By Dr Firas majeed INTESTINAL OBSTRUCTION classification Intestinal

By Dr. Firas majeed INTESTINAL OBSTRUCTION

classification Intestinal obstruction may be classified into two types: • Dynamic, in which peristalsis is working against a mechanical obstruction. It may occur in an acute or a chronic form. • Adynamic, in which peristalsis may be absent (e. g. paralytic ileus) or it may be present in a non-propulsive form (e. g. mesenteric vascular occlusion or pseudoobstruction). In both types a mechanical element is absent

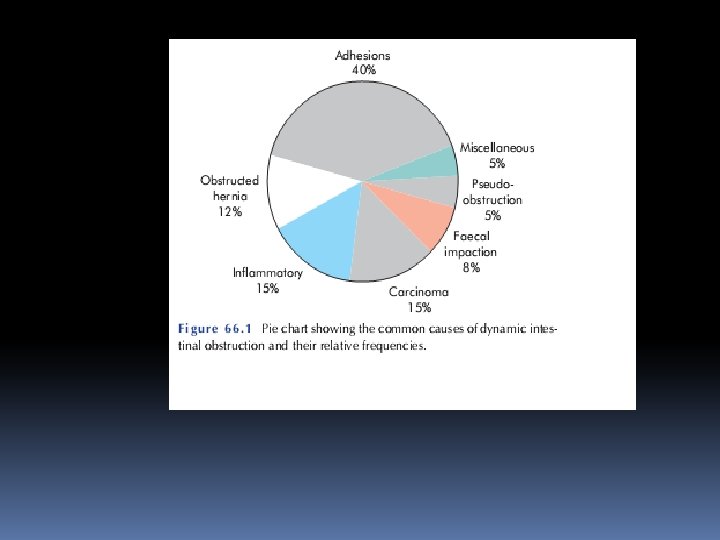
Causes of intestinal obstruction Dynamic ■ Intraluminal Impaction Foreign bodies Bezoars Gallstones ■ Intramural Stricture Malignancy ■ Extramural Bands/adhesions Hernia Volvulus Intussusception :

Adynamic ■ Paralytic ileus ■ Mesenteric vascular occlusion ■ Pseudo-obstruct

PATHOPHYSIOLOGY Irrespective of aetiology or acuteness of onset, in dynamic (mechanical) obstruction the proximal bowel dilates and devel-ops an altered motility. Below the obstruction the bowel exhibits normal peristalsis and absorption until it becomes empty, at which point it contracts and becomes immobile. Initially, proxi-mal peristalsis is increased to overcome the obstruction, in direct proportion to the distance of the obstruction.

If the obstruction is not relieved, the bowel begins to dilate, peristaltic strength, ultimately resulting in flaccidity and paralysis. This is a protective phenomenon to prevent vascular damage sec-ondary to increased intraluminal pressure.

The distension proximal to an obstruction is produced by two factors: • Gas: there is a significant overgrowth of both aerobic and anaerobic organisms, resulting in considerable gas production. Following the reabsorption of oxygen and carbon dioxide, the majority is made up of nitrogen (90%) and hydrogen sulphide. • Fluid: this is made up of the various digestive juices. Following obstruction, fluid accumulates within the bowel wall and any excess is secreted into the lumen, whilst absorption from the gut is retarded. Dehydration and electrolyte loss are therefore due to: – reduced oral intake; – defective intestinal absorption; – losses as a result of vomiting; – sequestration in the bowel lumen

STRANGULATION Causes of strangulation External ■ Hernial orifices ■ Adhesions/bands Interrupted blood flow ■ Volvulus ■ Intussusception Increased intraluminal pressure ■ Closed-loop obstruction Primary ■ Mesenteric infarction

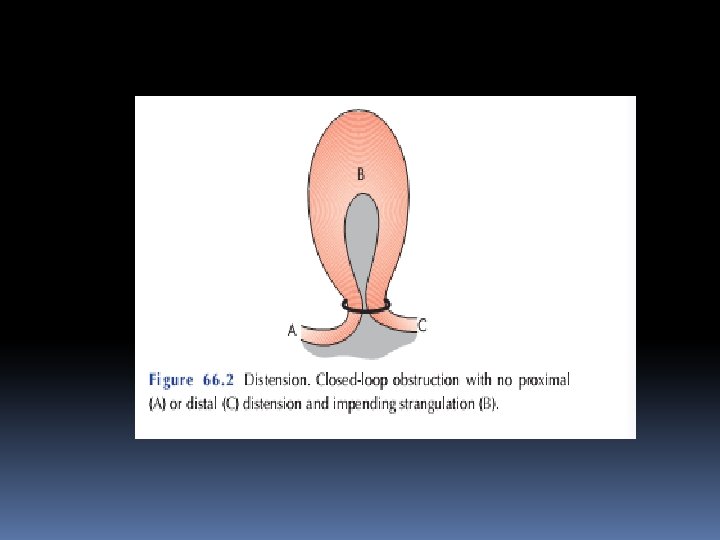
Closed-loop obstruction This occurs when the bowel is obstructed at both the proximal and distal points( A classic form of closed-loop obstruction is seen in the pres-ence of a malignant stricture of the right colon with a competent ileocaecal valve (present in up to one-third of individuals). The inability of the distended colon to decompress itself into the small bowel results in an increase in luminal pressure, which is greatest at the caecum, with subsequent impairment of blood supply. Unrelieved, this results in necrosis and perforation

Features of obstruction ■ In high small bowel obstruction, vomiting occurs early and is profuse with rapid dehydration. Distension is minimal with little evidence of fluid levels on abdominal radiography ■ In low small bowel obstruction, pain is predominant with central distension. Vomiting is delayed. Multiple central fluid levels are seen on radiography ■ In large bowel obstruction, distension is early and pronounced. Pain is mild and vomiting and dehydration are late. The proximal colon and caecum are distended on abdominal radiography.

