C 2 Bonding Structure the Properties of Matter

C 2 Bonding, Structure & the Properties of Matter
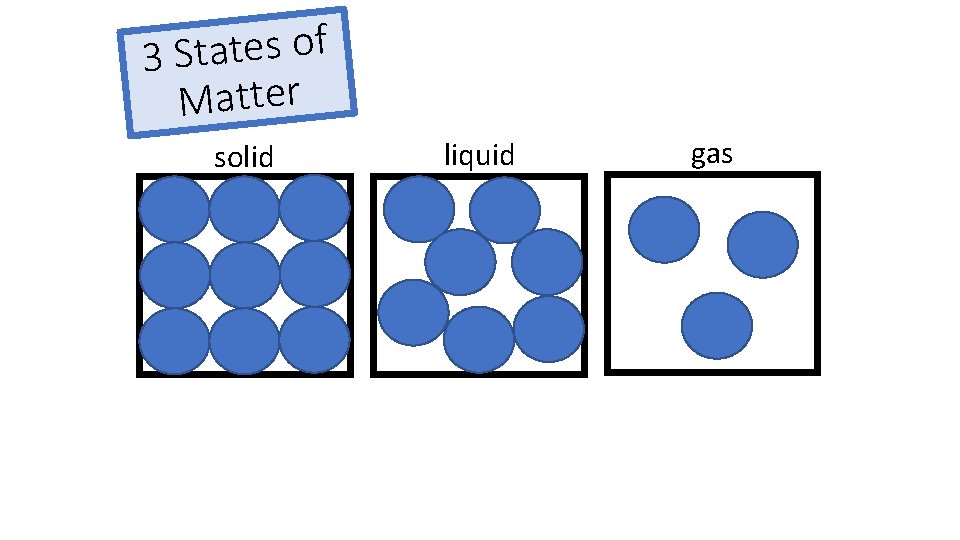
f o s e t a t 3 S Matter solid liquid gas
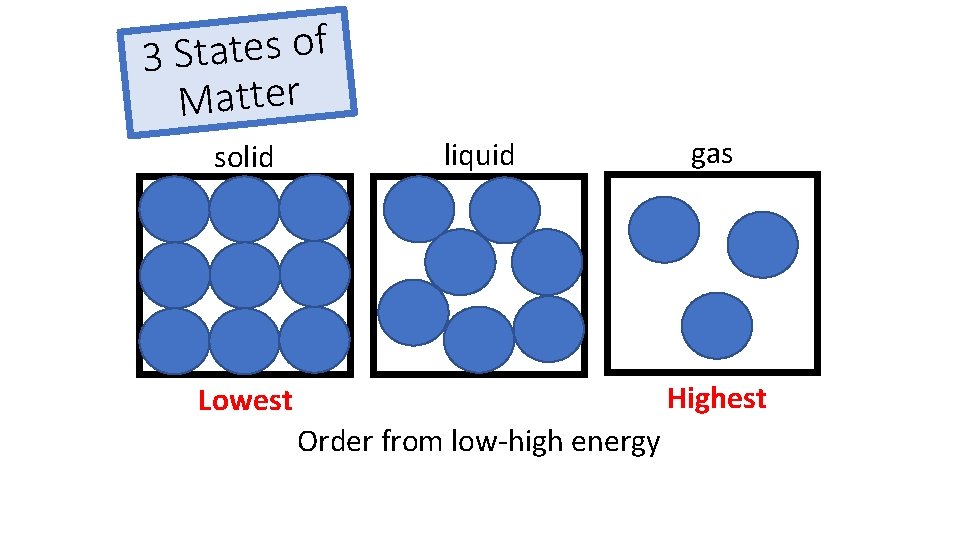
f o s e t a t 3 S Matter solid liquid gas Highest Lowest Order from low-high energy
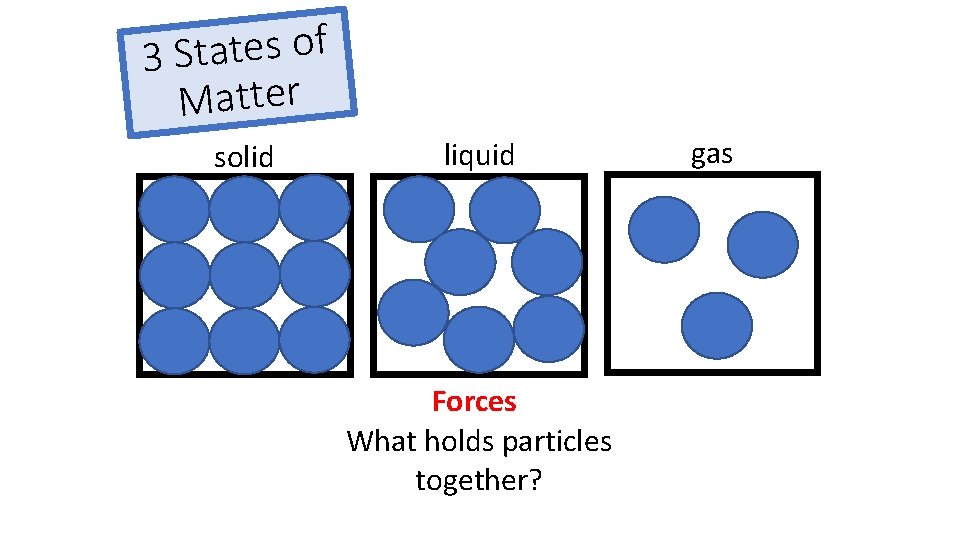
f o s e t a t 3 S Matter solid liquid Forces What holds particles together? gas
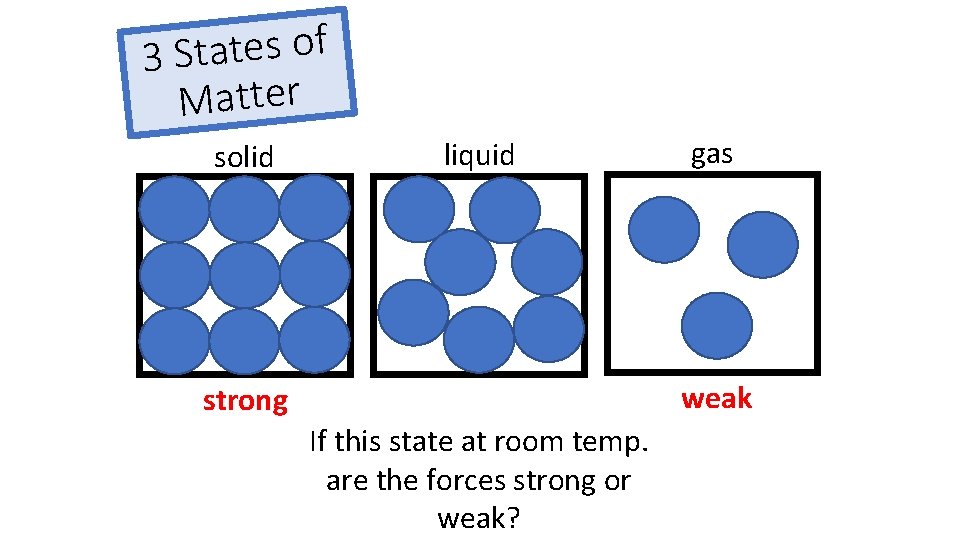
f o s e t a t 3 S Matter solid liquid gas weak strong If this state at room temp. are the forces strong or weak?

