Byetta Victoza Cycloset and Metformin Lipid and CVD

Byetta, Victoza, Cycloset and Metformin Lipid and CVD Benefits

Byetta (exenatide) • Efficacy and safety of exenatide once weekly versus sitagliptin or pioglitazone as an adjunct to metformin for treatment of type 2 diabetes (DURATION-2): a randomised trial – Bergenstal RB et al. Lancet 2010 Aug 7; 376(9739): 431 -9.
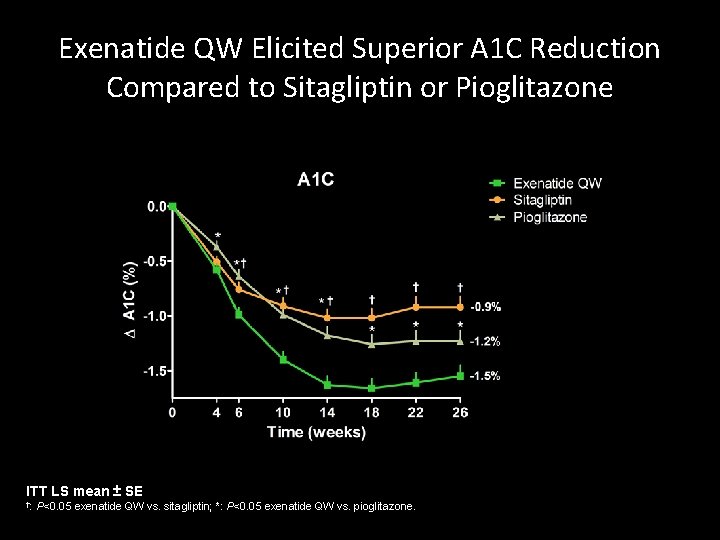
Exenatide QW Elicited Superior A 1 C Reduction Compared to Sitagliptin or Pioglitazone ITT LS mean ± SE †: P<0. 05 exenatide QW vs. sitagliptin; *: P<0. 05 exenatide QW vs. pioglitazone.
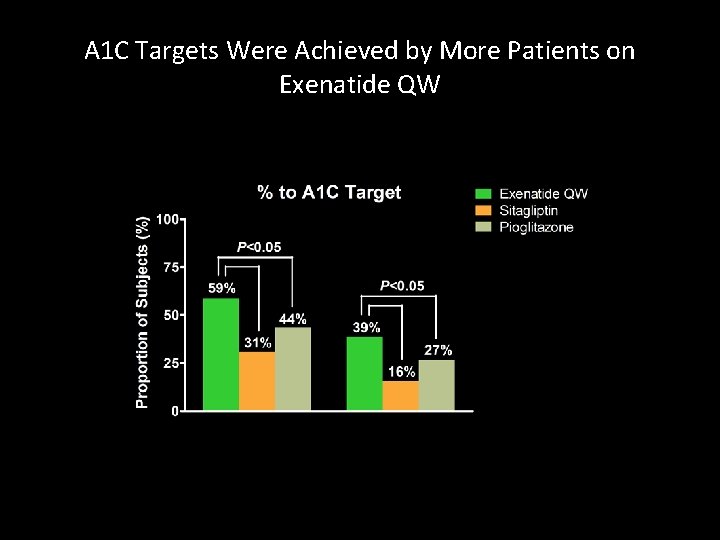
A 1 C Targets Were Achieved by More Patients on Exenatide QW
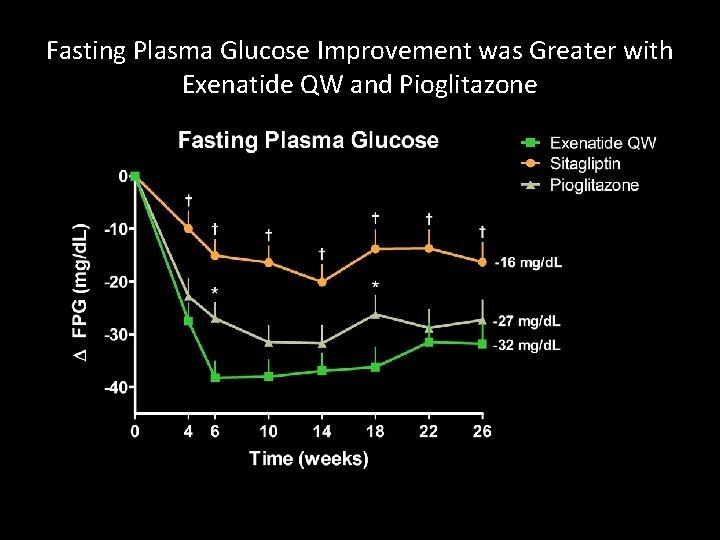
Fasting Plasma Glucose Improvement was Greater with Exenatide QW and Pioglitazone
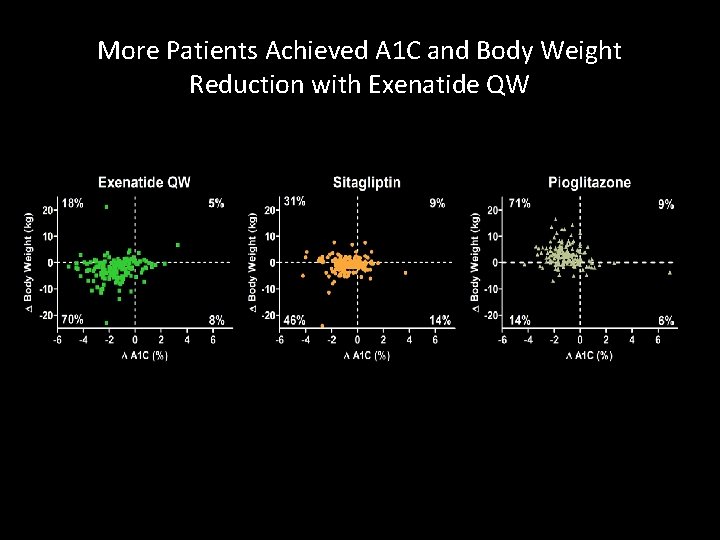
More Patients Achieved A 1 C and Body Weight Reduction with Exenatide QW
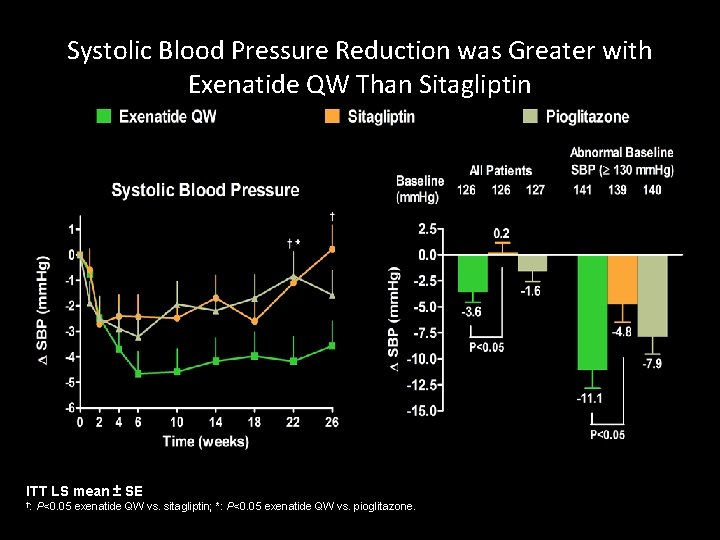
Systolic Blood Pressure Reduction was Greater with Exenatide QW Than Sitagliptin ITT LS mean ± SE †: P<0. 05 exenatide QW vs. sitagliptin; *: P<0. 05 exenatide QW vs. pioglitazone.
