C I 3 6 Optical Isomerism Types of







- Slides: 14

C. I. 3. 6 Optical Isomerism

Types of isomerism Isomerism Structural isomerism Stereoisomerism Geometric isomerism Optical isomerism

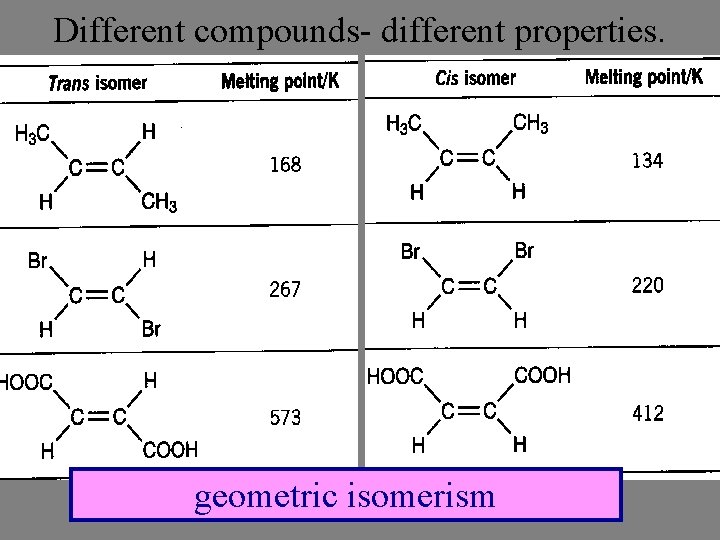
Different compounds- different properties. geometric isomerism

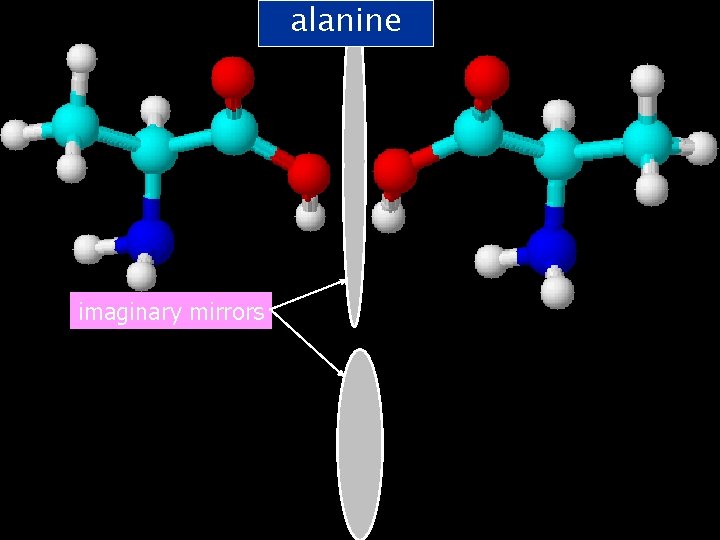
Optical Isomerism Arises because of the different ways you can arrange four different groups around a carbon atom. Hang on!! Aren’t -amino acids a group of compounds that have four different groups around a carbon atom?

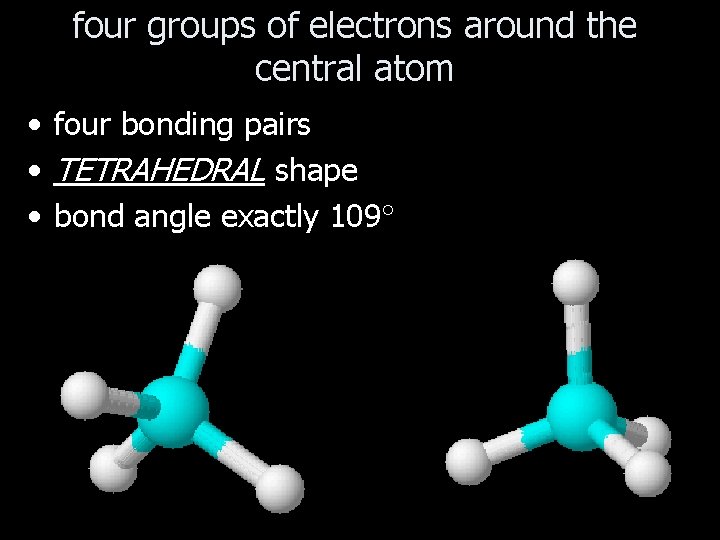
four groups of electrons around the central atom • four bonding pairs • TETRAHEDRAL shape • bond angle exactly 109