State Symbols (s) liquid (l) aqueous (g) solid (aq) gas

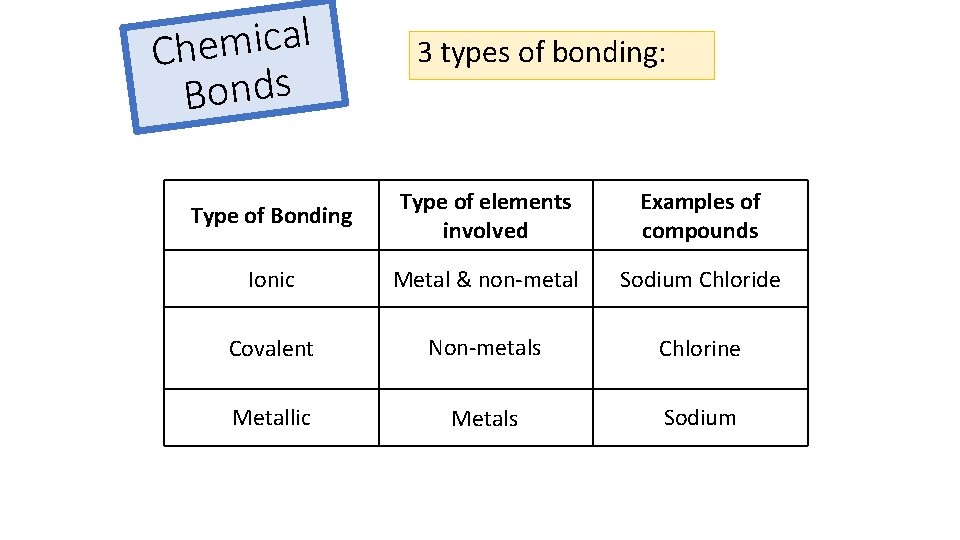
l a c i m e Ch Bonds 3 types of bonding: Type of Bonding Type of elements involved Examples of compounds Ionic Metal & non-metal Sodium Chloride Covalent Non-metals Chlorine Metallic Metals Sodium
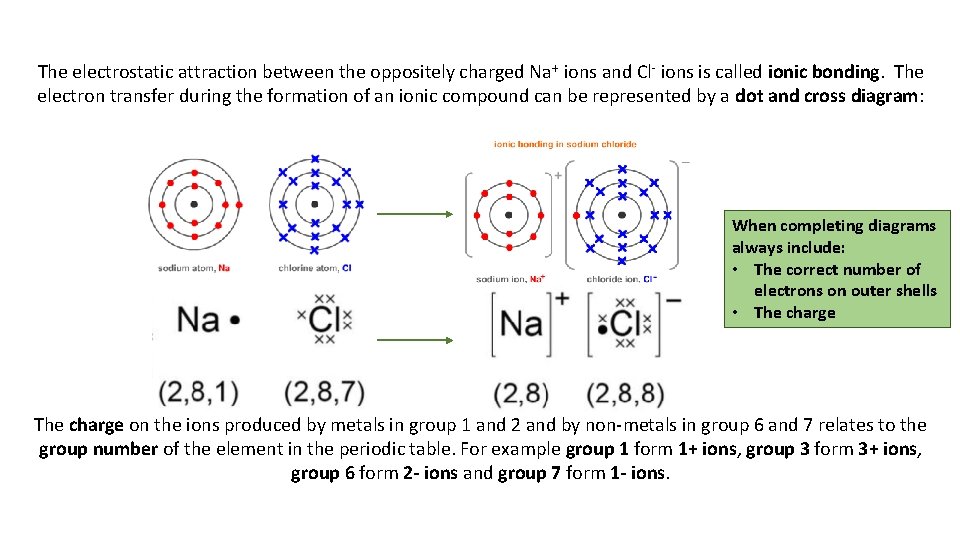
The electrostatic attraction between the oppositely charged Na + ions and Cl- ions is called ionic bonding. The electron transfer during the formation of an ionic compound can be represented by a dot and cross diagram: When completing diagrams always include: • The correct number of electrons on outer shells • The charge on the ions produced by metals in group 1 and 2 and by non-metals in group 6 and 7 relates to the group number of the element in the periodic table. For example group 1 form 1+ ions, group 3 form 3+ ions, group 6 form 2 - ions and group 7 form 1 - ions.