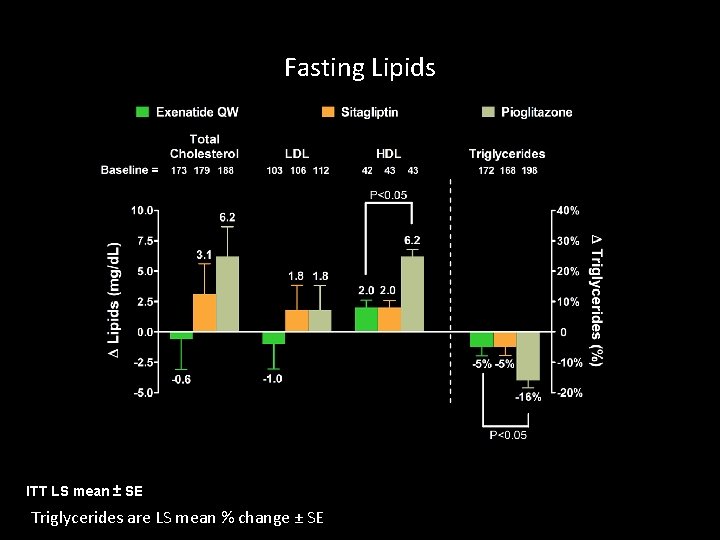
Fasting Lipids ITT LS mean ± SE Triglycerides are LS mean % change ± SE
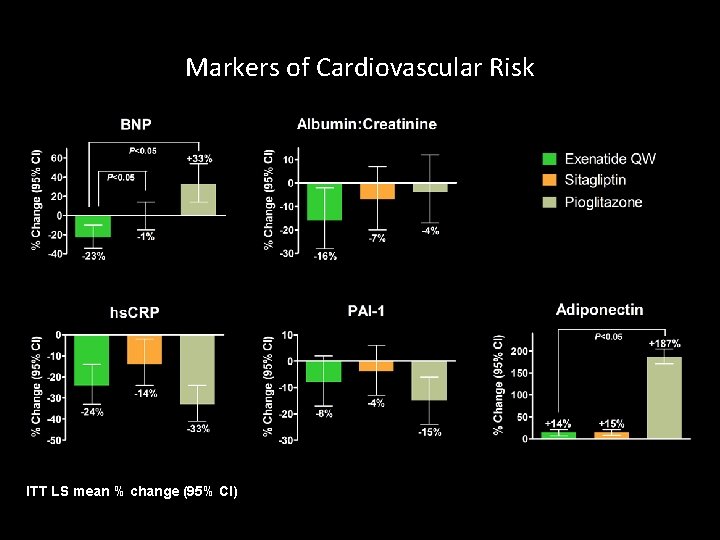
Markers of Cardiovascular Risk ITT LS mean % change (95% CI)

Summary • Once-weekly exenatide elicited superior improvements in: – Glucose control – Body weight reduction • All treatments were associated with: – Low rates of hypoglycemia – Low rates of withdrawal due to gastrointestinal AEs • Exenatide QW was the only treatment that improved SBP • All treatments improved HDL, hs. CRP, and adiponectin – Exenatide QW: improved BNP and albumin: creatinine – Pioglitazone: greatest HDL, triglyceride, and PAI-1 improvement, but was associated with increased total cholesterol and BNP

Byetta (exenatide) DURATION Program: Once Weekly Effects on the Cardiovascular System • Efficacy and safety of exenatide once weekly versus sitagliptin or pioglitazone as an adjunct to metformin for treatment of type 2 diabetes (DURATION-2): a randomised trial – Bergenstal RB et al. Lancet 2010 Aug 7; 376(9739): 431 -9. • Exenatide once weekly versus twice daily for the treatment of type 2 diabetes: a randomised open-label, non-inferiority study. – Drucker DJ, Buse JB, Taylor K, et al. Lancet 2008 Oct 4; 372(9645): 1240 -50. • Once weekly exenatide compared with insulin glargine titrated to target in patients with type 2 diabetes (DURATION 3): an open-label randomised trial. – Diamant M, Van Gaal L, Northrup J, et al. Lancet. 2010 Jun 26; 375(9733): 2234 -43.
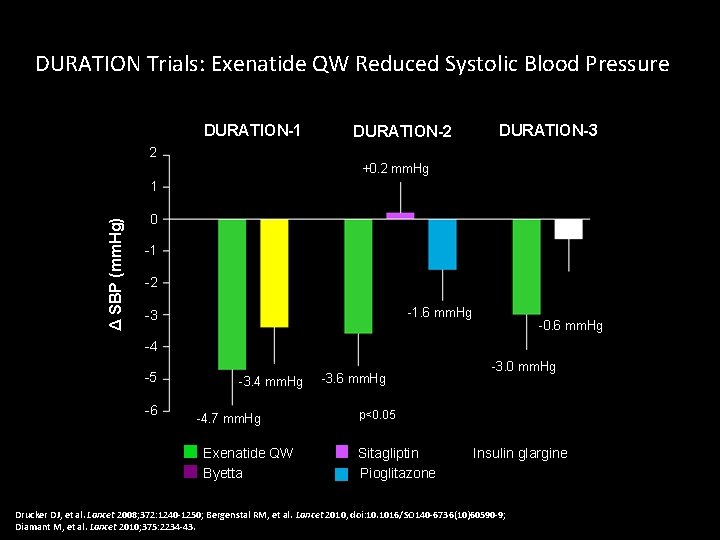
DURATION Trials: Exenatide QW Reduced Systolic Blood Pressure DURATION-1 DURATION-2 DURATION-3 2 +0. 2 mm. Hg Δ SBP (mm. Hg) 1 0 -1 -2 -1. 6 mm. Hg -3 -0. 6 mm. Hg -4 -5 -6 -3. 4 mm. Hg -4. 7 mm. Hg Exenatide QW Byetta -3. 6 mm. Hg -3. 0 mm. Hg p<0. 05 Sitagliptin Pioglitazone Insulin glargine Drucker. DJ, et etal. Lancet 2008; 372: 1240 -1250; Bergenstal. RM, et etal. Lancet 2010, doi: 10. 1016/SO 140 -6736(10)60590 -9; Diamant. M, M, et etal. Lancet 2010; 375: 2234 -43.