The diagnosis of dynamic intestinal obstruction is based on theclassic quartet of pain, distension, vomiting and absolute constipation. Obstruction may be classified clinically into two types: • small bowel obstruction – high or low; • large bowel obstruction

The nature of the presentation will also be influenced by whether the obstruction is: • acute; • chronic; • acute on chronic; • subacute. Acute obstructionusually occurs in small bowel obstruction, with sudden onset of severe colicky central abdominal pain, distension and early vomiting and constipation.

Chronic obstruction is usually seen in large bowel obstruction , with lower abdominal colic and absolute constipation followedby distension. In acute on chronic obstruction there is a short history of distension and vomiting against a background of pain constipation. Subacute obstruction implies an incomplete obstruction.

Cardinal clinical features of acute obstruction ■ Abdominal pain ■ Distension ■ Vomiting ■ Absolute constipation Late manifestations of intestinal obstruction that may be encoun-tered include dehydration, oliguria, hypovolaemic shock, pyrexia, septicaemia, respiratory embarrassment and peritonism. In allcases of suspected intestinal obstruction, all hernial orifices must be examine

Clinical features of strangulation ■ Constant pain ■ Tenderness with rigidity ■ Shock Pyrexia in the presence of obstruction may indicate: • the onset of ischaemia; • intestinal perforation; • inflammation associated with the obstructing disease. Hypothermia indicates septicaemic shock

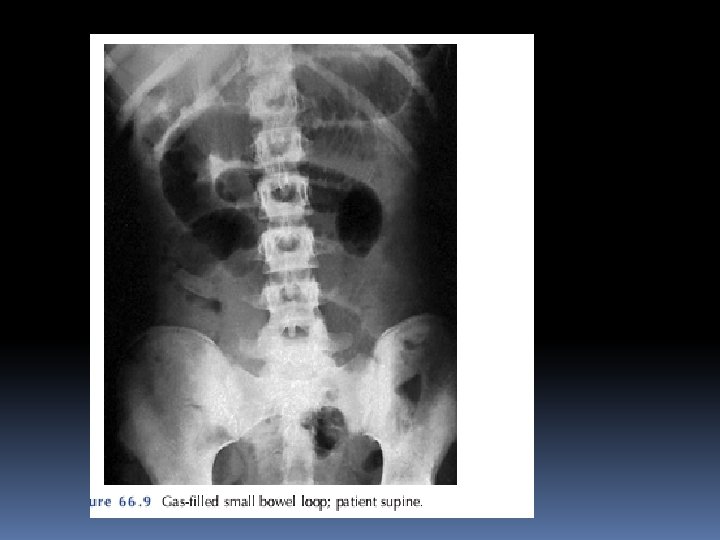
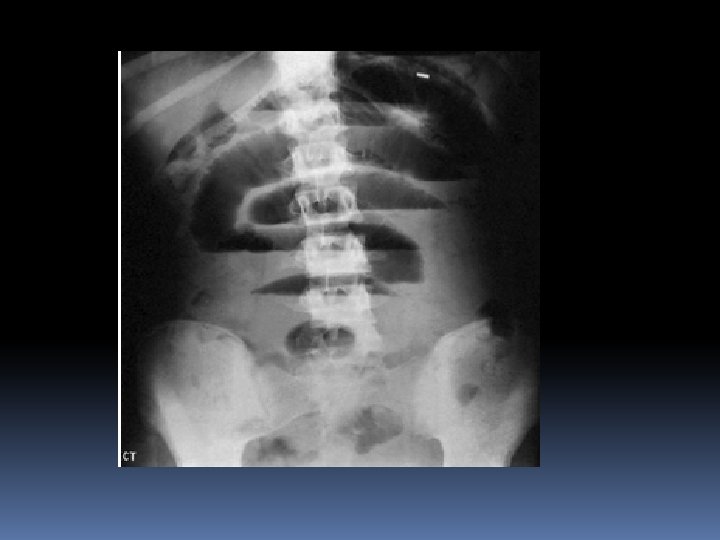
Radiological features of obstruction ■ The obstructed small bowel is characterised by straight segments that are generally central and lie transversely. No gas is seen in the colon ■ The jejunum is characterised by its valvulae conniventes, which completely pass across the width of the bowel and are regularly spaced, giving a ‘concertina’ or ladder effect ■ Ileum – the distal ileum has been piquantly described by Wangensteen as featureless

■ Caecum – a distended caecum is shown by a rounded gas shadow in the right iliac fossa ■ Large bowel, except for the caecum, shows haustral folds, which, unlike valvulae conniventes, are spaced irregularly, do not cross the whole diameter of the bowel and do not have indentations placed opposite one another