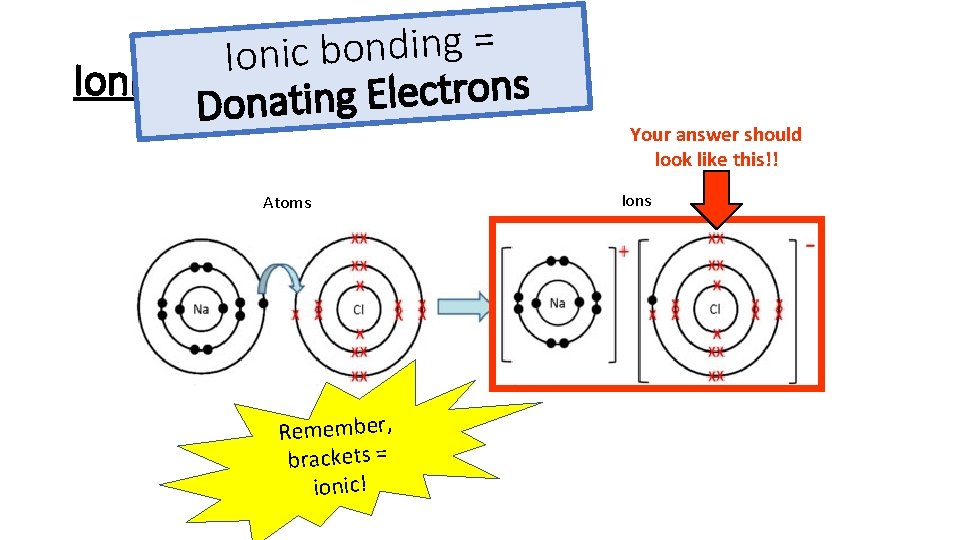
= g n i d n o b c i Ionic Bonding s n o r t c e l E g Donatin Atoms Remember, brackets = ionic! Your answer should look like this!! Ions
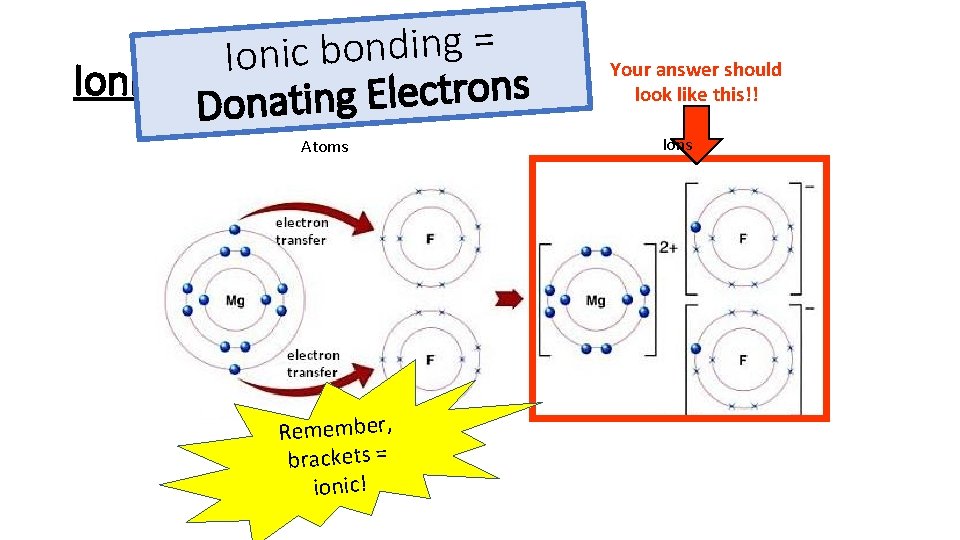
= g n i d n o b c i Ionic Bonding s n o r t c e l E g Donatin Atoms Remember, brackets = ionic! Your answer should look like this!! Ions

g n i d n o b c i Ionic. DBonding escribing ion 1. Which has lost e-? How many? 2. Positive of negative ion formed? 3. Which has gained e-? How many? 4. Positive or negative ion formed? 5. Do you need any other atoms? 6. Held together by electrostatic forces
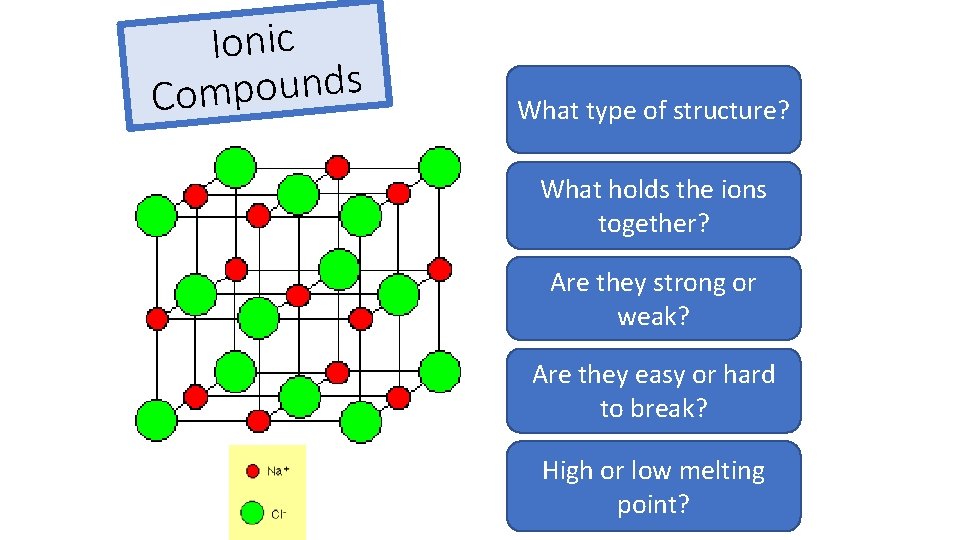
Ionic s d n u o p m Co What Giant type ionic of structure? lattice What holds the ions Electrostatic forces together? Are they strong or Strong weak? Are they easy or hard Hard to break? High or low melting High point?

