c2003 BrooksCole a division of Thomson Learning Inc
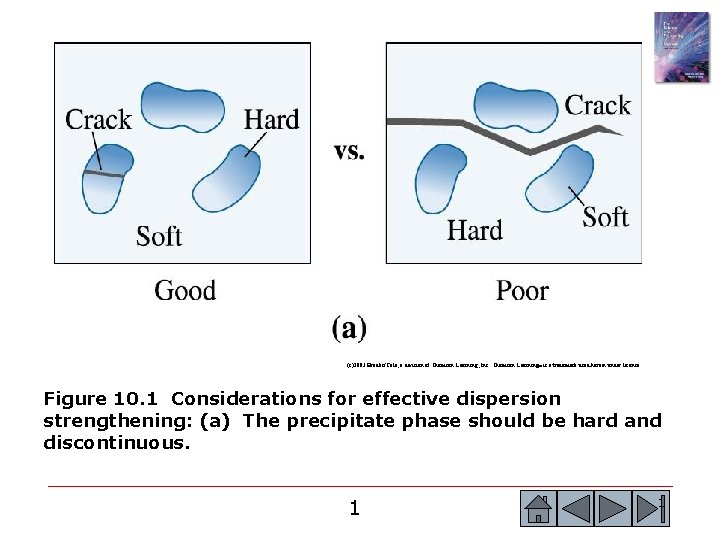
(c)2003 Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning™ is a trademark used herein under license. Figure 10. 1 Considerations for effective dispersion strengthening: (a) The precipitate phase should be hard and discontinuous. 1 1
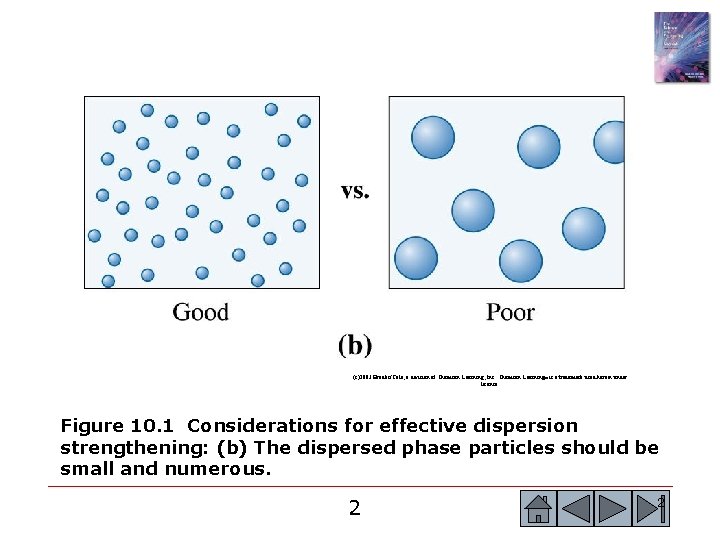
(c)2003 Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning™ is a trademark used herein under license. Figure 10. 1 Considerations for effective dispersion strengthening: (b) The dispersed phase particles should be small and numerous. 2 2
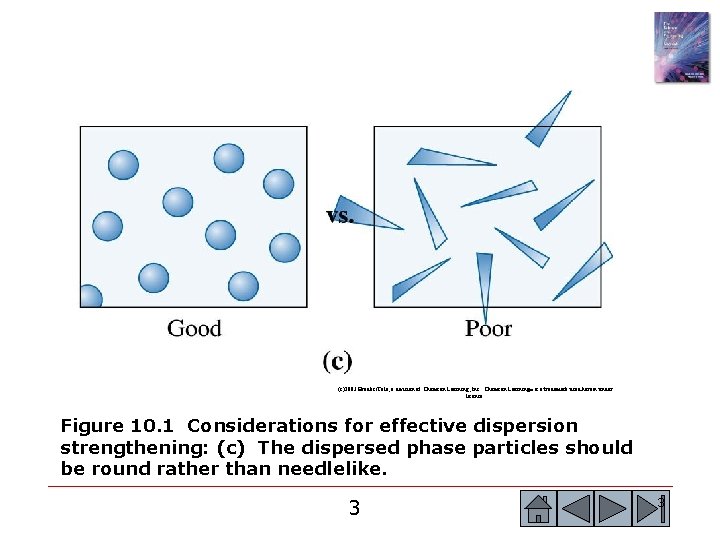
(c)2003 Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning™ is a trademark used herein under license. Figure 10. 1 Considerations for effective dispersion strengthening: (c) The dispersed phase particles should be round rather than needlelike. 3 3