Treatment of acute intestinal obstruction ■ Gastrointestinal drainage ■ Fluid and electrolyte replacement ■ Relief of obstruction ■ Surgical treatment is necessary for most cases of intestinal obstruction but should be delayed until resuscitation is complete, provided there is no sign of strangulation or evidence of closed-loop obstruction

Principles of surgical intervention for obstruction Management of: ■ The segment at the site of obstruction ■ The distended proximal bowel ■ The underlying cause of obstruction Indications for early surgical intervention ■ Obstructed or strangulated external hernia ■ Internal intestinal strangulation ■ Acute obstruction

The classic clinical advice that ‘the sun should not both rise and set’ on a case of unrelieved acute intestinal obstruction is soundand should be followed unless there are positive reasons for delay. Such cases may include obstruction secondary to adhesions when there is no pain or tenderness, despite continued radiological evidence of obstruction. In these circumstances, conservativemanagement may be continued for up to 72 hours in the hope of spontaneous resolution.

If the site of obstruction is unknown, adequate exposure is best achieved by a midline incision. Assessment is directed to: • the site of obstruction; • the nature of the obstruction; • the viability of the gut.

Identification and assessment of the caecum is the best initial manoeuvre. If it is collapsed, the lesion is in the small bowel and may be identified by careful retrograde assessment. A dilated caecum indicates large bowel obstruction. To display the cause of obstruction, distended loops of small bowel should be displaced with care and covered with warm moist abdominal packs.


SPECIAL TYPES OF MECHANICAL INTESTINAL OBSTRUCTION Internal herniation occurs when a portion of the small intestine becomes entrapped in one of the retroperitoneal fossae or in a congenital mesenteric defect. The following are potential sites of internal herniation: • the foramen of Winslow; • a hole in the mesenter

• a hole in the transverse mesocolon; • defects in the broad ligament; • congenital or acquired diaphragmatic hernia; • duodenal retroperitoneal fossae – left paraduodenal and right duodenojejunal; • caecal/appendiceal retroperitoneal fossae – superior, inferior and retrocaecal; • intersigmoid fossa

Obstruction from enteric strictures Small bowel strictures usually occur secondary to tuberculosis or Crohn’s disease. Malignant strictures associated with lymphoma are common, whereas carcinoma and sarcoma are rare. Present-ation is usually subacute or chronic. Standard surgical manage-ment consists of resection and anastomosis. In Crohn’s disease, strictureplasty may be considered in the presence of short multiple strictures without active sepsis.
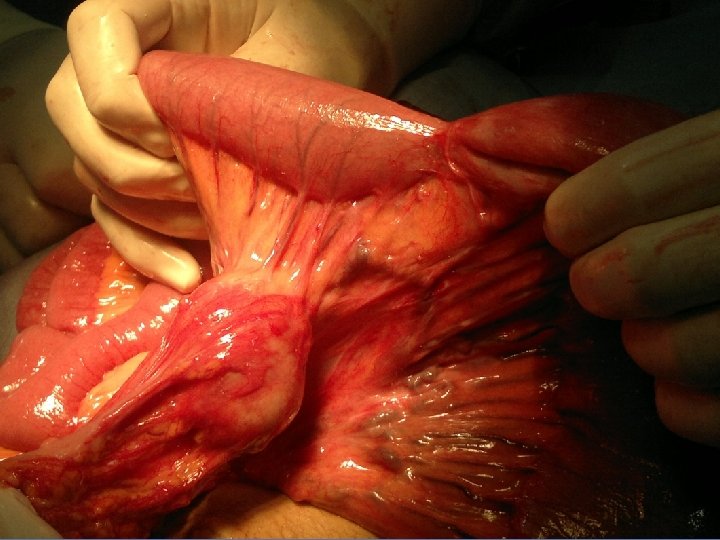

Bolus obstruction in the small bowel may be caused by food, gall-stones, trichobezoar, phytobezoar, stercoliths and worms. Gallstone. This type of obstruction tends to occur in the elderly secondary to erosion of a large gallstone through the gall bladder into the duodenum. Classically, there is impaction about 60 cm proximal to the ileocaecal valve. The patient may have recurrent attacks as the obstruction is frequently incomplete or relapsing as a result of a ball-valve effect. A radiograph will show evidence of small bowel obstruction with a diagnostic air–fluid level in the biliary tree. The stone may not be visible.

At laparotomy it may be poss-ible to crush the stone within the bowel lumen, after milking it proximally. If not, the intestine is opened and the gallstoneremoved. If the gallstone is faceted, a careful check for other enteric stones should be made. The region of the gall bladder should not be explored Food Bolus obstruction may occur after partial or total gastrectomy when unchewed articles can pass directly into the small bowel. Fruit and vegetables are particularly liable to cause obstruction. The management is similar to that for gallstone, with intra-luminal crushing usually being successful.