Ionic s d n u o p m Co Conduct electricity No when solid? • Ions cannot move Why? • Cannot carry charge Molten Whenordoes aqueous it? • Ions free to move Why? • Can carry charge
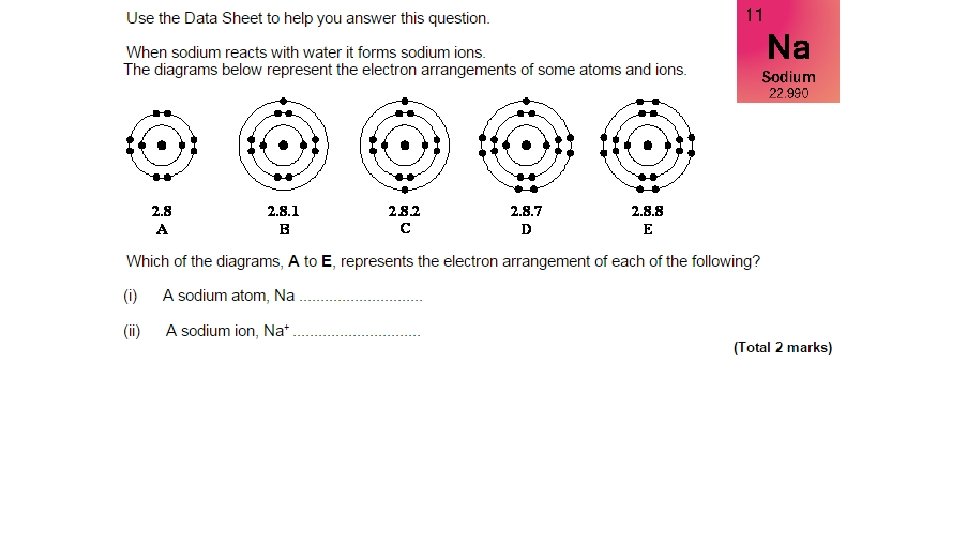
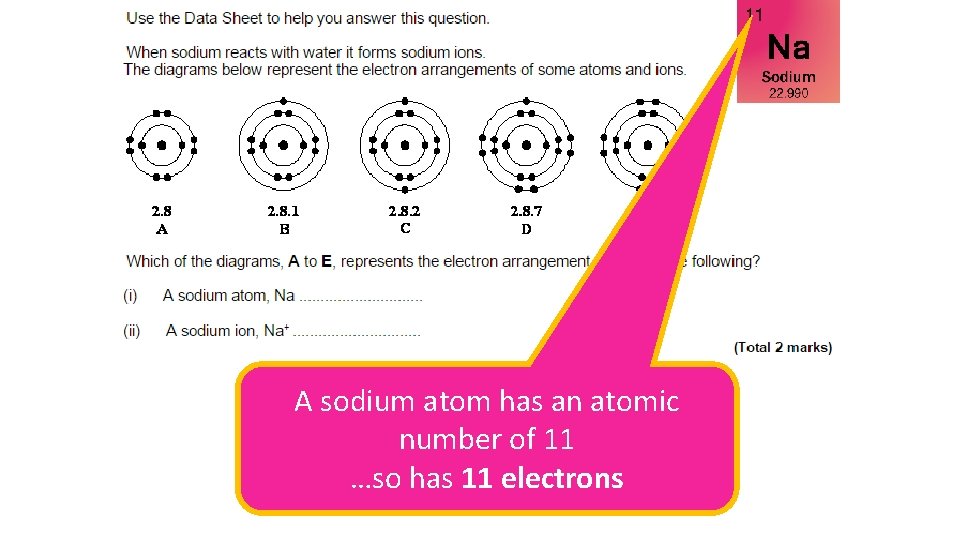
A sodium atom has an atomic number of 11 …so has 11 electrons
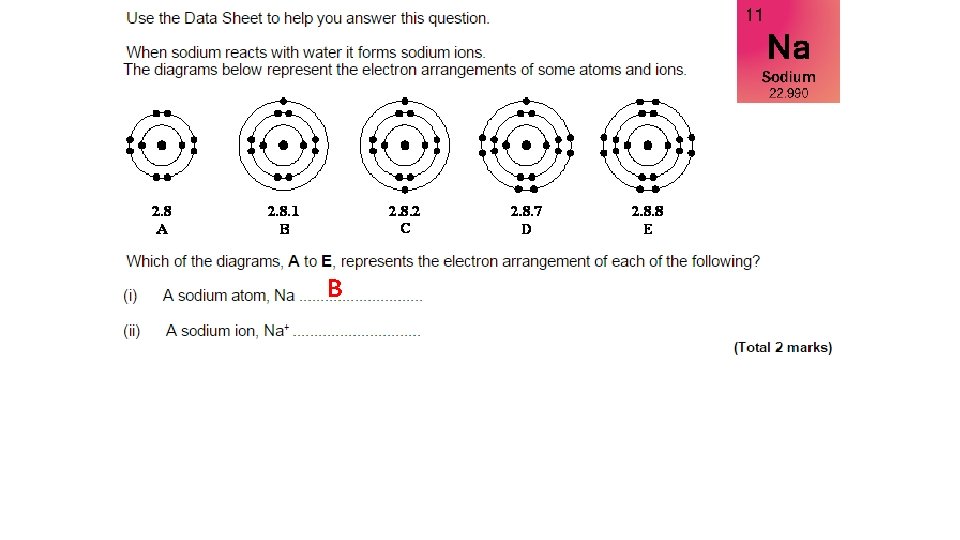
B
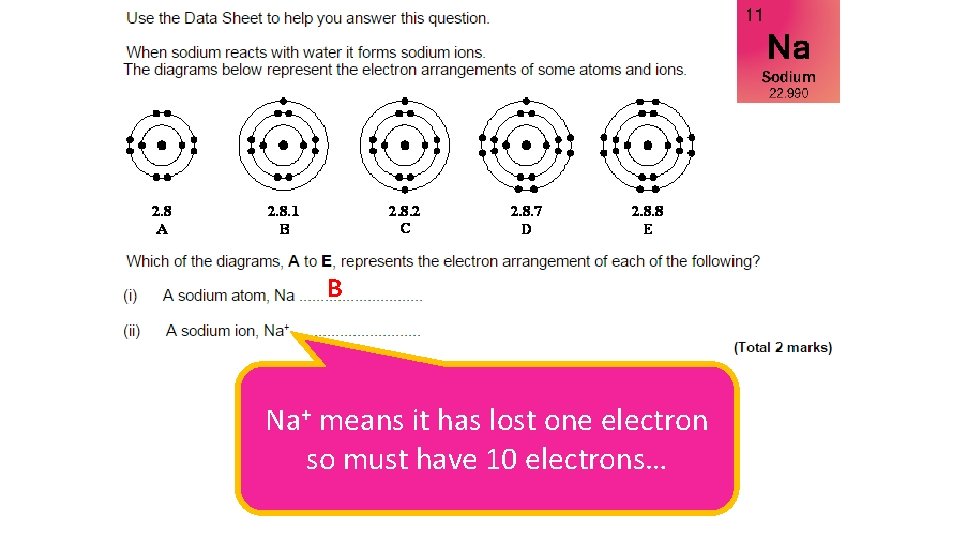
B Na+ means it has lost one electron so must have 10 electrons…
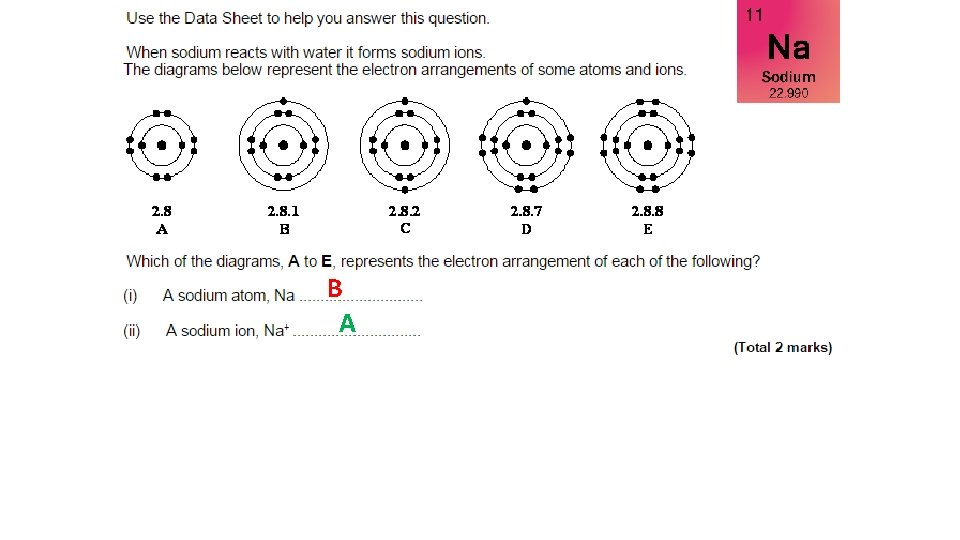
B A

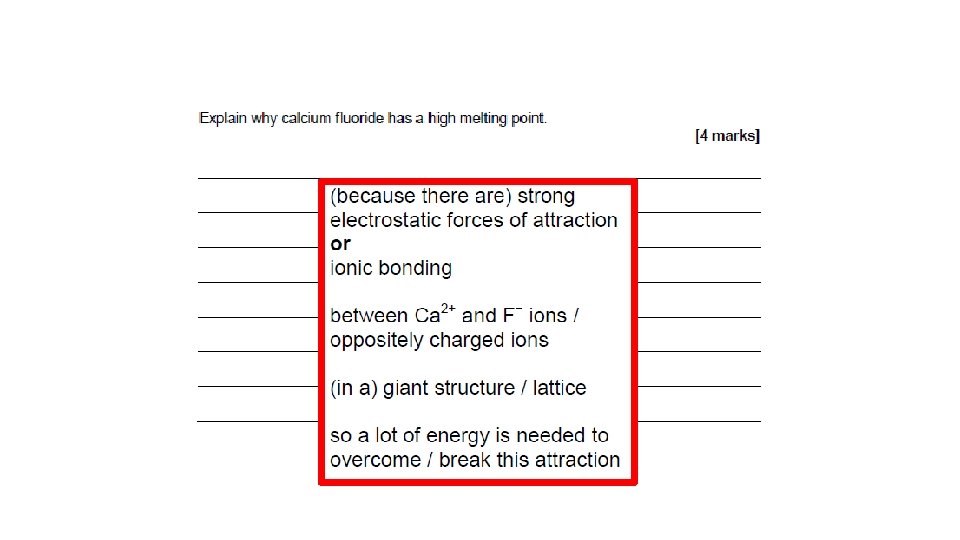
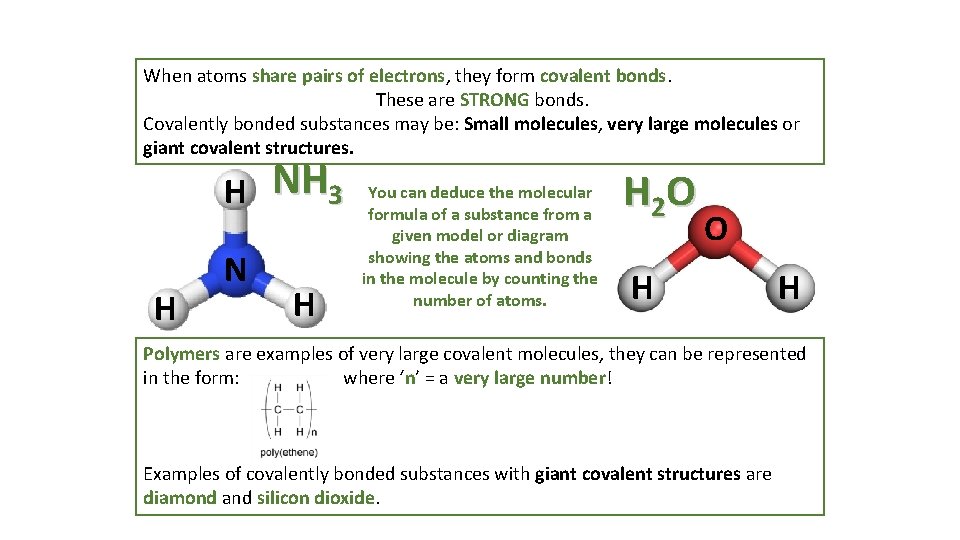
When atoms share pairs of electrons, they form covalent bonds. These are STRONG bonds. Covalently bonded substances may be: Small molecules, very large molecules or giant covalent structures. H H N NH 3 H You can deduce the molecular formula of a substance from a given model or diagram showing the atoms and bonds in the molecule by counting the number of atoms. H 2 O O H H Polymers are examples of very large covalent molecules, they can be represented in the form: where ‘n’ = a very large number! Examples of covalently bonded substances with giant covalent structures are diamond and silicon dioxide.
