Business Review Day 2006 Introduction and Injectables Divestment

Business Review Day 2006 Introduction and Injectables Divestment Frank Condella Chief Executive 21 June 2006 NASDAQ: SKYE LSE: SKP www. skyepharma. com UK tel: +44 (0)207 491 1777 US tel: +1 (212) 753 5780

Legal statement This presentation does not constitute or form part of any offer for sale or subscription or solicitation of any offer to buy or subscribe for any securities in Skye. Pharma PLC nor shall it or any part of it form the basis of or be relied on in connection with any contract or commitment whatsoever. This presentation is being made only to and is directed at (a) persons who have professional experience in matters relating to investments who fall within Article 19(1) of the Financial Services and Markets Act 2000 (Financial Promotion) Order 2005 (the “Order”); or (b) high net worth entities and other persons to whom it may otherwise lawfully be communicated, falling within Article 49(1) of the Order (all such persons together being referred to as “relevant persons”). Any person who is not a relevant person should not rely on this presentation or any of its contents. This presentation includes certain forward-looking statements with respect to certain development projects, potential collaborative partnerships, results of operations and certain plans and objectives of Skye. Pharma including, in particular, the statements regarding potential sales revenues from Paxil CR, targeted sales revenues from other products both currently marketed and under development, possible launch dates for new products, and any revenue and profit guidance. By their very nature forward-looking statements involve risk and uncertainty that could cause actual results and developments to differ materially from those expressed or implied. The significant risks related to Skye. Pharma’s business which could cause our actual results and developments to differ materially from those forward-looking statements are discussed in Skye. Pharma’s SEC filings under the caption “Risk Factors”. Skye. Pharma reports under IFRS. Where US dollar equivalents of Skye. Pharma’s reported figures have been provided for convenience in this presentation, a fixed exchange rate of $1. 88 = £ 1 has been used throughout. These dollar equivalent numbers do not imply restatement from IFRS to US GAAP. This presentation was updated on 20 June 2006 2
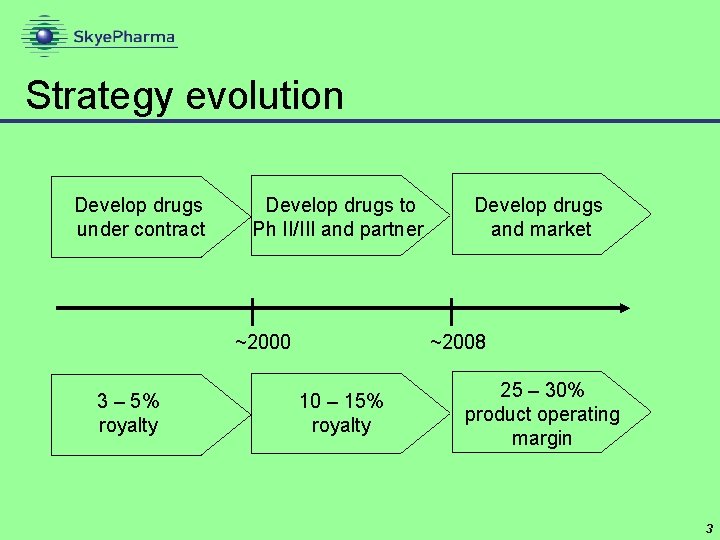
Strategy evolution Develop drugs under contract Develop drugs to Ph II/III and partner ~2000 3 – 5% royalty Develop drugs and market ~2008 10 – 15% royalty 25 – 30% product operating margin 3

Strategic plan 2006 • new leadership • divest injectables unit • continue Phase III for Flutiform™ and outlicense this year • focus on core oral/inhalation unit and expand pipeline • improve operational efficiency • longer term aim to market own products in selected therapeutic area 4
Two main business units London (HQ) ORAL & INHALATION Basel Lyon San Diego INJECTABLE 5

Divest injectables unit • two complementary injectable delivery technologies • Depo. Foam™ • Biospheres™ • two approved products • Depo. Cyt (cancer) • Depo. Dur (post-operative pain) • strong pipeline • Depo. Bupivacaine™ (post-operative pain; Ph II completed) • Biologics (SR injectable formulations of therapeutic proteins; pre-clinical) • stand-alone operation with R&D and manufacture in San Diego • unit requires significant investment in R&D and capex 6