alanine imaginary mirrors

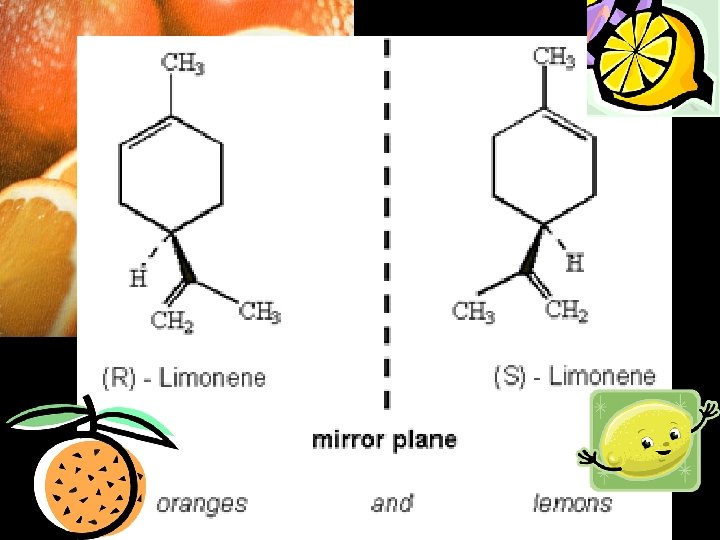
All molecules have mirror images – however they don’t all exist as two isomers. What makes an isomer is the fact that the mirror image and the original molecule are non-superimposable! The only way to make these two superimpose is to break & reform bonds.

Left and right hands are an example of non-superimposable mirror images.

Enantiomers ? üMolecules such as alanine that exist in these two forms = optical isomers or enantiomers. ü We distinguish between the two enantiomers of a molecule by +/-, D/L or more correctly R/S. ü A 50/50 mixture of the two enantiomers is called a racemic mixture or a racemate.

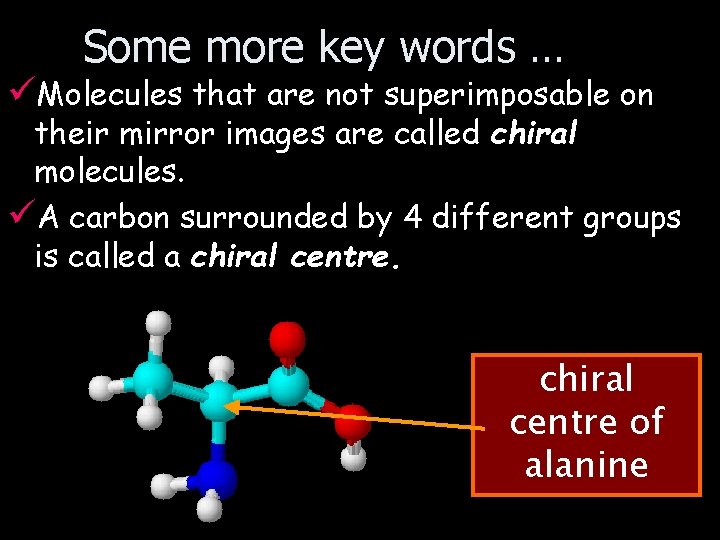
Some more key words … üMolecules that are not superimposable on their mirror images are called chiral molecules. üA carbon surrounded by 4 different groups is called a chiral centre of alanine

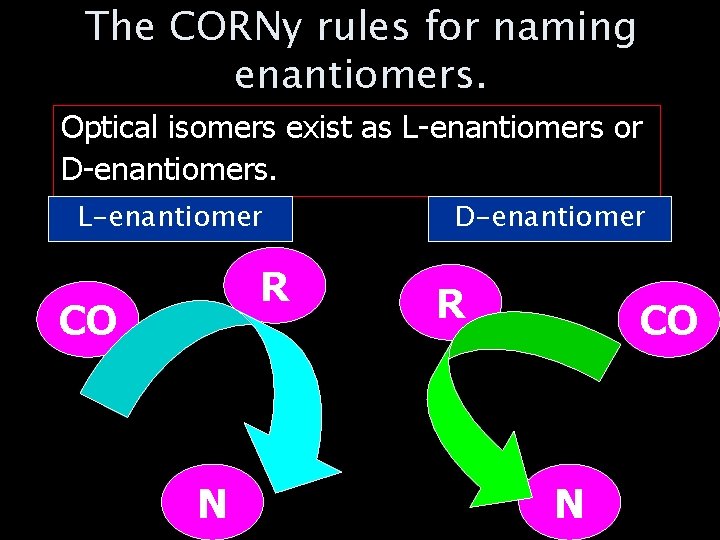
The CORNy rules for naming enantiomers. Optical isomers exist as L-enantiomers or D-enantiomers. L-enantiomer R CO N D-enantiomer R CO N

The CORNy rules for naming enantiomers. 1. Imagine looking down on the molecule with the single H atom point straight up towards you. 2. Label the other three groups a. COOH = CO b. R-group = R c. NH 2 = N 3. If CORN is arranged in a CLOCKWISE direction it is the L-amino acid. 4. If CORN is arranged ANTI-CLOCKWISE it is a D-amino acid.

How do enantiomers differ? • Behave identically in ordinary test-tube chemical reactions. • Have same physical properties. BUT!! • Behave differently in presence of other chiral molecules. – e. g. Taste-buds are ‘chiral’ D-amino acids taste sweet, L-amino acids are tasteless or bitter.