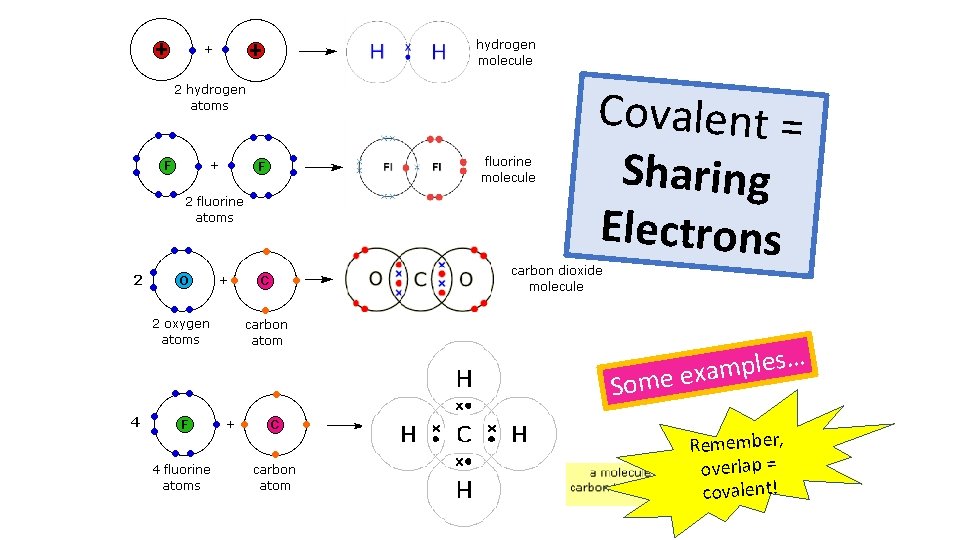
Covalent = Sharing Electrons … s e l p m a Some ex Remember, overlap = covalent!
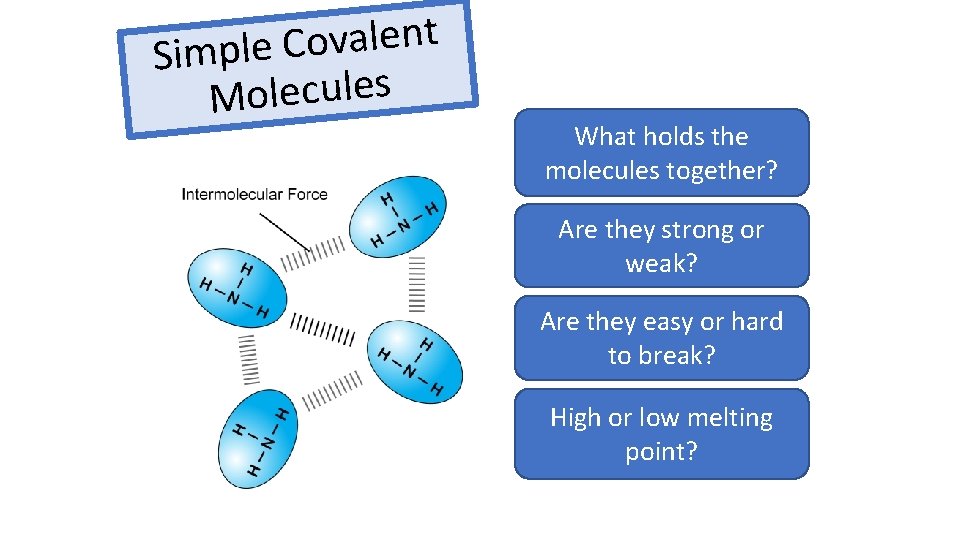
t n e l a v o C Simple s e l u c e l o M What holds the Intermolecular forces molecules together? Are they strong or Weak weak? Are they easy or hard Easy to break? High or low melting Low point?
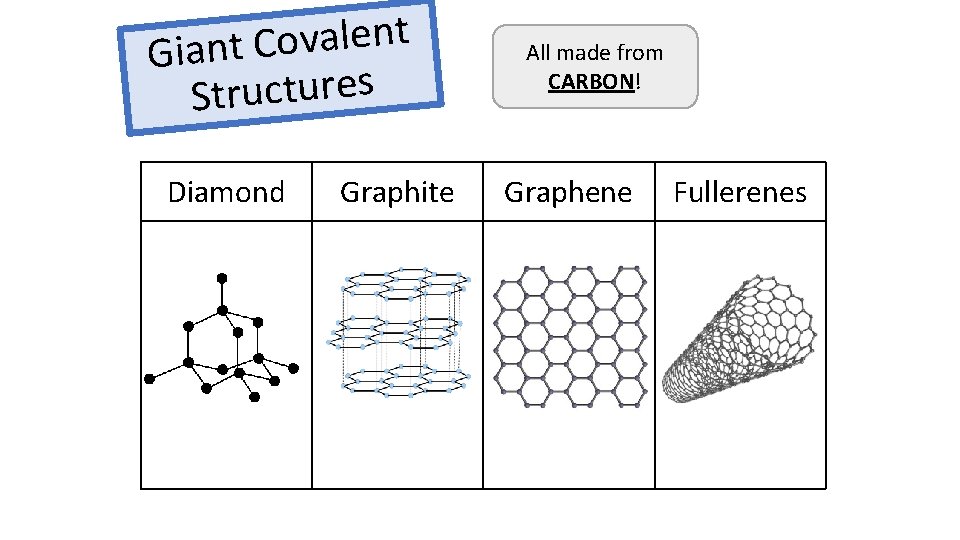
t n e l a v o C t Gian s e r u t c u r St Diamond Graphite All made from CARBON! Graphene Fullerenes
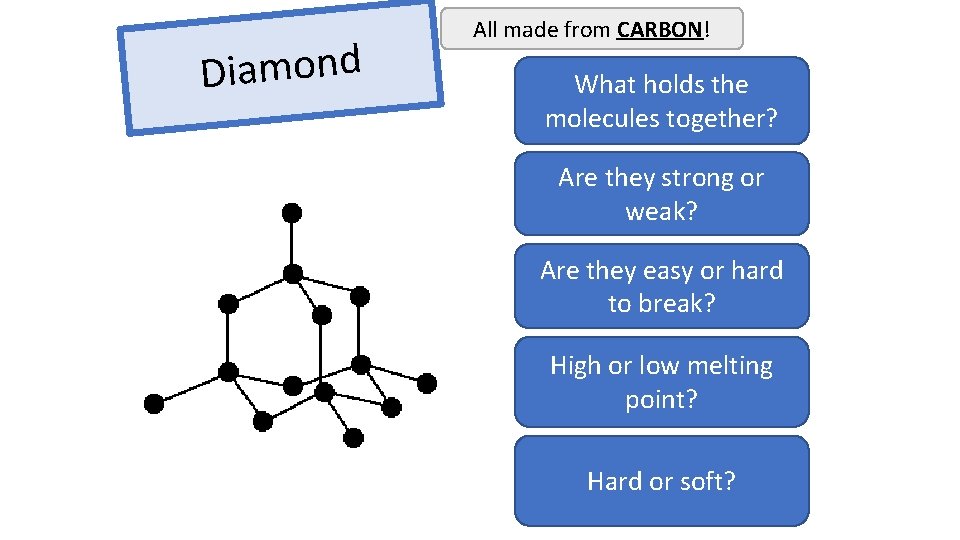
Diamond All made from CARBON! What holds the Covalent bonds molecules together? Are they strong or Very stong weak? Are they easy or hard Hard to break? High or low melting Very high point? Hard or soft?