Depo. Bupivacaine ™ ™ Depo. Bupivacaine has blockbuster potential 7

™ Depo. Bupivacaine - issues with bupivacaine • bupivacaine is the “gold standard” local anaesthetic but has the drawback of a short duration of action • all patients require post-operative analgesia after surgery • local anaesthetics are used for: • • pre-operative anaesthesia post-operative analgesia • bupivacaine most commonly used local anaesthetic for significant surgical procedures - considered the “gold standard” • longest-lasting bupivacaine duration of effect currently 6 -10 hours *Source: 2004 Verispan Data 8

™ Depo. Bupivacaine - extended pain relief • Depo. Bupivacaine™ offers prolonged pain relief through a variety of routes of administration • innovation in local/regional analgesia provides sustained duration of analgesia (target: >48 hours) • • sustained-release bupivacaine via a single-dose administration favored analgesic matched with sustained-release Depo. Foam™ delivery technology • previously approved in both US and EU (Depo. Dur and Depo. Cyt ) • 3 potential routes of administration • wound infiltration - soft tissue incisional procedures (eg hernia repair, hysterectomies, etc) • • nerve blocks - thoracic, upper and lower limb surgeries epidural - significant orthopedic procedures (eg hip and knee replacement, etc) 9

™ Depo. Bupivacaine - major product benefits • simplified post-operative pain management after various surgical procedures • helps address patient pain for 2 -4 days after surgery • significant reduction in need for supplemental opioid pain medications • fewer narcotic side effects • ease of use • no need for catheters, pumps, or devices • potential pharmacoeconomic benefits • • • easier patient management earlier discharge faster rehabilitation 10

™ Depo. Bupivacaine - $1 bn market potential • • potential $1 billion opportunity exists for Depo. Bupivacaine™ • • significant established market, expressed clinical and commercial needs • safety comparable to bupivacaine solution has been shown at 2 -6 x free bupivacaine dose • • production scale-up approach already cleared by regulatory agencies >40 mn annual US in- & out-patient surgical procedures (hospitals and outpatient surgical centers) - all patients require post-operative analgesia safety and potential duration of action out to 96 hours currently established by PK levels one possible competitor • • Durect – now in Ph III trials due to start 2 H ‘ 06 16

Biologics Substantial potential for Biologics 17
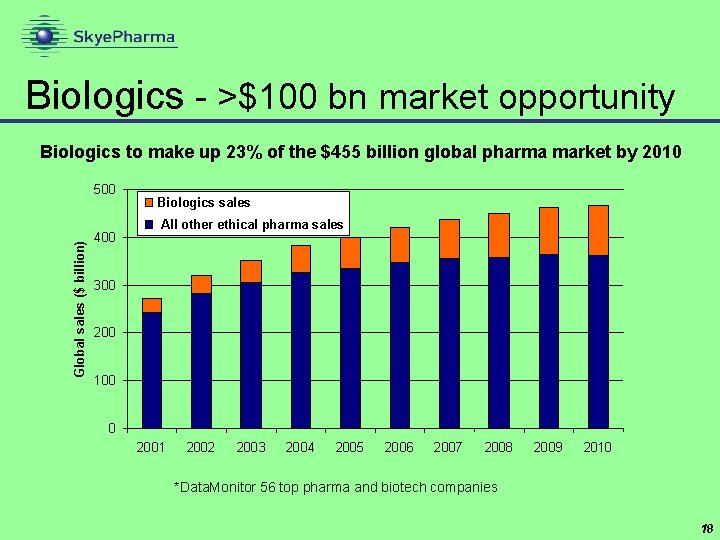
Biologics - >$100 bn market opportunity Biologics to make up 23% of the $455 billion global pharma market by 2010 Global sales ($ billion) 500 400 Biologics sales All other ethical pharma sales 300 200 100 0 2001 2002 2003 2004 2005 2006 2007 2008 2009 2010 *Data. Monitor 56 top pharma and biotech companies 18
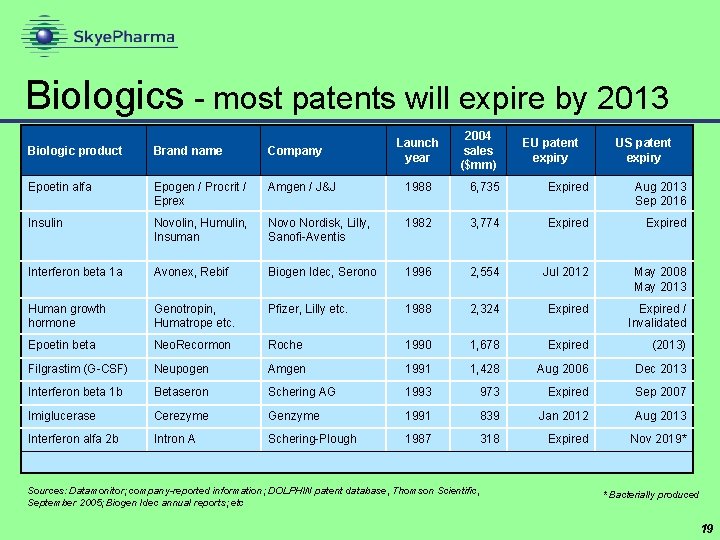
Biologics - most patents will expire by 2013 Launch year 2004 sales ($mm) EU patent expiry US patent expiry Biologic product Brand name Company Epoetin alfa Epogen / Procrit / Eprex Amgen / J&J 1988 6, 735 Expired Aug 2013 Sep 2016 Insulin Novolin, Humulin, Insuman Novo Nordisk, Lilly, Sanofi-Aventis 1982 3, 774 Expired Interferon beta 1 a Avonex, Rebif Biogen Idec, Serono 1996 2, 554 Jul 2012 May 2008 May 2013 Human growth hormone Genotropin, Humatrope etc. Pfizer, Lilly etc. 1988 2, 324 Expired / Invalidated Epoetin beta Neo. Recormon Roche 1990 1, 678 Expired (2013) Filgrastim (G-CSF) Neupogen Amgen 1991 1, 428 Aug 2006 Dec 2013 Interferon beta 1 b Betaseron Schering AG 1993 973 Expired Sep 2007 Imiglucerase Cerezyme Genzyme 1991 839 Jan 2012 Aug 2013 Interferon alfa 2 b Intron A Schering-Plough 1987 318 Expired Nov 2019* Sources: Datamonitor; company-reported information; DOLPHIN patent database, Thomson Scientific, September 2005; Biogen Idec annual reports; etc * Bacterially produced 19