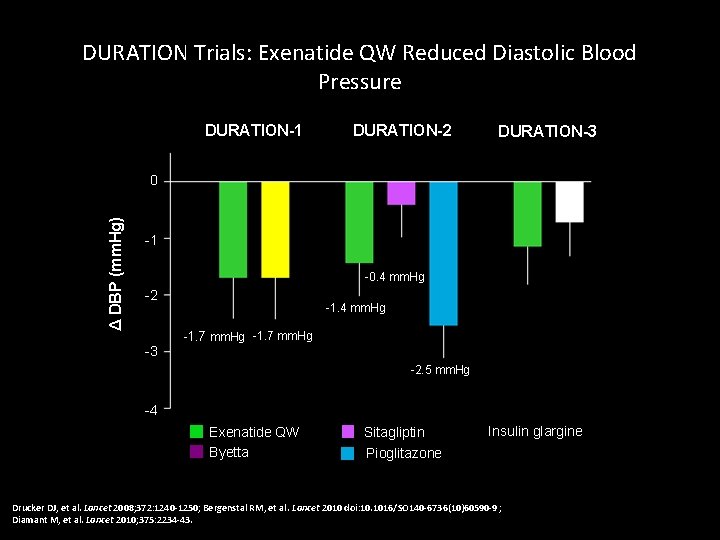
DURATION Trials: Exenatide QW Reduced Diastolic Blood Pressure DURATION-1 DURATION-2 DURATION-3 Δ DBP (mm. Hg) 0 -1 -0. 7 mm. Hg -0. 4 mm. Hg -2 -3 -1. 2 mm. Hg -1. 4 mm. Hg -1. 7 mm. Hg -2. 5 mm. Hg -4 Exenatide QW Byetta Sitagliptin Pioglitazone Insulin glargine Drucker DJ, et al. Lancet 2008; 372: 1240 -1250; Bergenstal RM, et al. Lancet 2010 doi: 10. 1016/SO 140 -6736(10)60590 -9 ; Diamant M, et al. Lancet 2010; 375: 2234 -43.
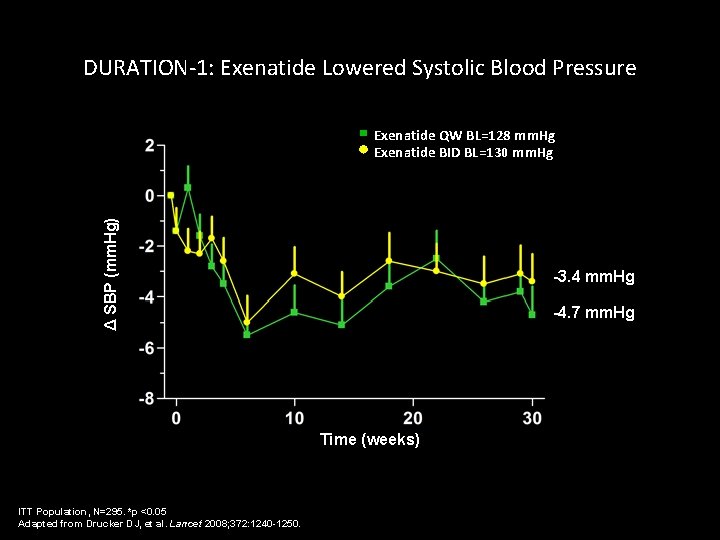
DURATION-1: Exenatide Lowered Systolic Blood Pressure Δ SBP (mm. Hg) * Exenatide QW BL=128 mm. Hg Exenatide BID BL=130 mm. Hg -3. 4 mm. Hg -4. 7 mm. Hg Time (weeks) ITT Population, N=295. *p *p <0. 05 ITT Adapted from Drucker DJ, et al. Lancet 2008; 372: 1240 -1250. Adapted from Drucker DJ, et
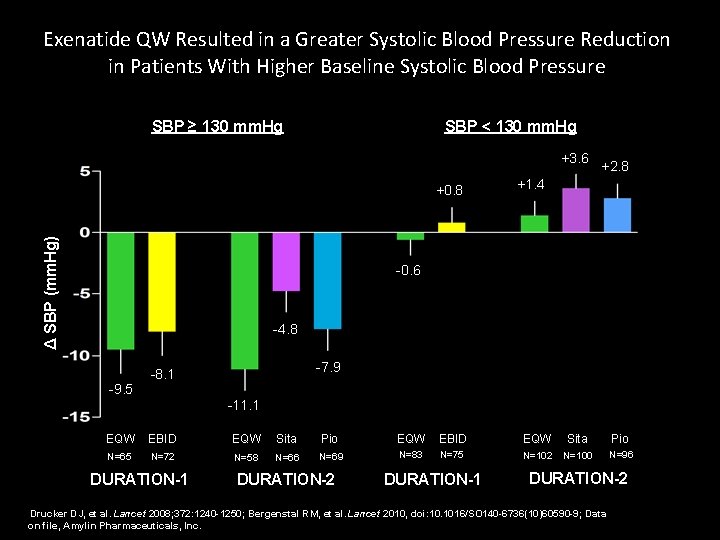
Exenatide QW Resulted in a Greater Systolic Blood Pressure Reduction in Patients With Higher Baseline Systolic Blood Pressure SBP ≥ 130 mm. Hg SBP < 130 mm. Hg +3. 6 Δ SBP (mm. Hg) +0. 8 +2. 8 +1. 4 -0. 6 -4. 8 -9. 5 -7. 9 -8. 1 -11. 1 EQW EBID EQW Sita Pio N=65 N=72 N=58 N=66 N=69 N=83 N=75 N=102 N=100 N=96 DURATION-1 DURATION-2 Drucker et al. Lancet 2008; 372: 1240 -1250; Bergenstal RM, et al. Lancet 2010, doi: 10. 1016/SO 140 -6736(10)60590 -9; Data Drucker DJ, et al. Lancet 2008; 372: 1240 -1250; Bergenstal RM, et al. Lancet 2010, doi: 10. 1016/SO 140 -6736(10)60590 -9; Data file, Amylin Pharmaceuticals, Inc. onon file, Amylin Pharmaceuticals, Inc.