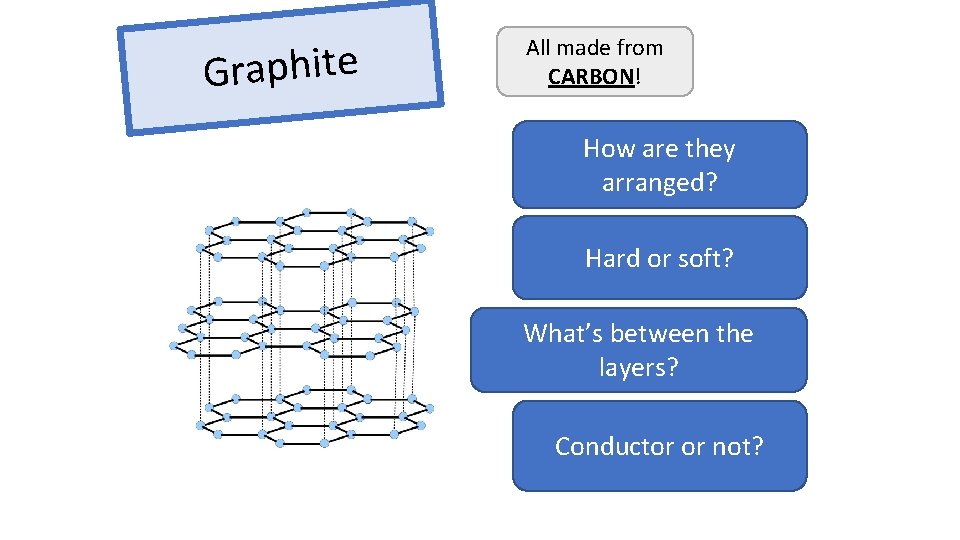
Graphite All made from CARBON! How are they Layers, can slide arranged? Hard. Soft or soft? Delocalised What’s between electrons, thecan carrylayers? the charge Conductor electricity or not?
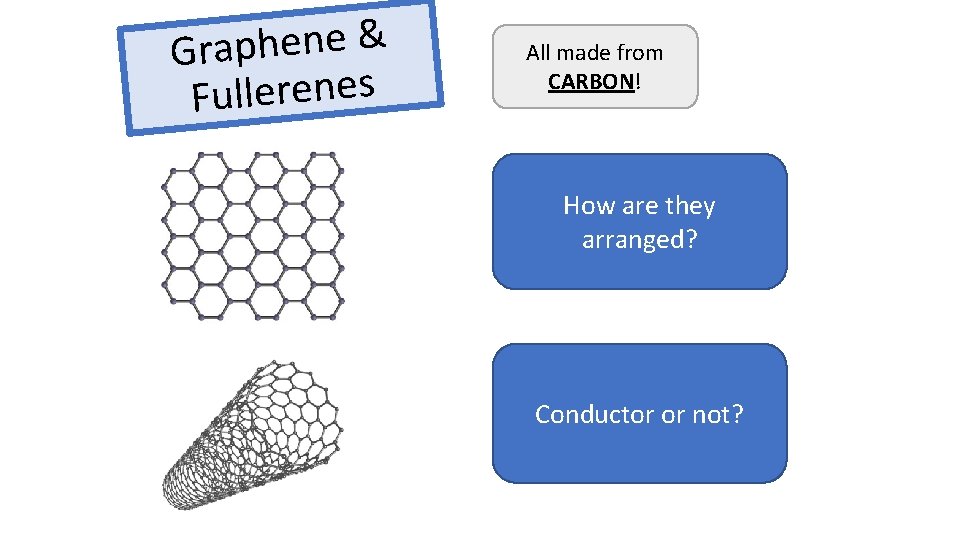
& e n e h p a r G s e n e r e l l u F All made from CARBON! How are they 1 atom thick arranged? Free electrons, carry Conductor or not? charge, conduct electricity
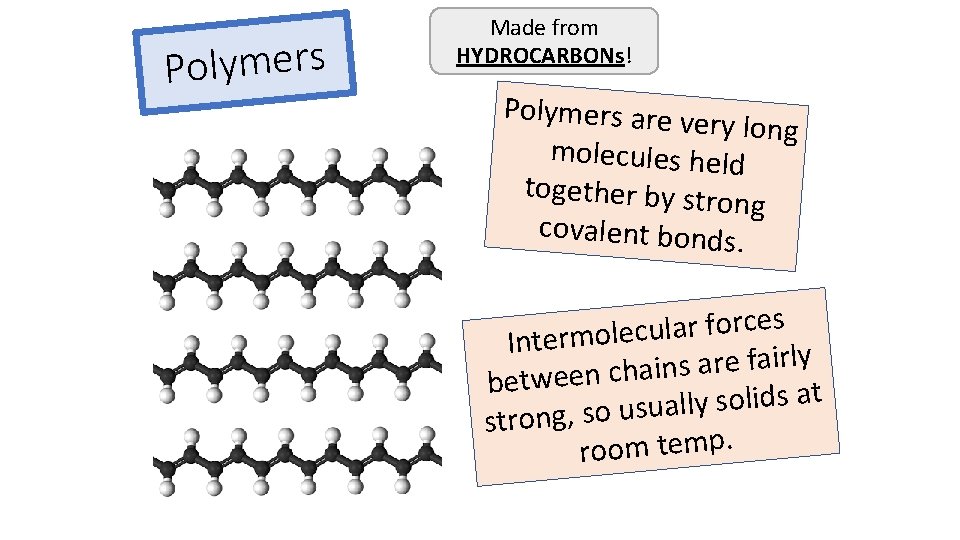
Polymers Made from HYDROCARBONs! Polymers are ve ry long molecules held together by stro ng covalent bonds. s e c r o f r a l u c e l Intermo y l r i a f e r a s n i a between ch t a s d i l o s y l l a u s u o s , g n o r t s room temp.