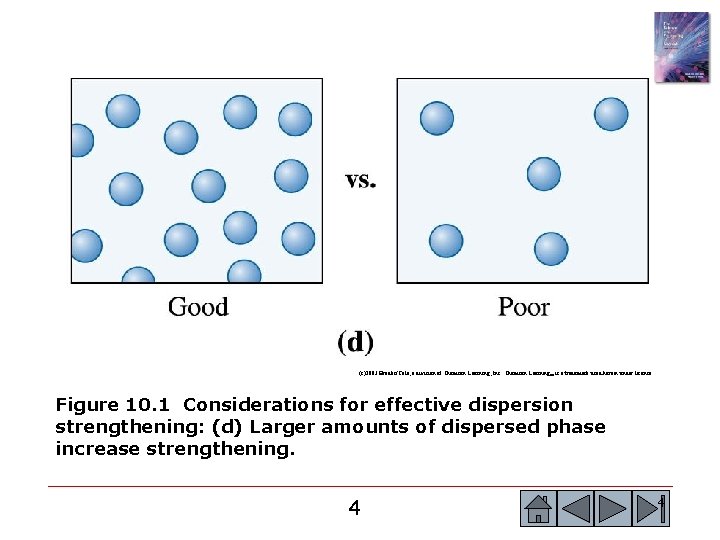
(c)2003 Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning™ is a trademark used herein under license. Figure 10. 1 Considerations for effective dispersion strengthening: (d) Larger amounts of dispersed phase increase strengthening. 4 4
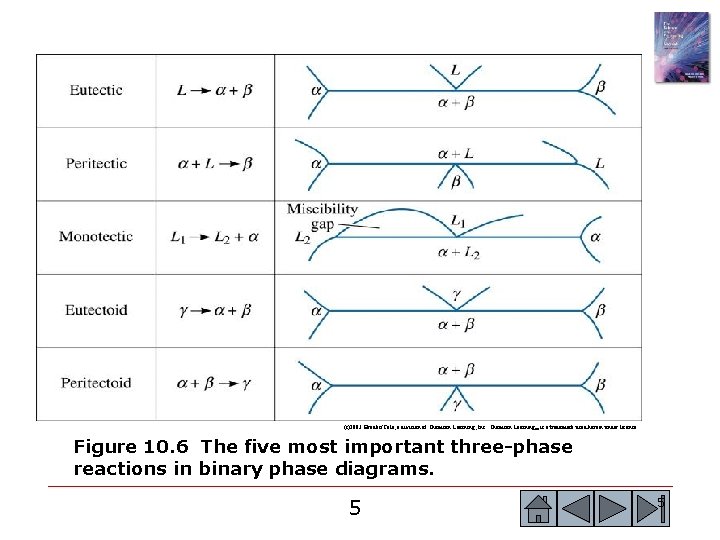
(c)2003 Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning™ is a trademark used herein under license. Figure 10. 6 The five most important three-phase reactions in binary phase diagrams. 5 5

Section 10. 4 The Eutectic Phase Diagram o Solvus - A solubility curve that separates a single-solid phase region from a two-solid phase region in the phase diagram. o Hypoeutectic alloy - An alloy composition between that of the left-hand-side end of the tie line defining the eutectic reaction and the eutectic