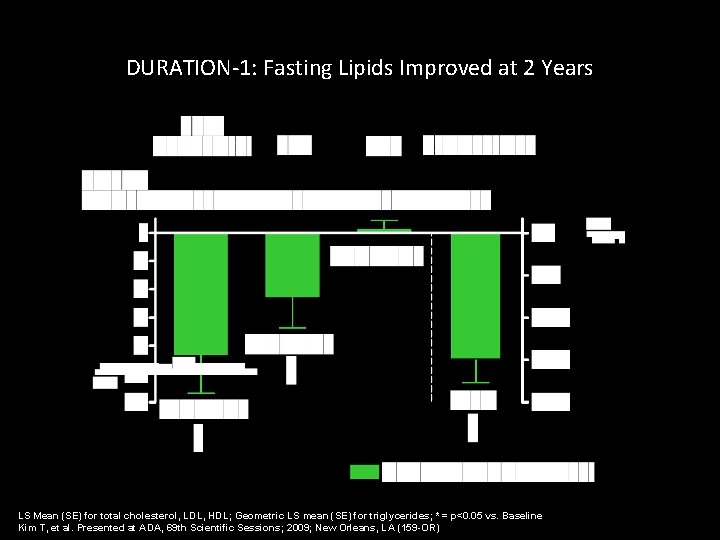
DURATION-1: Fasting Lipids Improved at 2 Years LS Mean (SE) for total cholesterol, LDL, HDL; Geometric LS mean (SE) for triglycerides; * = p<0. 05 vs. Baseline Kim T, et al. Presented at ADA, 69 th Scientific Sessions; 2009; New Orleans, LA (159 -OR)
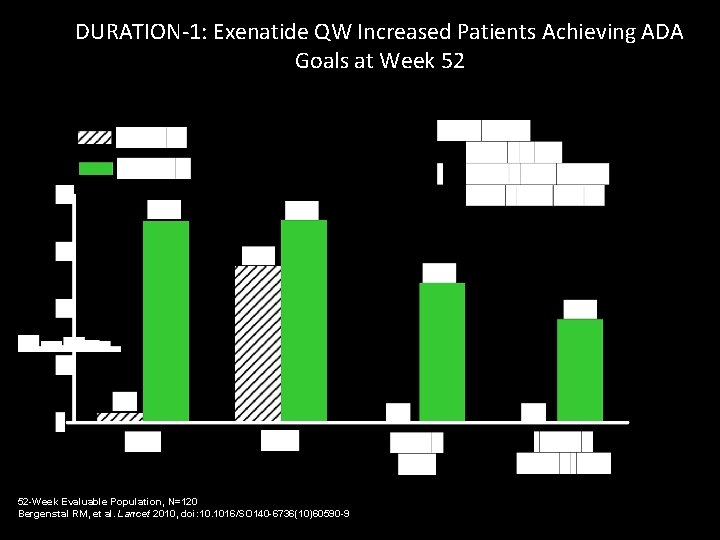
DURATION-1: Exenatide QW Increased Patients Achieving ADA Goals at Week 52 52 -Week Evaluable Population, N=120 Bergenstal RM, et al. Lancet 2010, doi: 10. 1016/SO 140 -6736(10)60590 -9
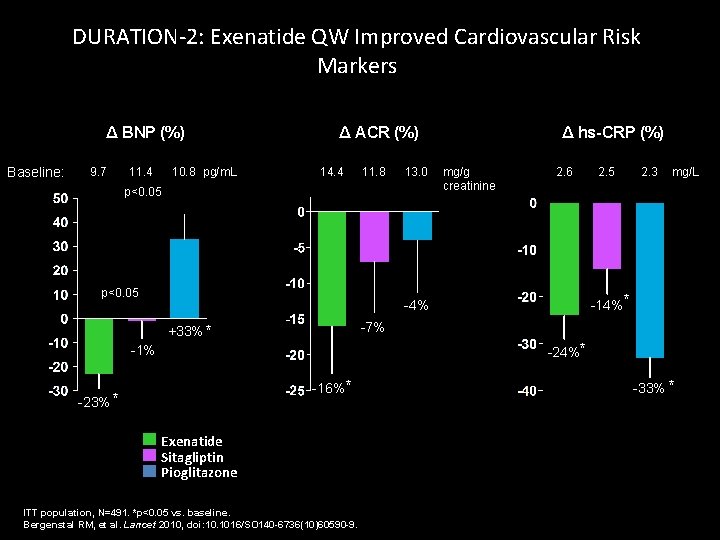
DURATION-2: Exenatide QW Improved Cardiovascular Risk Markers Δ BNP (%) Baseline: 9. 7 11. 4 10. 8 pg/m. L Δ ACR (%) 14. 4 11. 8 13. 0 p<0. 05 Δ hs-CRP (%) mg/g creatinine 2. 6 2. 3 mg/L -14%* -4% -7% +33% * -24%* -1% -16% * -23% * 2. 5 Exenatide QW, N=160 Sitagliptin, N=166 Pioglitazone, N=165 ITTpopulation, N=491. *p<0. 05 vs. baseline. ITT Bergenstal RM, et al. Lancet 2010, doi: 10. 1016/SO 140 -6736(10)60590 -9. -33% *

Cardiovascular Risk and Exenatide QW Systolic Blood Pressure Reduced Lipid profile Improved Cardiovascular risk markers Reduced

Victoza (liraglutide)

Cycloset (bromocriptine) • Bromocriptine-unique formulation of a dopamine agonist for the treatment of type 2 diabetes. – Scranton R and A Cincotta. Expert Opin Pharmacother. 2010 Feb; 11(2): 269 -79. • Quick release bromocriptin (Cycloset) a novel treatment for type 2 diabetes also demonstrates improvements in blood pressure. – Scranton R, Erzoki M, Farwell W, et al. IDF 2009 -20 th World Diabetes Congress North American Poster and Oral Abstracts. • Cycloset and Reduction of Major Adverse Cardiovascular Events (MACE) in T 2 DM Subjects – Vero. Science Data on File
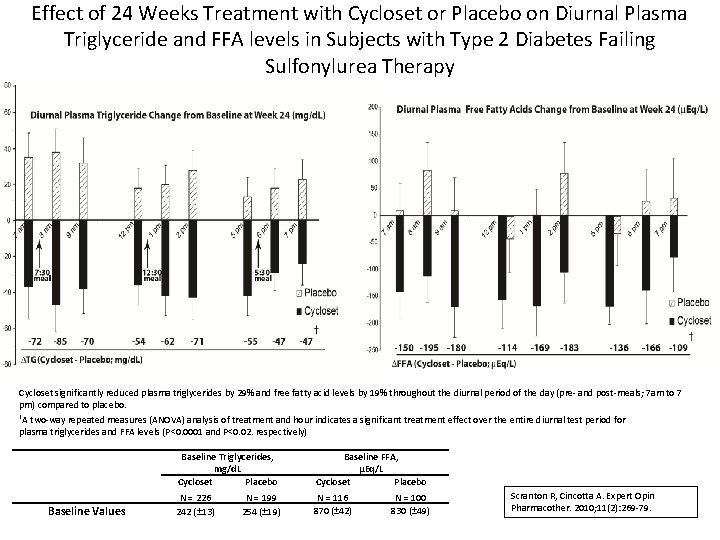
Effect of 24 Weeks Treatment with Cycloset or Placebo on Diurnal Plasma Triglyceride and FFA levels in Subjects with Type 2 Diabetes Failing Sulfonylurea Therapy Cycloset significantly reduced plasma triglycerides by 29% and free fatty acid levels by 19% throughout the diurnal period of the day (pre- and post-meals; 7 am to 7 pm) compared to placebo. †A two-way repeated measures (ANOVA) analysis of treatment and hour indicates a significant treatment effect over the entire diurnal test period for plasma triglycerides and FFA levels (P<0. 0001 and P<0. 02. respectively) Baseline Values Baseline Triglycerides, mg/d. L Cycloset Placebo Baseline FFA, µEq/L Cycloset Placebo N = 226 242 (± 13) N = 116 870 (± 42) N = 199 254 (± 19) N = 100 830 (± 49) Scranton R, Cincotta A. Expert Opin Pharmacother. 2010; 11(2): 269 -79.