Trychobezoars and phytobezoars These are firm masses of undigested hair balls and fruit/vegetable fibre respectively. The former is due to persistent hair chewing sucking, and may be associated with an underlying psychiatric abnormality. Predisposition to phytobezoars results from a high fibre intake, inadequate chewing, previous gastric surgery, hypochlorhydria and loss of the gastric pump mechanism. When possible, the lesion may be kneaded into the caecum, otherwise open removal is required Stercoliths These are usually found in the small bowel in association with a jejunal diverticulum or ileal stricture. Presentation and manage-ment are identical to that of gallstones

Worms Ascaris lumbricoides may cause low small bowel obstruction, particularly in children, the institutionalised and those near the tropics. An attack frequently follows the initiation of anti-helminthic therapy. Debility is frequently out of proportion to that produced by the obstruction. If worms are not seen in the stool or vomitus the diagnosis may be indicated by eosinophilia or the sight of worms within gas-filled small bowel loops on a plain radiograph (Naik). At laparotomy it may be possible to knead the tangled mass into the caecum; if not it should be removed. Occasionally, worms may cause a perforation and peritonitis, especially if the enteric wall is weakened by such conditions as ameobiasis.


Obstruction by adhesions and bands . Adhesions In western countries where abdominal operations are common, adhesions and bands are the most common cause of intestinal obstruction. Furthermore, in the early postoperative period, the onset of such a mechanical obstruction may be difficult to differentiate from paralytic ileus. Any source of peritoneal irritation results in local fibrin production, which produces adhesions between apposed surfaces. Early fibrinous adhesions may disappear when the cause is removed or they may become vascularised and be replaced by mature fibrous tissue.

The common causes of intra-abdominal adhesions • Ischaemic areas Sites of anastomoses, reperitonealisation of raw areas, trauma, vascular occlusion • Foreign material Talc, starch, gauze, silk • Infection Peritonitis, tuberculosis • Inflammatory conditions Crohn’s disease • Radiation enteritis

Prevention of adhesions Factors that may limit adhesion formation include: ■ Good surgical technique ■ Washing of the peritoneal cavity with saline to remove clots, etc. ■ Minimising contact with gauze ■ Covering anastomosis and raw peritoneal surfaces

Adhesions may be classified into various types by virtue of whether they are early (fibrinous) or late (fibrous) or by under -lying aetiology. From a practical perspective there are only two types – ‘easy’ flimsy ones and ‘difficult’ dense ones Postoperative adhesions giving rise to intestinal obstruction usually involve the lower small bowel. Operations for appendicitis and gynaecological procedures are the most common precursors and are an indication for early intervention.

Bands Usually one band is culpable. This may be: • congenital, e. g. obliterated vitellointestinal duct; • a string band following previous bacterial peritonitis; • a portion of greater omentum, usually adherent to the pariete

Treatment of adhesive obstruction ■ Initially treat conservatively provided there are no signs of strangulation; should rarely continue conservative treatment for longer than 72 hours ■ At operation, divide only the causative adhesion(s) and limit dissection ■ Cover serosal tears; invaginate (or resect) areas of doubtful viability ■ Laparoscopic adhesiolysis may have a role in chronic cases

Postoperative intestinal obstruction Differentiation between persistent paralytic ileus and early mechanical obstruction may be difficult in the early postoperative period. In practice, the latter is probably more common. evidence of obstruction (days 1– 5) is usually due to nonstrangu-lating causes such as fibrinous adhesions and oedema. Obstruction is usually incomplete and the majority settle withcontinued conservative management. Late postoperativeobstruction (> 7 days) is usually more significant in nature, and timely surgical intervention is usually required.

Acute intussusception This occurs when one portion of the gut becomes invaginated within an immediately adjacent segment; almost invariably, it is the proximal into the distal. The condition is encountered most commonly in children, with a peak incidence between 5 and 10 months of age. About 90% of cases are idiopathic but an associated upper respiratory tract infection or gastroenteritis may precede the condition.

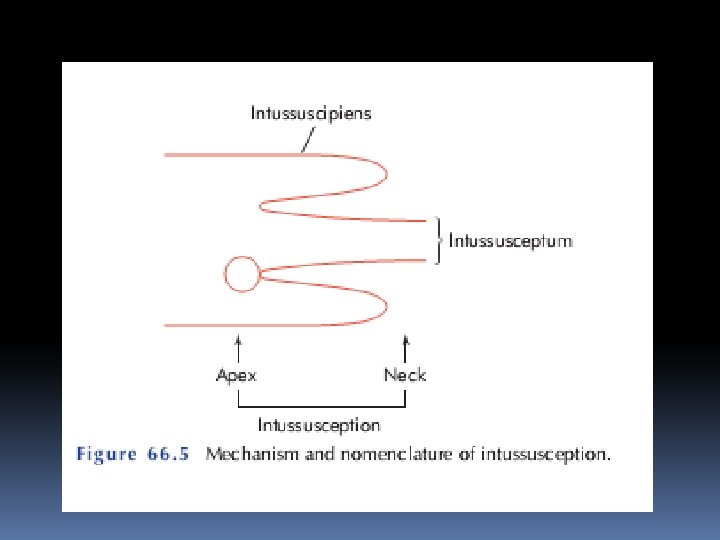
Children with intussusception associated with apathological lead point such as Meckel’s diverticulum, polyp, duplication, Henoch–Schönlein purpura or appendix are usually older than those with idiopathic disease. After the age of 2 years, a patholog-ical lead point is found in at least one-third of affected children. Adult cases are invariably associated with a lead point, which is usually a polyp (e. g. Peutz–Jeghers syndrome), a submucosal lipo-ma or other tumour. Pathology An intussusception is composed of three parts. • the entering or inner tube; • the returning or middle tube; • the sheath or outer tube (intussuscipiens)

■ Most common in children ■ Primary or secondary to intestinal pathology, e. g. polyp, Meckel’s diverticulum ■ Ileocolic is the commonest variety ■ Can lead to an ischaemic segment ■ Radiological reduction is indicated in most cases ■ The remainder require surgery In adults, colocolic intussusception is common.