composition. o Hypereutectic alloys - An alloy composition between that of the right-hand-side end of the tie line defining the eutectic reaction and the eutectic composition. 6 6
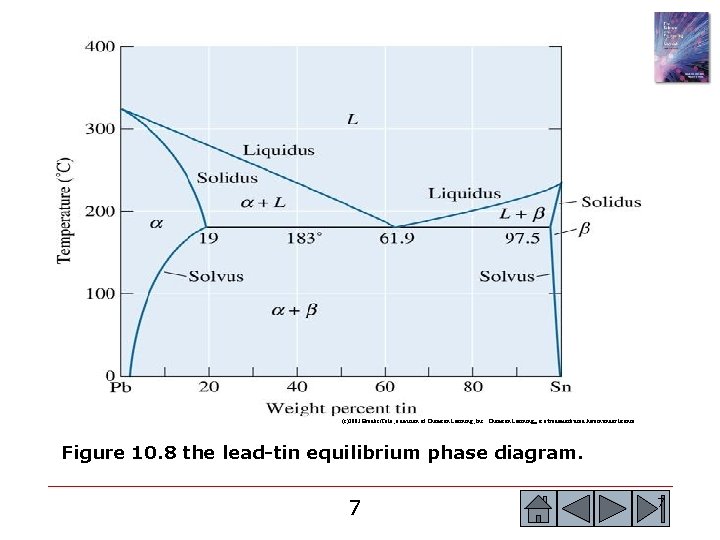
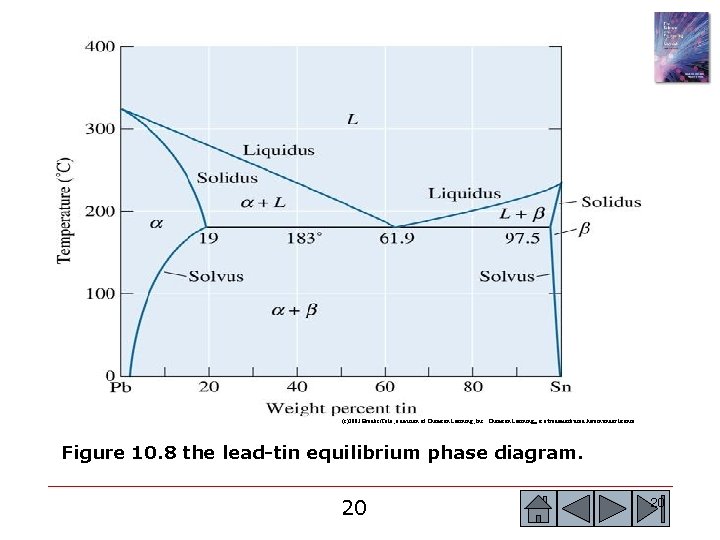
(c)2003 Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning™ is a trademark used herein under license. Figure 10. 8 the lead-tin equilibrium phase diagram. 7 7
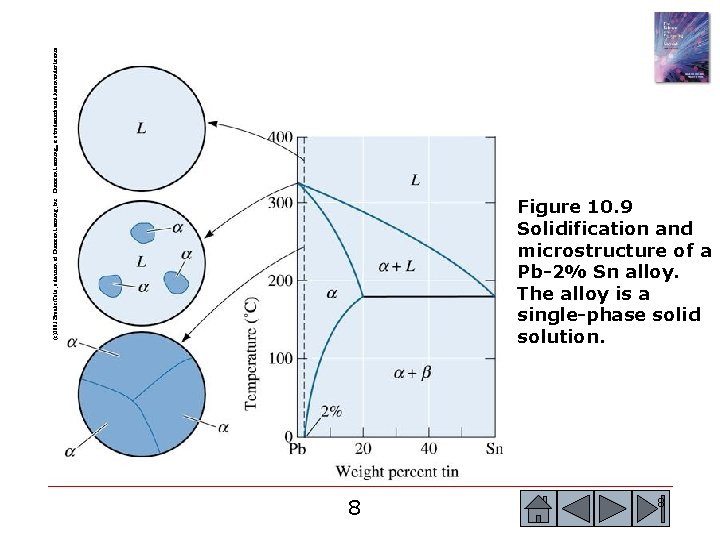
(c)2003 Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning™ is a trademark used herein under license. Figure 10. 9 Solidification and microstructure of a Pb-2% Sn alloy. The alloy is a single-phase solid solution. 8 8
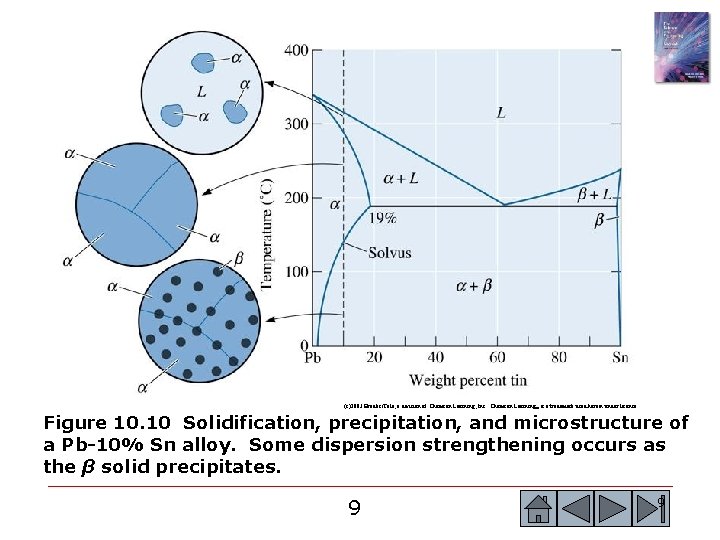
(c)2003 Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning™ is a trademark used herein under license. Figure 10. 10 Solidification, precipitation, and microstructure of a Pb-10% Sn alloy. Some dispersion strengthening occurs as the β solid precipitates. 9 9