Biologics - limited ways to exploit patent expiry • biogeneric / biosimilar • mimic originator product • adulterate or enhance protein • modify protein to increase circulation half-life (eg PEGylation) • enhance delivery of protein • maintain originator product but extend duration of action via sustained-release delivery system (eg Depo. Foam™ or Biosphere™) 20

Biologics – not like small molecule generics • the biosimilars market presents higher barriers to entry than for small molecule generics - including cost, risk and time needed for clinical development High up-front investment Clinical trial requirements Specialist marketing New challenges representing increasing barriers to biogeneric entry Biotechnology expertise Pharmacovigilance studies Regulatory experience Legal / patent expertise Controlling manufacturing cost Skills required in a traditional generic business model 21

Biologics - portfolio of enhanced biosimilars • Phase I • h. GH-Biosphere • feasibility studies complete: • • Depo-IFN-α Depo-EPO • feasibility studies in progress: • • Depo-G-CSF Depo-h. GH Depo-IFN-β a number of proprietary proteins and peptides for third parties 22

Benefits of divesting injectable unit • improve Skye. Pharma P&L • reduce cash requirements of ongoing business • strengthen balance sheet • permits Skye. Pharma to explore strategic options • focus management on oral/inhalation unit 23
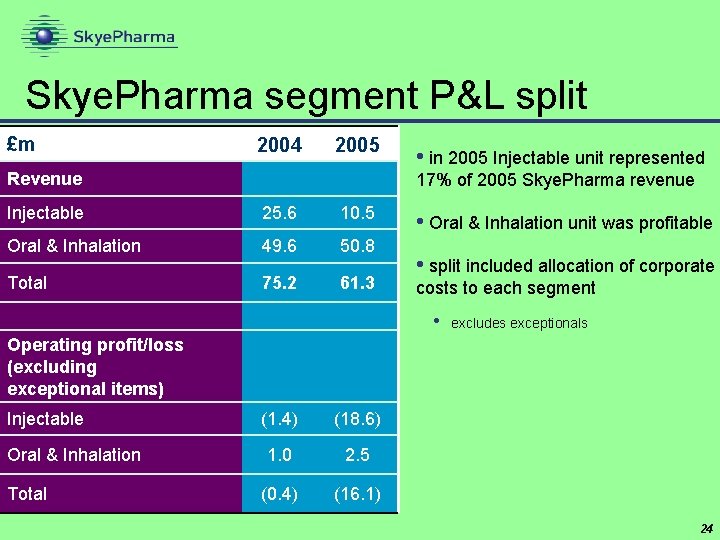
Skye. Pharma segment P&L split £m 2004 2005 Revenue • in 2005 Injectable unit represented 17% of 2005 Skye. Pharma revenue Injectable 25. 6 10. 5 Oral & Inhalation 49. 6 50. 8 Total 75. 2 61. 3 • Oral & Inhalation unit was profitable • split included allocation of corporate costs to each segment • excludes exceptionals Operating profit/loss (excluding exceptional items) Injectable Oral & Inhalation Total (1. 4) (18. 6) 1. 0 2. 5 (0. 4) (16. 1) 24