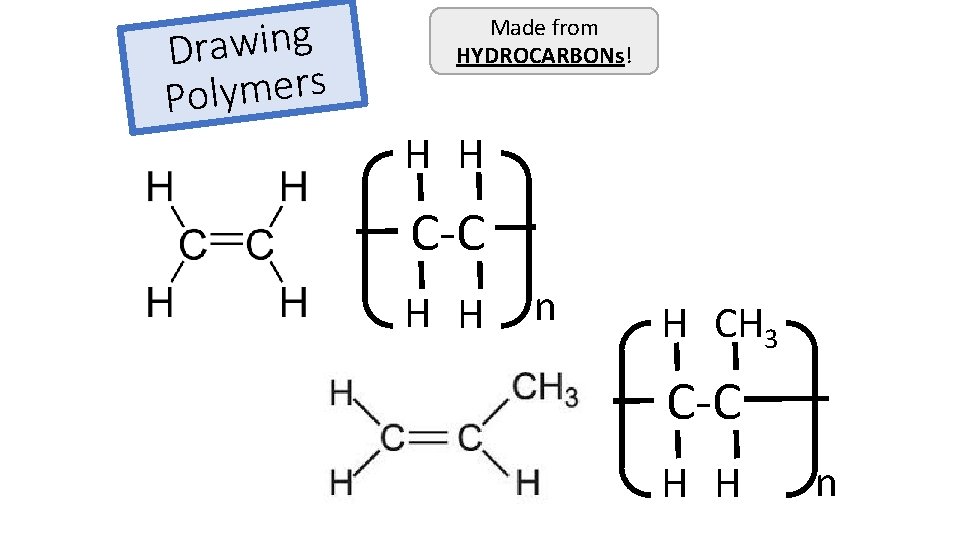
g n i w a r D s r e m y l Po Made from HYDROCARBONs! H H C-C H H n H CH 3 C-C H H n
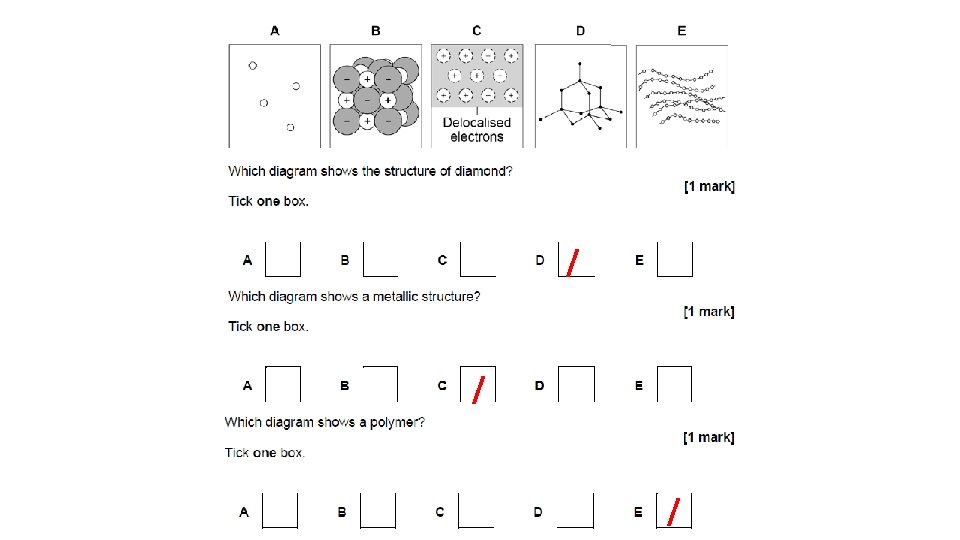


= g n i d n o B Metallic d e s i l a c o l e d electrons What holds the Electrostatic forces molecules together? Are they strong or Strong weak? Are they easy or hard Hard to break? High or low melting High point? Delocalised electrons, carry Does it conduct electricity? charge, conduct electricity

f o s e i t r e p Pro s y o l l A & s l a t Me Pure metals are arranged in layers. Layers can slide over each other. Therefore pure metals are malleable (bendable). The layers of alloys are disrupted, so cannot slide over each other. So alloys are harder than the pure metals they come from.


& s e l c i t r a P Sizes of s e i t r e p o r P their They have a large surface area to volume ratio. Nano = 1 -100 nm Fine = 100 -2500 nm Coarse = 1 x 10 -5 -1 x 10 -6 m Nanopar ticles hav e different propertie s to the same materials in bulk. Triple Only

Triple Only Nanoscience is the study of small particles that are between 1 and 100 nanometres in size. Particles consisting of fewer than 100 atoms are often called nanoclusters. The size of a typical nanoparticle is … 1 nanometre (1 nm) = 1 x 10 -9 metres (0. 000 001 m or a billionth of a metre). Nanoparticles are smaller than fine particles (PM 2. 5) which have diameters between 1 x 10 -7 metres and 2. 5 x 10 -6. To comprehend how small this is, coarse particles, like dust, have diameters between 1 x 10 -5 and 2. 5 x 10 -6. … to a football as a football is … …to the Earth

Uses of s e l c i t r a p Nano medicine Triple Only electronics Sun cream deodorants catalysts cosmetics
- Slides: 38