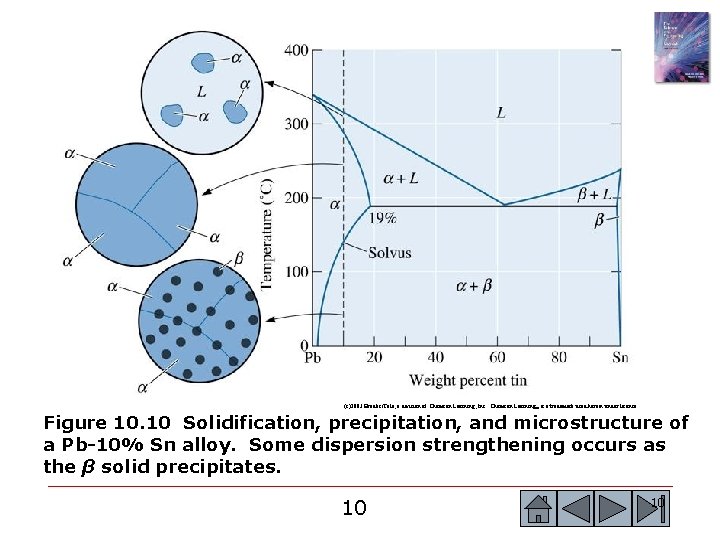
(c)2003 Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning™ is a trademark used herein under license. Figure 10. 10 Solidification, precipitation, and microstructure of a Pb-10% Sn alloy. Some dispersion strengthening occurs as the β solid precipitates. 10 10
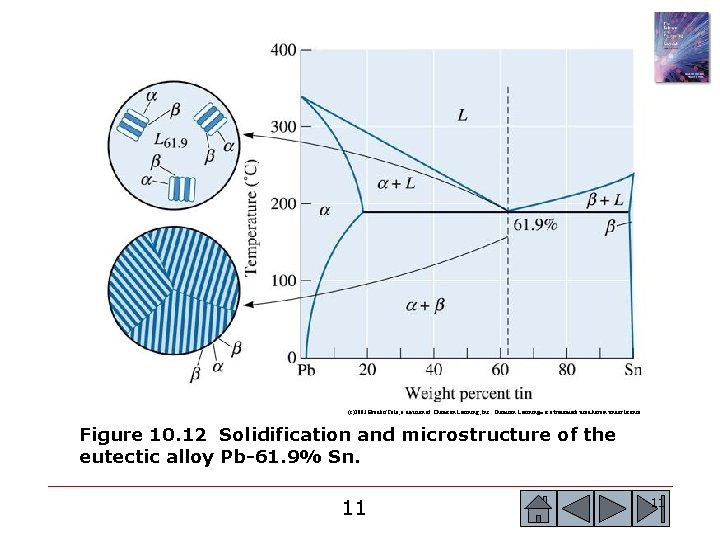
(c)2003 Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning™ is a trademark used herein under license. Figure 10. 12 Solidification and microstructure of the eutectic alloy Pb-61. 9% Sn. 11 11
12 12
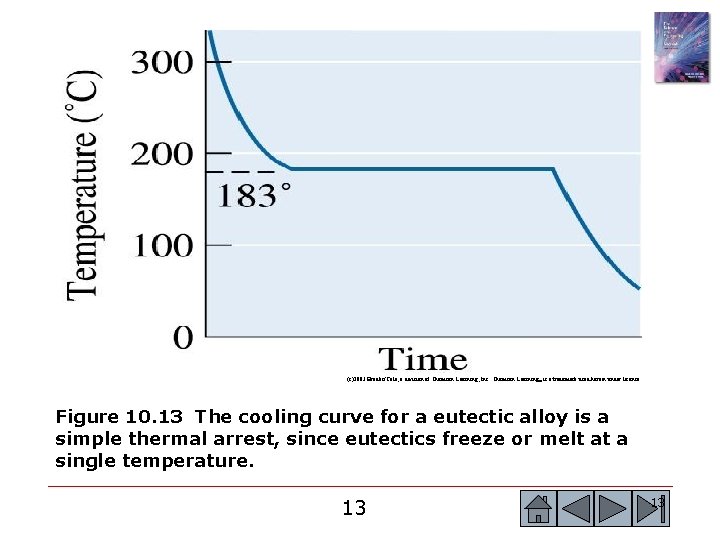
(c)2003 Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning™ is a trademark used herein under license. Figure 10. 13 The cooling curve for a eutectic alloy is a simple thermal arrest, since eutectics freeze or melt at a single temperature. 13 13