Sale of injectables unit - status • due diligence materials prepared • UBS selected as investment bank • several expressions of interest already received • management presentations and due diligence ongoing 25
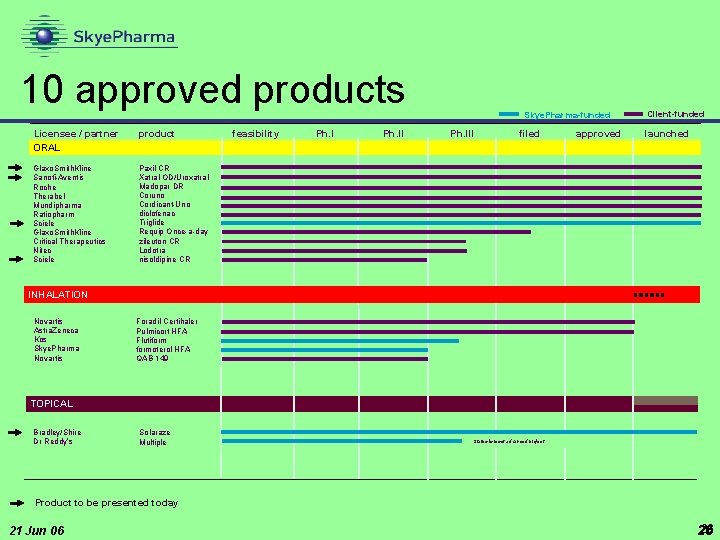
10 approved products Licensee / partner ORAL product Glaxo. Smith. Kline Sanofi-Aventis Roche Therabel Mundipharma Ratiopharm Sciele Glaxo. Smith. Kline Critical Therapeutics Nitec Sciele Paxil CR Xatral OD/Uroxatral Madopar DR Coruno Cordicant-Uno diclofenac Triglide Requip Once-a-day zileuton CR Lodotra nisoldipine CR feasibility Ph. II Skye. Pharma-funded Ph. III filed approved Client-funded launched INHALATION Novartis Astra. Zeneca Kos Skye. Pharma Novartis Foradil Certihaler Pulmicort HFA Flutiformoterol HFA QAB 149 TOPICAL Bradley/Shire Dr Reddy’s Solaraze Multiple Status is most advanced project Product to be presented today 21 Jun 06 26 26

Business Review Day 2006 Marketed products Dr Werner Enz VP Commercial Patrick Fourteau CEO, Sciele NASDAQ: SKYE LSE: SKP www. skyepharma. com UK tel: +44 (0)207 491 1777 US tel: +1 (212) 753 5780 27

Key marketed products Paxil CR® (Glaxo. Smith. Kline) Xatral® OD / Uroxatral® (Sanofi-Aventis) Solaraze® (Bradley; Shire) Triglide™ & nisoldipine CR will be covered by Patrick Fourteau, CEO 21 Jun 06 28 28

™ Paxil CR Glaxo. Smith. Kline • Geomatrix™ version of GSK’s SSRI Paxil® (paroxetine) • FDA approved for the treatment of depression, social anxiety disorder, premenstrual dysphoric disorder, and panic disorder • all SSRI antidepressants take about a month to take effect – but sideeffects (notably nausea) start immediately • nausea major reason for therapy switching and non-compliance • Paxil CR™ designed to reduce nausea • active passes through stomach unchanged and is released in lower intestine • clinically proven benefits of Paxil CR™ over Paxil® IR: • reduced nausea • improved compliance 21 Jun 06 • improved efficacy 29 29