Types of intussusception in children (after R. E. Gross) (n= 702) Percentage of series Ileoileal 5 Ileocolic 77 Ileoileocolic 12 Colocolic 2 Multiple 1 Retrograde 0. 2 Others 2. 8

Clinical features of intussusception The classical presentation of is with intussusception episodes of screaming and drawing up of the legs in a previously well male infant. The attacks last for a few minutes and recur repeatedly. During attacks the child appears pale, whereas between episodes he may be listless. Vomiting may or may not occur at the outset but becomes conspicuous and bile-stained with time. Initially, thepassage of stool may be normal, whereas, later, blood and mucus are evacuated – the ‘redcurrant jelly’ stool.

Whenever possible, examination should be undertaken between episodes of colic , without disturbing the child. Classically, the abdomen is not initially distended a lump that hardens on palpation may be discerned but this is present in only 60% of cases). There may be an associated feeling of emptiness in the right iliac fossa (the sign of Dance). On rectalexamination, blood-stained mucus may be found on the finger. Occasionally, in extensive ileocolic or colocolic intussusception, the apex may be palpable or even protrude from the anus. -
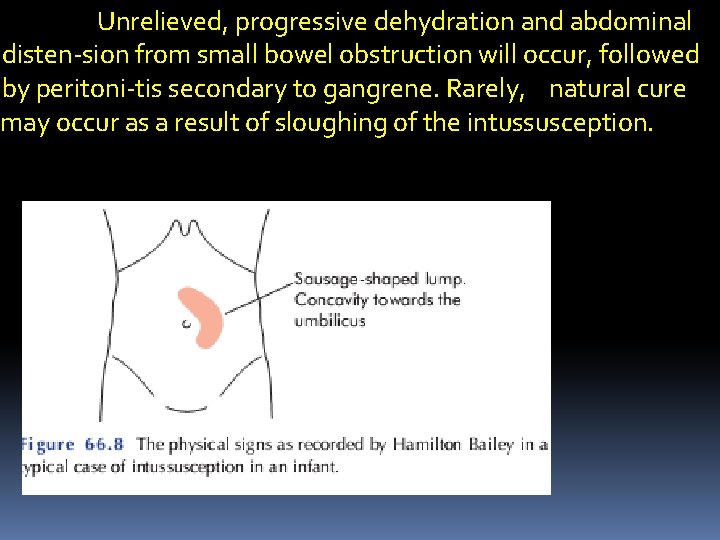
Unrelieved, progressive dehydration and abdominal disten-sion from small bowel obstruction will occur, followed by peritoni-tis secondary to gangrene. Rarely, natural cure may occur as a result of sloughing of the intussusception.

Imaging in intussusception A plain abdominal field usually reveals evidence of small or large bowel obstruction with an absent caecal gas shadow in ileocolic cases. A soft tissue opacity is often visible in children. A barium enema may be used to diagnose the presence of an ileocolic intussusception (the claw sign) (Fig. 66. 11) but does not demonstrate small bowel intussusception. An abdominal ultrasound scan has a high diagnostic sensitivity in children, demonstrating the typical doughnut appearance of concentric rings in transverse section. A computerised tomography (CT) scan is also.
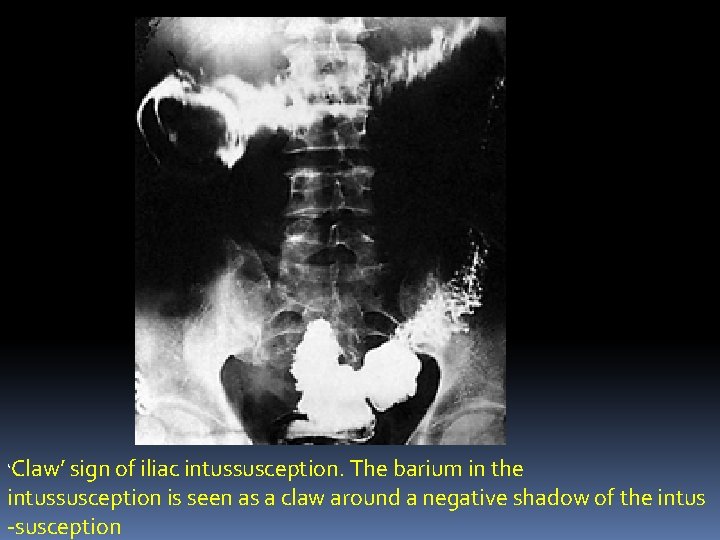
‘Claw’ sign of iliac intussusception. The barium in the intussusception is seen as a claw around a negative shadow of the intus -susception

Treatment of intussusception In the infant with ileocolic intussusception, after resuscitation with intravenous fluids, broad-spectrum antibiotics and naso-gastric drainage, non-operative reduction can be attempted using an air or barium enema Successful reduction can only be accepted if there is free reflux of air or bar-ium into the small bowel, together with resolution of symptom