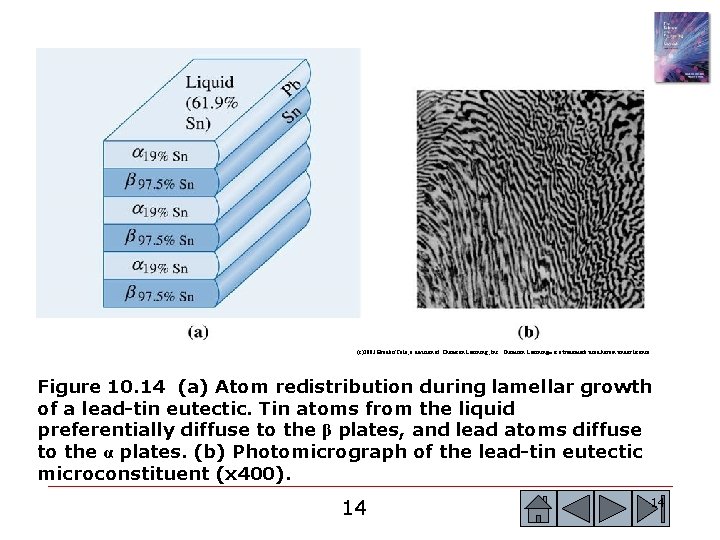
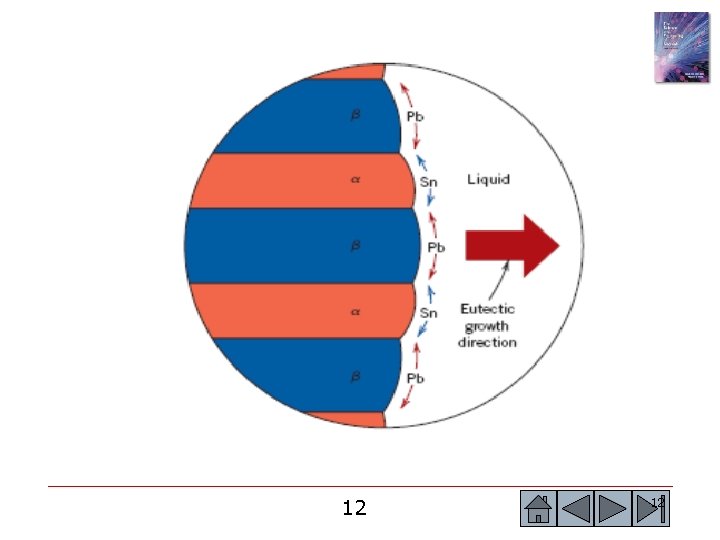
(c)2003 Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning™ is a trademark used herein under license. Figure 10. 14 (a) Atom redistribution during lamellar growth of a lead-tin eutectic. Tin atoms from the liquid preferentially diffuse to the β plates, and lead atoms diffuse to the α plates. (b) Photomicrograph of the lead-tin eutectic microconstituent (x 400). 14 14
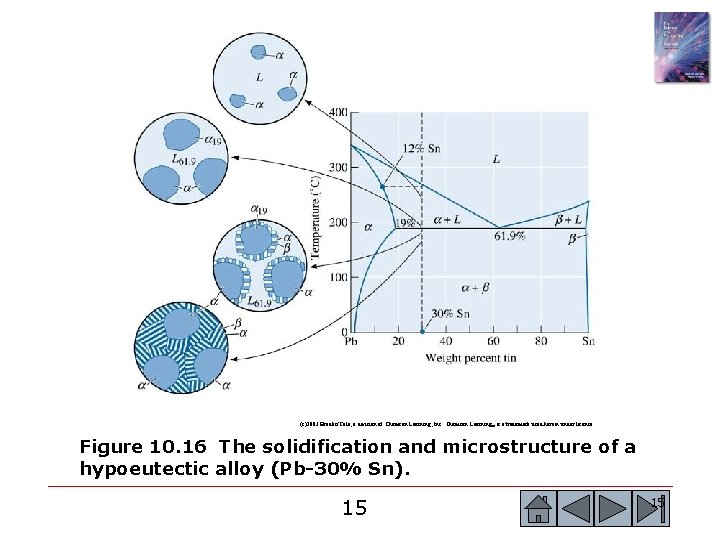
(c)2003 Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning™ is a trademark used herein under license. Figure 10. 16 The solidification and microstructure of a hypoeutectic alloy (Pb-30% Sn). 15 15

