2012; 10. Stintzing, et al. ECC 2013; Regorafenib 9 11. Venook, et al. ASCO 2014






Ca ΣΤΟΜΑΧΟΥ Μεταστατική νόσος • Ανακουφιστική χημειοθεραπεία – Cisplatin, 5 -Fluorouracil, Anthracyclines – Irinotecan, Oxaliplatin, Docetaxel, Paclitaxel, Capecitabine – Προσθήκη Trastuzumab σε HER 2 (+) νόσο – Ramucirumab






- Slides: 34