Non-operative reduction is contraindi-cated if there are signs of peritonitis or perforation, there is a known pathological lead point or in the presence of profound shock. In experienced units, more than 70% of intussusceptions can be reduced non-operatively. Strangulated bowel and patho-logical lead points are unlikely to reduce. Surgery is required when radiological reduction has failed or is contraindicated.
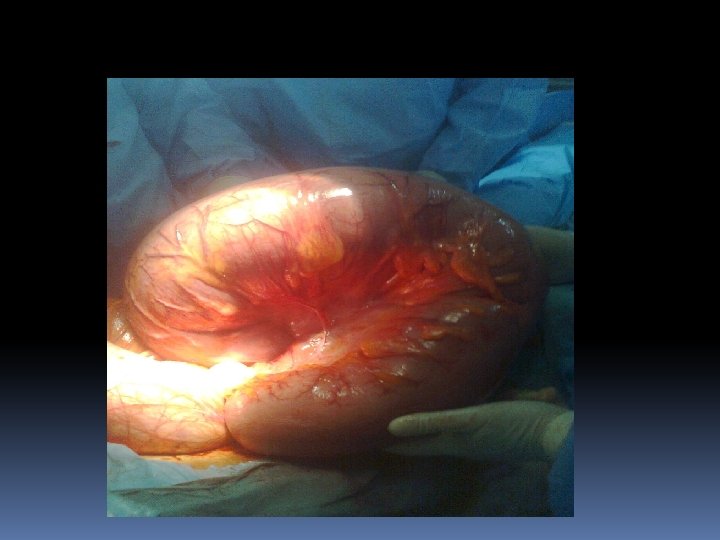
Volvulus A volvulus is a twisting or axial rotation of a portion ofbowelabout its mesentery. When complete it forms aclosedloop of obstruction with resultant ischaemia secondary to vascular occlu-sion. Volvuli may be primary or secondary. The primary form occurs secondary to congenital malrotation of the gut, abnormal mesen-teric attachments or congenital bands. Examples include volvu-lus neonatorum, caecal volvulus and sigmoid volvulus. secondary volvulus, which is the more common variety, is due to rotation of a piece of bowel around an acquired adhesion or stoma.

Volvulus ■ May involve the small intestine, caecum or sigmoid colon; neonatal midgut volvulus secondary to midgut malrotation is life-threatening ■ The commonest spontaneous type in adults is sigmoid ■ Sigmoid volvulus can be relieved by decompression per anum ■ Surgery is required to prevent or relieve ischaemia

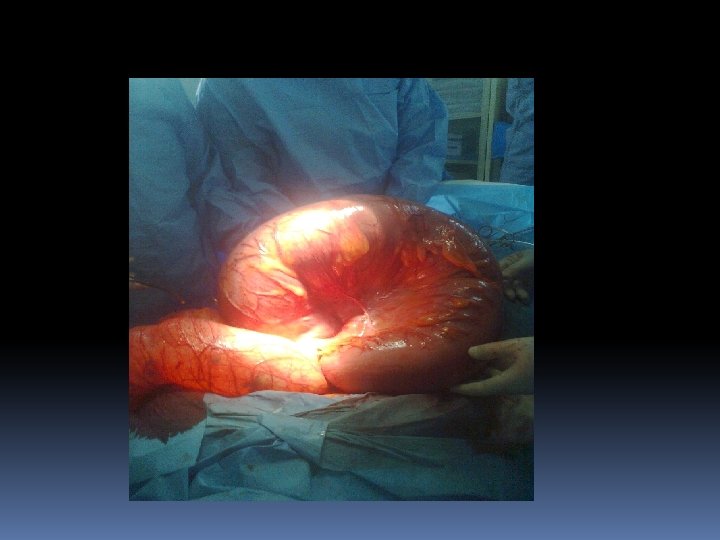
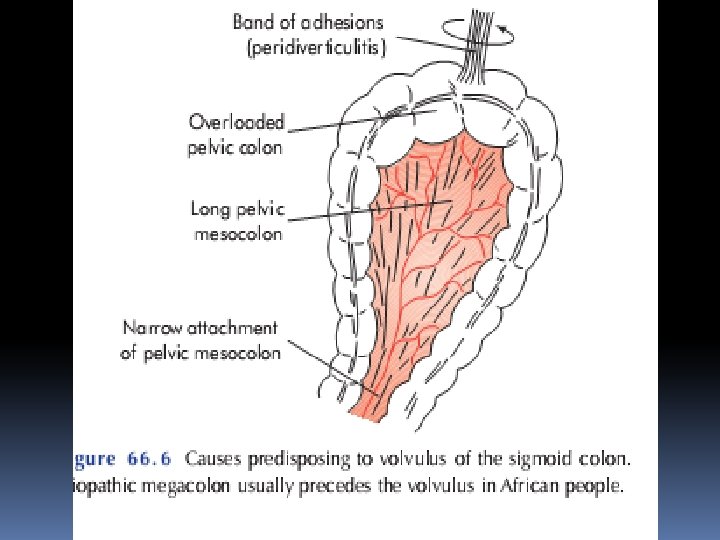
Volvulus neonatorum This occurs secondary to intestinal malrotation) and is potentially catastrophic. Sigmoid volvulus This is rare in Europe and the USA but more common in Eastern Europe and Africa. Indeed, it is the most comcause of large bowel obstruction in the indigenous black African pop-ulation. Rotation nearly always occurs in the anticlockwis direction.