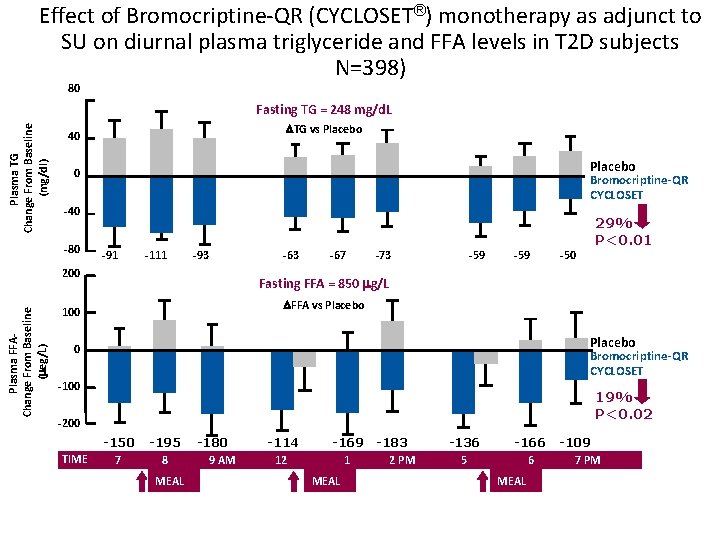
Effect of Bromocriptine-QR (CYCLOSET®) monotherapy as adjunct to SU on diurnal plasma triglyceride and FFA levels in T 2 D subjects N=398) 80 Plasma TG Change From Baseline (mg/dl) Fasting TG = 248 mg/d. L TG vs Placebo 40 Placebo 0 Bromocriptine-QR CYCLOSET -40 -80 29% P<0. 01 -91 -111 -93 Plasma FFAChange From Baseline ( eg/L) 200 -63 -67 -73 -59 Fasting FFA = 850 g/L FFA vs Placebo 100 Placebo 0 Bromocriptine-QR CYCLOSET -100 19% P<0. 02 -200 TIME -50 -150 7 -195 8 MEAL -180 9 AM -114 12 -169 1 MEAL -183 2 PM -136 5 -166 6 MEAL -109 7 PM
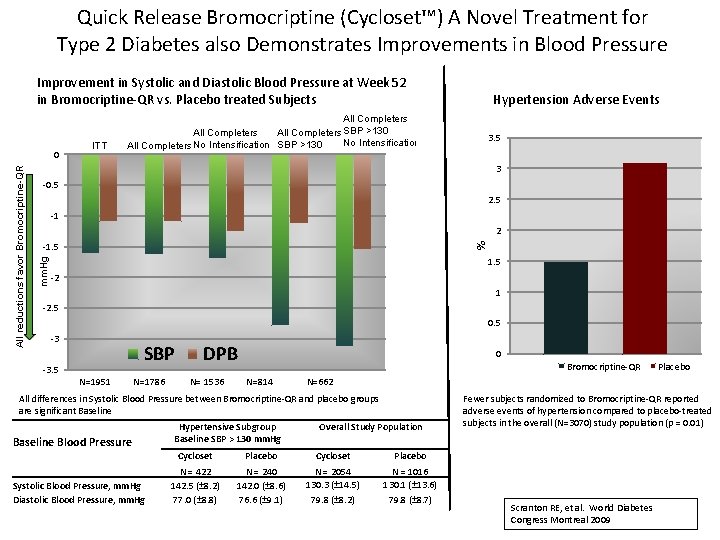
Quick Release Bromocriptine (Cycloset™) A Novel Treatment for Type 2 Diabetes also Demonstrates Improvements in Blood Pressure Improvement in Systolic and Diastolic Blood Pressure at Week 52 in Bromocriptine-QR vs. Placebo treated Subjects All Completers SBP >130 No Intensification All Completers No Intensification SBP >130 3. 5 3 -0. 5 2. 5 -1 2 % -1. 5 mm. Hg All reductions favor Bromocriptine-QR 0 ITT Hypertension Adverse Events 1. 5 -2 1 -2. 5 0. 5 -3 -3. 5 SBP DPB 0 Bromocriptine-QR Placebo N=1951 N=1786 N= 1536 N=814 N=662 All differences in Systolic Blood Pressure between Bromocriptine-QR and placebo groups are significant Baseline Blood Pressure Systolic Blood Pressure, mm. Hg Diastolic Blood Pressure, mm. Hg Hypertensive Subgroup Baseline SBP > 130 mm. Hg Overall Study Population Cycloset Placebo N = 422 142. 5 (± 8. 2) 77. 0 (± 8. 8) N = 240 142. 0 (± 8. 6) 76. 6 (± 9. 1) N = 2054 130. 3 (± 14. 5) 79. 8 (± 8. 2) N = 1016 130. 1 (± 13. 6) 79. 8 (± 8. 7) Fewer subjects randomized to Bromocriptine-QR reported adverse events of hypertension compared to placebo-treated subjects in the overall (N=3070) study population (p = 0. 01) Scranton RE, et al. World Diabetes Congress Montreal 2009
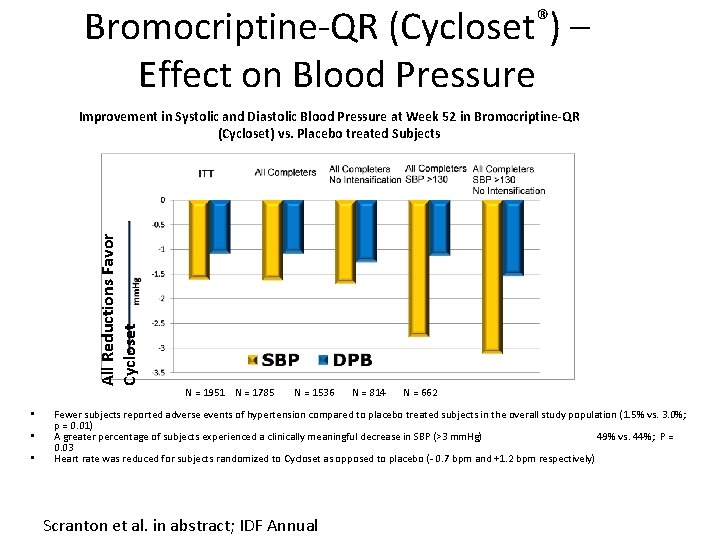
Bromocriptine-QR (Cycloset®) – Effect on Blood Pressure All Reductions Favor Cycloset Improvement in Systolic and Diastolic Blood Pressure at Week 52 in Bromocriptine-QR (Cycloset) vs. Placebo treated Subjects • • • N = 1951 N = 1785 N = 1536 N = 814 N = 662 Fewer subjects reported adverse events of hypertension compared to placebo treated subjects in the overall study population (1. 