Example 10. 5 Determination of Phases and Amounts in a Pb-30% Sn Hypoeutectic Alloy For a Pb-30% Sn alloy, determine the phases present, their amounts, and their compositions at 300 o. C, 200 o. C, 184 o. C, 182 o. C, and 0 o. C. 16 16

Example 10. 5 SOLUTION 17 17
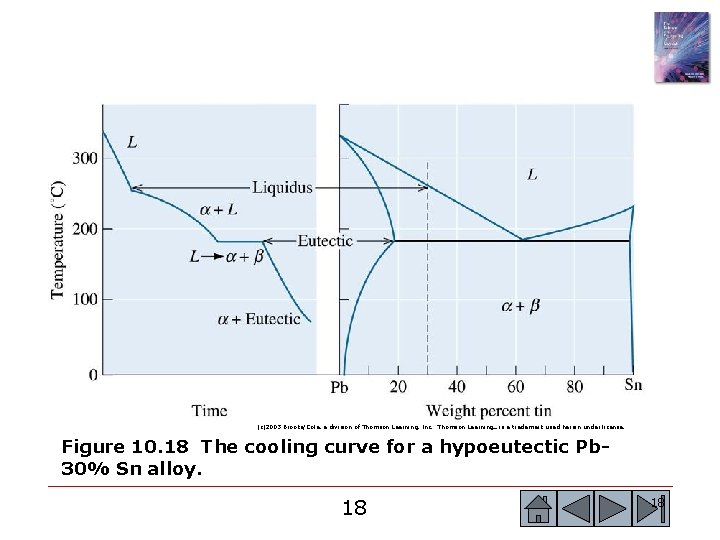
(c)2003 Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning™ is a trademark used herein under license. Figure 10. 18 The cooling curve for a hypoeutectic Pb 30% Sn alloy. 18 18
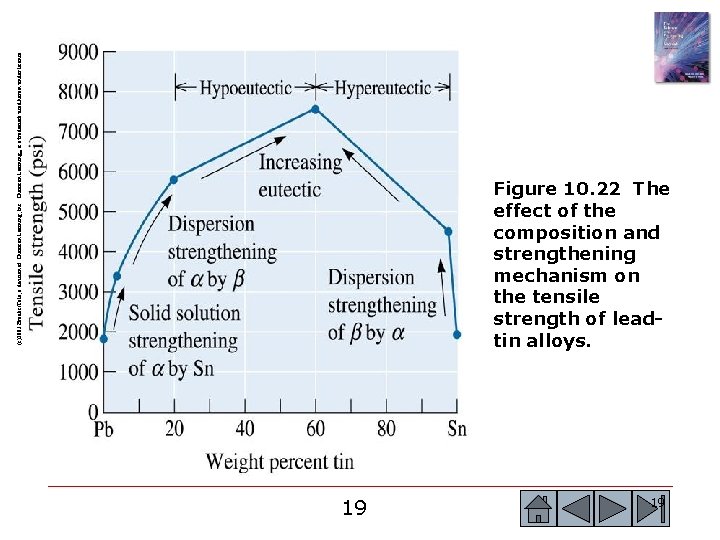
(c)2003 Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning™ is a trademark used herein under license. Figure 10. 22 The effect of the composition and strengthening mechanism on the tensile strength of leadtin alloys. 19 19
(c)2003 Brooks/Cole, a division of Thomson Learning, Inc. Thomson Learning™ is a trademark used herein under license. Figure 10. 8 the lead-tin equilibrium phase diagram. 20 20
- Slides: 20