Compound volvulus This is a rare condition also known as ileosigmoid knotting. Thesigmoid colon, resulting in gangrene of either or both segments of bowel. The patient present withb acute intestinal obstruction but distension is Plain radiography Reveals distended ileal loops in a distended sigmoid colon. At operation, decompression, resection and anastomosis are required.

Clinical features of volvulus Volvulus of the small intestine This may be primary or secondary and usually occurs in the lower ileum. It may occur spontaneously in African people, particularly following the consumption of a large volume of vegetable matter, whereas in the west it is usually secondary to adhesions passing to the parietes or female pelvic organs. Caecal volvulus This may occur as part of volvulus neonatorum or de novo and is usually a clockwise twist. It is more common in females and usually presents acutely with the classic features of obstruction. At first the obstruction may be partial, with the passage of flatus and faeces. In 25% of cases, examination may reveal a palpable tympanic swelling in the midline or left side of the abdomen.

Sigmoid volvulus The symptoms are of large bowel obstruction, which may initially be intermittent followed by the passage of large quantities of flatus and faeces. Presentation varies in severity and acuteness, with younger patients appearing to develop the more acute form. Abdominal distension is an early and progressive sign, which may be associated with hiccough and retching; vomiting occurs late. Constipation is absolute. In the elderly, a more chronic form maybe seen.

Imaging in volvulus In caecal volvulus, radiography may reveal a gas-filled ileum and occasionally a distended caecum. A barium enema may be used to confirm the diagnosis, with an absence of barium in the caecum and a bird beak deformity. In sigmoid volvulus, a plain radiograph shows massive colonic distension. The classic appearance is of a dilated loop of bowelrunning diagonally across the abdomen from right to left, with two fluid levels seen, one within each loop of bowel. In volvulus neonatorium, the abdominal radiograph shows avariable appearance. Initially, it may appear normal or show evi-dence of duodenal obstruction but, as the intestinal strangulation progresses, the abdomen becomes relatively gasless

Treatment of caecal volvulus At operation the volvulus should be reduced. Sometimes, this can only be achieved after decompression of the caecum using a needle. Further management consists of fixation of the caecum to the right iliac fossa (caecopexy) and/or a caecostomy. If the caecum is ischaemic or gangrenous, a right hemicolectomy should be performed.

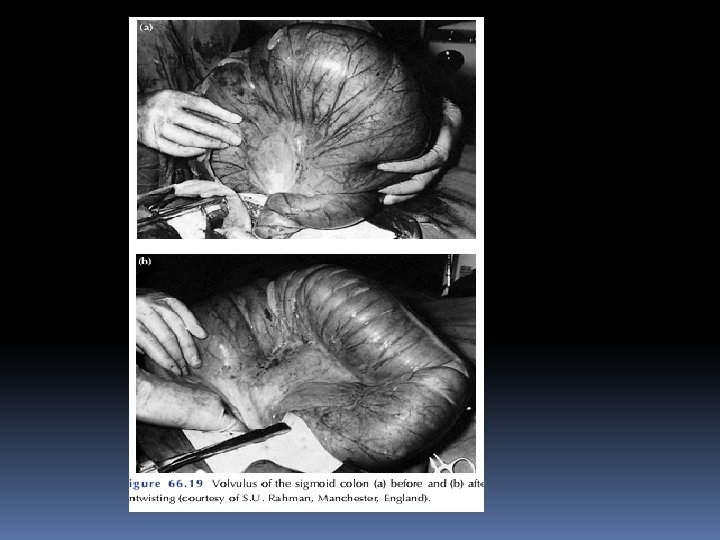
Treatment of sigmoid volvulus Flexible sigmoidoscopy or rigid sigmoidoscopy and insertion of a flatus tube should be carried out to allow deflation of the gut. Success, as long as ischaemic bowel is excluded, will provide temporary respite, allowing resuscitation and an elective proce-dure. Failure results in an early laparotomy, with untwisting of the loop and per anum decompression.

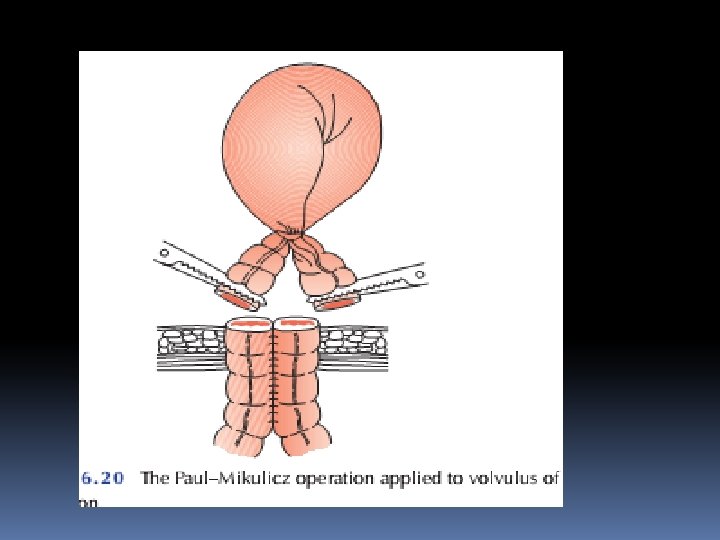
When the bowel is viable, fixation of the sigmoid colon to the posterior abdominal wall may be a safer manoeuvre in inexperienced hands. Resection is preferable if it can be achieved safely. A Paul–Mikulicz proce-dure is useful, particularly if there is suspicion of impending gangrene an alternative procedure is a sigmoid colec-tomy and, when anastomosis is considered unwise, a Hartmann’s procedure with subsequent reanastomosis can be carried out