5% vs. 3. 0%; p = 0. 01) A greater percentage of subjects experienced a clinically meaningful decrease in SBP (>3 mm. Hg) 49% vs. 44%; P = 0. 03 Heart rate was reduced for subjects randomized to Cycloset as opposed to placebo (- 0. 7 bpm and +1. 2 bpm respectively) Scranton et al. in abstract; IDF Annual
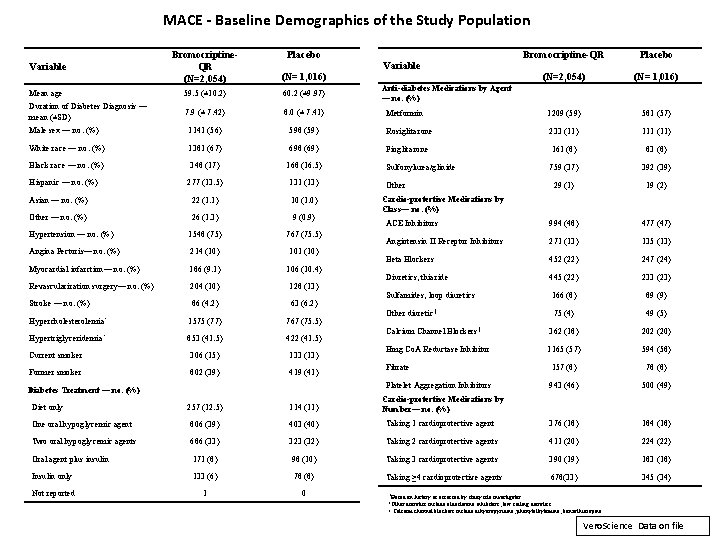
MACE - Baseline Demographics of the Study Population Bromocriptine. QR (N=2, 054) Bromocriptine-QR Placebo (N= 1, 016) (N=2, 054) (N= 1, 016) Mean age 59. 5 (± 10. 2) 60. 2 (± 9. 97) Duration of Diabetes Diagnosis — mean (±SD) 7. 9 (± 7. 42) 8. 0 (± 7. 41) Metformin 1209 (59) 581 (57) Male sex — no. (%) 1141 (56) 598 (59) Rosiglitazone 233 (11) 111 (11) White race — no. (%) 1381 (67) 698 (69) Pioglitazone 161 (8) 83 (8) Black race — no. (%) 348 (17) 168 (16. 5) Sulfonylurea/glinide 759 (37) 392 (39) 277 (13. 5) 131 (13) 29 (1) 19 (2) Asian — no. (%) 22 (1. 1) 10 (1. 0) Other — no. (%) 26 (1. 3) 9 (0. 9) ACE Inhibitors 994 (48) 477 (47) Hypertension — no. (%) 1548 (75) 767 (75. 5) Angiotensin II Receptor Inhibitors 271 (13) 135 (13) Angina Pectoris— no. (%) 214 (10) 101 (10) Beta Blockers 452 (22) 247 (24) Myocardial infarction— no. (%) 186 (9. 1) 106 (10. 4) Revascularization surgery— no. (%) 204 (10) 128 (13) Diuretics, thiazide 445 (22) 233 (23) Stroke — no. (%) 86 (4. 2) 63 (6. 2) Sulfamides, loop diuretics 166 (8) 89 (9) Hypercholesterolemia* 1575 (77) 767 (75. 5) Other diuretic† 75 (4) 49 (5) Hypertriglyceridemia* 853 (41. 5) 422 (41. 5) Calcium Channel Blockers‡ 362 (18) 202 (20) Current smoker 306 (15) 133 (13) Hmg Co. A Reductase Inhibitor 1165 (57) 594 (58) Former smoker 802 (39) 419 (41) Fibrate 157 (8) 78 (8) Platelet Aggregation Inhibitors 943 (46) 500 (49) Variable Hispanic — no. (%) Placebo Diabetes Treatment — no. (%) Diet only Variable Anti-diabetes Medications by Agent — no. (%) Other Cardio-protective Medications by Class— no. (%) Cardio-protective Medications by Number— no. (%) 257 (12. 5) 114 (11) One oral hypoglycemic agent 806 (39) 403 (40) Taking 1 cardioprotective agent 376 (18) 184 (18) Two oral hypoglycemic agents 686 (33) 323 (32) Taking 2 cardioprotective agents 411 (20) 224 (22) Oral agent plus insulin 171 (8) 98 (10) Taking 3 cardioprotective agents 390 (19) 183 (18) Insulin only 133 (6) 78 (8) Taking ≥ 4 cardioprotective agents 678(33) 345 (34) Not reported 1 0 *Based on history as assessed by study site investigator †Other diuretics include aldosterone inhibitors, low ceiling diuretics ‡ Calcium channel blockers include dihydropyridine, phenylalkylamine, benzothiazepine Vero. Science Data on file
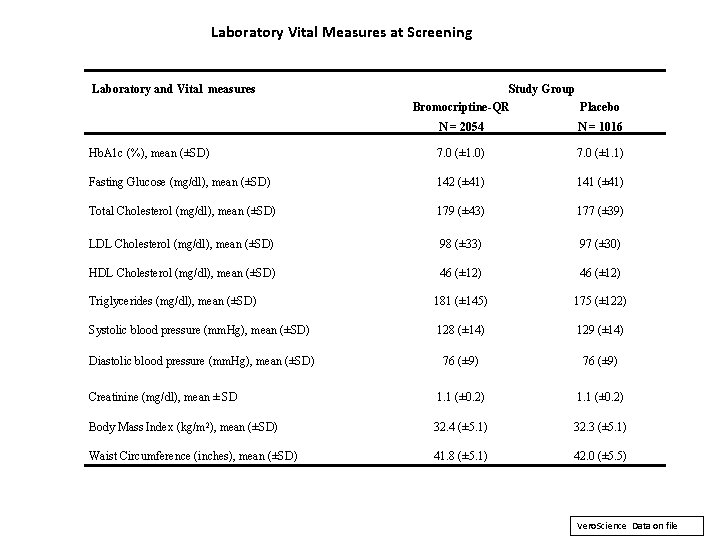
Laboratory Vital Measures at Screening Laboratory and Vital measures Study Group Bromocriptine-QR Placebo N = 2054 N = 1016 Hb. A 1 c (%), mean (±SD) 7. 0 (± 1. 0) 7. 0 (± 1. 1) Fasting Glucose (mg/dl), mean (±SD) 142 (± 41) 141 (± 41) Total Cholesterol (mg/dl), mean (±SD) 179 (± 43) 177 (± 39) LDL Cholesterol (mg/dl), mean (±SD) 98 (± 33) 97 (± 30) HDL Cholesterol (mg/dl), mean (±SD) 46 (± 12) Triglycerides (mg/dl), mean (±SD) 181 (± 145) 175 (± 122) Systolic blood pressure (mm. Hg), mean (±SD) 128 (± 14) 129 (± 14) Diastolic blood pressure (mm. Hg), mean (±SD) 76 (± 9) Creatinine (mg/dl), mean ± SD 1. 1 (± 0. 2) Body Mass Index (kg/m 2), mean (±SD) 32. 4 (± 5. 1) 32. 3 (± 5. 1) Waist Circumference (inches), mean (±SD) 41. 8 (± 5. 1) 42. 0 (± 5. 5) Vero. Science Data on file
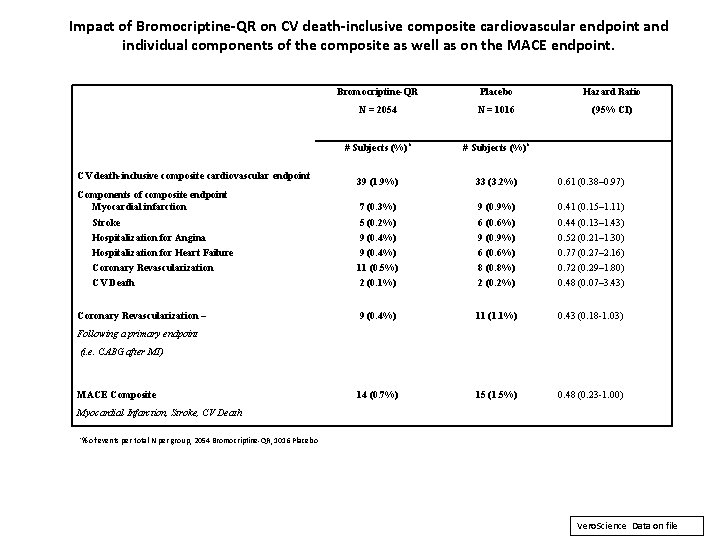
Impact of Bromocriptine-QR on CV death-inclusive composite cardiovascular endpoint and individual components of the composite as well as on the MACE endpoint. CV death-inclusive composite cardiovascular endpoint Bromocriptine-QR Placebo Hazard Ratio N = 2054 N = 1016 (95% CI) # Subjects (%)* 39 (1. 9%) 33 (3. 2%) 0. 61 (0. 38– 0. 97) Components of composite endpoint Myocardial infarction 7 (0. 3%) 9 (0. 9%) 0. 41 (0. 15– 1. 11) Stroke 5 (0. 2%) 6 (0. 6%) 0. 44 (0. 13– 1. 43) Hospitalization for Angina 9 (0. 4%) 9 (0. 9%) 0. 52 (0. 21– 1. 30) Hospitalization for Heart Failure 9 (0. 4%) 6 (0. 6%) 0. 77 (0. 27– 2. 16) Coronary Revascularization 11 (0. 5%) 8 (0. 8%) 0. 72 (0. 29– 1. 80) CV Death 2 (0. 1%) 2 (0. 2%) 0. 48 (0. 07– 3. 43) Coronary Revascularization – 9 (0. 4%) 11 (1. 1%) 0. 43 (0. 18 -1. 03) 14 (0. 7%) 15 (1. 5%) 0. 48 (0. 23 -1. 00) Following a primary endpoint (i. e. CABG after MI) MACE Composite Myocardial Infarction, Stroke, CV Death *% of events per total N per group, 2054 Bromocriptine-QR, 1016 Placebo Vero. Science Data on file