TREATMENT OF ACUTE LARGE BOWEL OBSTRUCTION Large bowel obstruction is usually caused by an underlying carci-noma or occasionally diverticular disease, and presents in an acute or chronic form. The condition of pseudo-obstruction should always be considered and excluded by a limited contrast study or a CT air scan to confirm organic obstruction

Management of left-sided large bowel obstruction Contraindications to immediate resection include: ■ Inexperienced surgeon ■ Moribund patient ■ Advanced disease

CHRONIC LARGE BOWEL OBSTRUCTION The symptoms of chronic intestinal obstruction may arise from two sources – the cause and the subsequent obstruction. The causes of obstruction may be organic: • intramural – faecal impaction; • mural – colorectal cancer, diverticulitis, strictures (Crohn’s disease, ischaemia), anastomotic stenosis; • extramural – metastatic deposits, endometriosis; or functional: • Hirschsprung’s disease, idiopathic megacolon, pseudoobstruction

Principles of investigation of possible large bowel obstruction ■ In the presence of large bowel obstruction, a single-contrast water-soluble enema or CT should be undertaken to exclude a functional cause.

ADYNAMIC OBSTRUCTION Paralytic ileus This may be defined as a state in which there is failure of trans-mission of peristaltic waves secondary to neuromuscular failure [i. e. in the myenteric (Auerbach’s) and submucous (Meissner’s)plexuses]. The resultant stasis leads to accumulation of fluid and gas within the bowel, with associated distension, vomiting, absence of bowel sounds and absolute constipation.

Varieties The following varieties are recognised. • Postoperative: a degree of ileus usually occurs after any abdominal procedure and is self-limiting, with a variable duration of 24– 72 hours. Postoperative ileus may be prolonged in the presence of hypoproteinaemia or metabolic abnormality (see below). • Infection: intra-abdominal sepsis may give rise to localised or generalised ileus. Resultant adhesions may contribute a mechanical element to the initial neurogenic aetiology.

• Reflex ileus: this may occur following fractures of the spine or ribs, retroperitoneal haemorrhage or even the application of a plaster jacket. • Metabolic: uraemia and hypokalaemia are the most common contributory factors Clinical features Paralytic ileus takes on a clinical significance if, 72 hours after laparotomy: • there has been no return of bowel sounds auscultation; • there has been no passage of flatus. Abdominal distension becomes more marked and tympanitic. Pain is not a feature. In the absence of gastric aspiration, effort-less vomiting may occur. Radiologically, the abdomen shows gas-filled loops of intestine with multiple fluid levels.

Management • The primary cause must be removed. • Gastrointestinal distension must be relieved by decompression. • Close attention to fluid and electrolyte balance is essential. • There is no place for the routine use of peristaltic stimulants. Rarely, in resistant cases, medical therapy with an adrenergic blocking agent in association with cholinergic stimulation, e. g. neostigmine (the Catchpole regimen), may be used, provided that an intraperitoneal cause has been excluded. • If paralytic ileus is prolonged and threatens life, a laparotomy should be considered to exclude a hidden cause and facilitate bowel decompression

Pseudo-obstruction Factors associated with pseudo-obstruction Idiopathic ■ Metabolic Diabetes: intermittent porphyria Acute hypokalaemia Uraemia Myxodoema ■ Severe trauma (especially to the lumbar spine and pelvis) ■ Shock Burns Myocardial infarction Stroke Septicaemia

■ Retroperitoneal irritation Blood Urine Enzymes (pancreatitis) Tumour ■ Drugs Tricyclic antidepressants Phenothiazines Laxatives ■ Secondary gastrointestinal involvement Scleroderma Chagas’ disease

Acute mesenteric ischaemia The superior mesenteric vessels are the visceral vessels most likely to be affected by embolisation or thrombosis, with the former being most common. Occlusion at the origin of the superior mesenteric artery (SMA) is almost invariably the result of thrombosis, whereas emboli lodge at the origin of the middle colic artery. Inferior mesenteric involvement is usually clinically silent because of a better collateral circulation
Clinical features The most important clue to an early diagnosis of acute mesenteric ischaemia is the sudden onset of severe abdominal pain in a patient with atrial fibrillation or atherosclerosis. The pain is typi-cally central and out of all proportion to physical findings. Persistent vomiting and defaecation occur early, with the subse-quent passage of altered blood. Hypovolaemic shock rapidly ensues. Abdominal tenderness may be mild initially with rigidity being a late feature.

Investigation will usually reveal a profound neutrophil leuco-cytosis with an absence of gas in the thickened small intestine on abdominal radiographs. The presence of gas bubbles in the mesenteric veins is rare but pathognomonic. Treatment needs to full resuscitation, embolectomy or revascularisation of the SMA may be considered in earlyembolic cases. The majority of cases, however, are diagnosed late. In the young, all affected bowel should be resected, whereas in the elderly or infirm the situation may be deemed incurable. Anti-coagulation should be implemented early in the postopera-tive period. t
- Slides: 85