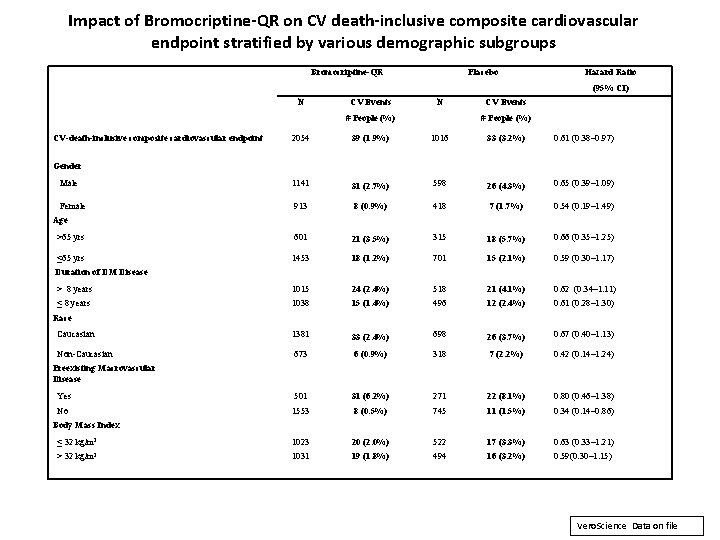
Impact of Bromocriptine-QR on CV death-inclusive composite cardiovascular endpoint stratified by various demographic subgroups Bromocriptine-QR Placebo Hazard Ratio (95% CI) N CV Events N # People (%) CV-death-inclusive composite cardiovascular endpoint CV Events # People (%) 2054 39 (1. 9%) 1016 33 (3. 2%) 0. 61 (0. 38– 0. 97) Male 1141 31 (2. 7%) 598 26 (4. 3%) 0. 65 (0. 39– 1. 09) Female 913 8 (0. 9%) 418 7 (1. 7%) 0. 54 (0. 19– 1. 49) >65 yrs 601 21 (3. 5%) 315 18 (5. 7%) 0. 66 (0. 35– 1. 25) ≤ 65 yrs 1453 18 (1. 2%) 701 15 (2. 1%) 0. 59 (0. 30– 1. 17) > 8 years 1015 24 (2. 4%) 518 21 (4. 1%) 0. 62 (0. 34– 1. 11) ≤ 8 years 1038 15 (1. 4%) 496 12 (2. 4%) 0. 61 (0. 28– 1. 30) Caucasian 1381 33 (2. 4%) 698 26 (3. 7%) 0. 67 (0. 40– 1. 13) Non-Caucasian 673 6 (0. 9%) 318 7 (2. 2%) 0. 42 (0. 14– 1. 24) Yes 501 31 (6. 2%) 271 22 (8. 1%) 0. 80 (0. 46– 1. 38) No 1553 8 (0. 5%) 745 11 (1. 5%) 0. 34 (0. 14– 0. 86) ≤ 32 kg/m 2 1023 20 (2. 0%) 522 17 (3. 3%) 0. 63 (0. 33– 1. 21) > 32 kg/m 2 1031 19 (1. 8%) 494 16 (3. 2%) 0. 59(0. 30– 1. 15) Gender Age Duration of DM Disease Race Preexisting Macrovascular Disease Body Mass Index Vero. Science Data on file
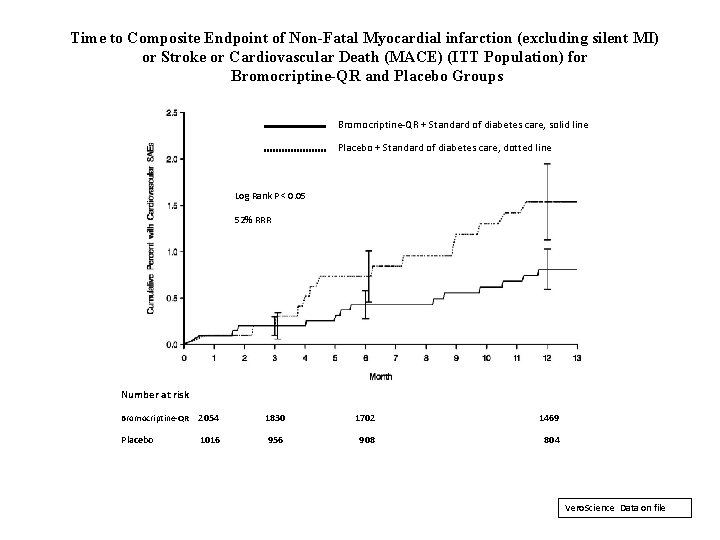
Time to Composite Endpoint of Non-Fatal Myocardial infarction (excluding silent MI) or Stroke or Cardiovascular Death (MACE) (ITT Population) for Bromocriptine-QR and Placebo Groups Bromocriptine-QR + Standard of diabetes care, solid line Placebo + Standard of diabetes care, dotted line Log Rank P < 0. 05 52% RRR Number at risk Bromocriptine-QR 2054 1830 1702 1469 Placebo 1016 956 908 804 Vero. Science Data on file
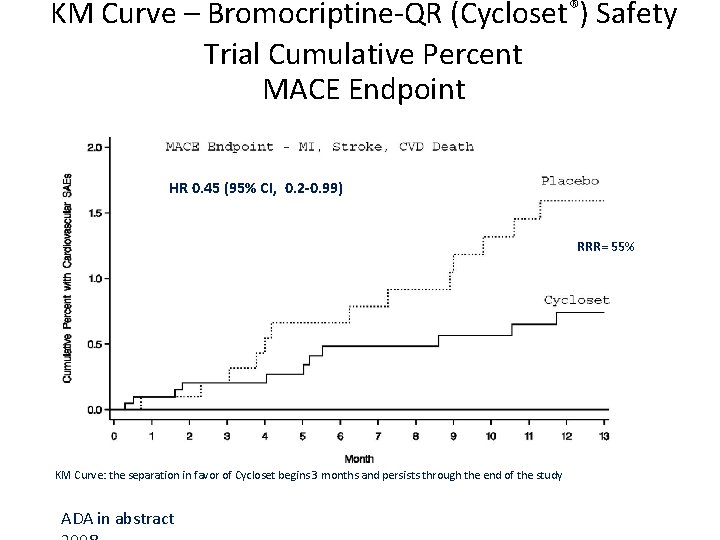
KM Curve – Bromocriptine-QR (Cycloset®) Safety Trial Cumulative Percent MACE Endpoint HR 0. 45 (95% CI, 0. 2 -0. 99) RRR= 55% KM Curve: the separation in favor of Cycloset begins 3 months and persists through the end of the study ADA in abstract
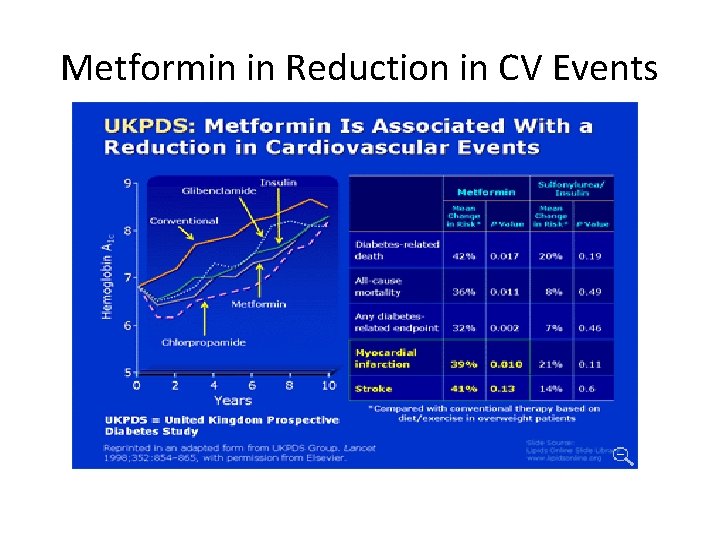
Metformin in Reduction in CV Events
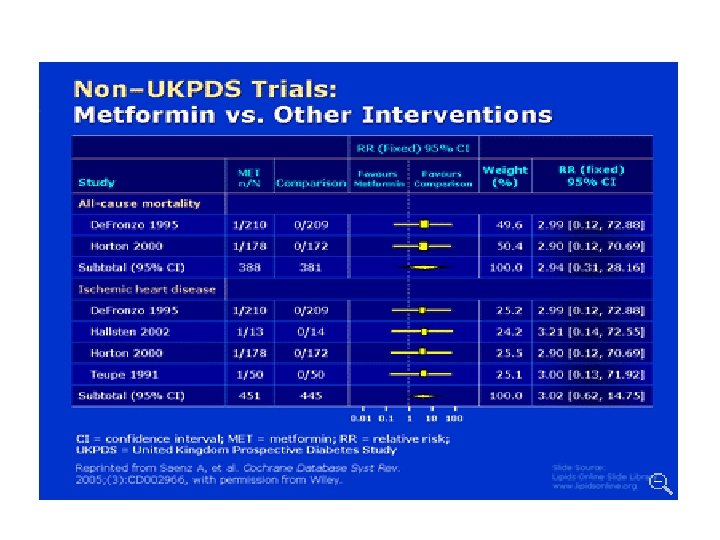
- Slides: 33