™ Paxil CR Glaxo. Smith. Kline • FDA approval Feb ‘ 99 - but GSK did not launch until Apr ’ 02 • first generic competition for Paxil® (old IR version) Sep ’ 03 • Paxil CR™ clinically differentiated and not substitutable, so generic paroxetine had minimal impact on Paxil CR™ Rx share 21 Jun 06 31 31
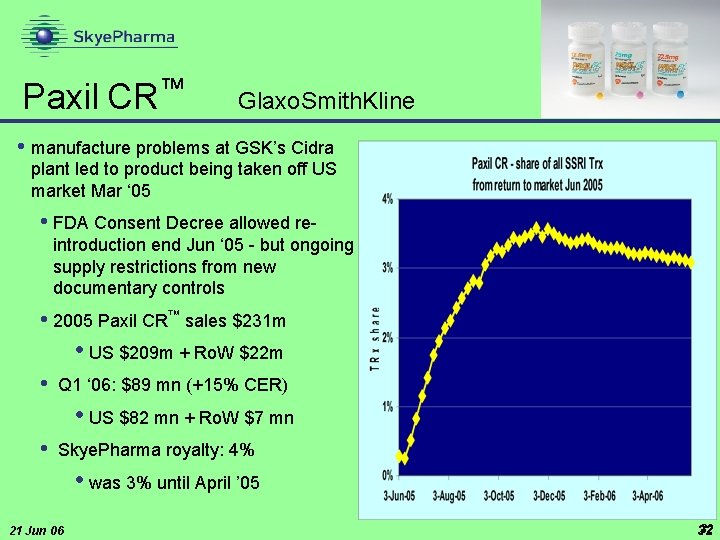
™ Paxil CR Glaxo. Smith. Kline • manufacture problems at GSK’s Cidra plant led to product being taken off US market Mar ‘ 05 • FDA Consent Decree allowed reintroduction end Jun ‘ 05 - but ongoing supply restrictions from new documentary controls • 2005 Paxil CR™ sales $231 m • US $209 m + Ro. W $22 m • Q 1 ‘ 06: $89 mn (+15% CER) • US $82 mn + Ro. W $7 mn • Skye. Pharma royalty: 4% • was 3% until April ’ 05 21 Jun 06 32 32

™ Paxil CR Glaxo. Smith. Kline • Mylan filed Para IV certification for “paroxetine hydrochloride controlled release” in late 2005 • Mylan is not challenging GSK’s paroxetine hydrochloride hemihydrate crystalline form patent (expires Jun ’ 07) • GSK has decided not to invoke 30 month stay of approval • earliest possible generic entry: July ’ 07 • no certainty that Mylan will be able to match PK profile of Paxil CR™ • Skye. Pharma not aware of any other generic versions pending • Skye. Pharma has other patents on technologies used in Paxil CR™ 21 Jun 06 33 33

™ Paxil CR Glaxo. Smith. Kline Conclusions • sales >$700 mn before withdrawal • sales slowly recuperating - but unlikely to reach previous level • sales still growing Q over Q • generic paroxetine has had minimal impact • earliest generic CR entry could be Q 3 ‘ 07 21 Jun 06 35 35

Xatral® OD/Uroxatral® Sanofi-Aventis • once-daily Geomatrix™ formulation of alfuzosin • alpha-blocker for treating the urinary symptoms of benign prostatic hypertrophy (BPH) • in BPH slow growth in size of prostate gradually constricts the urethra • patient unable to empty bladder despite frequent urination • urination problems normal reason for patient to consult doctor BPH diagnosis • BPH is a very common condition in males from middle age onwards • 20% of males aged 50 -60, >40% of males aged >70 • pharmaceutical treatments for BPH: • 5 -alpha reductase inhibitors • slowly shrink prostate • little or no short-term benefit on urination • alpha-blockers for symptomatic relief • relax smooth muscle, open urethra and allow the bladder to empty 21 Jun 06 36 36

Xatral® OD/Uroxatral® Sanofi-Aventis • alfuzosin has two USPs: • highly uroselective • avoids postural hypotension seen with older alpha-blockers • low cardiovascular risk • can be used safely by patients taking PDE 5 inhibitors • minimal impact on sexual function • contrast with tamsulosin (retrograde ejaculation affects ~20% of patients) • late-stage BPH results in total blockage of urethra - Acute Urinary Retention (AUR) • severe complication requiring immediate surgery for insertion of catheter • Xatral® OD now approved in Europe and over 40 other markets for AUR • adjuvant (after surgery) and prophylaxis • indication no longer being pursued in USA 21 Jun 06 37 37
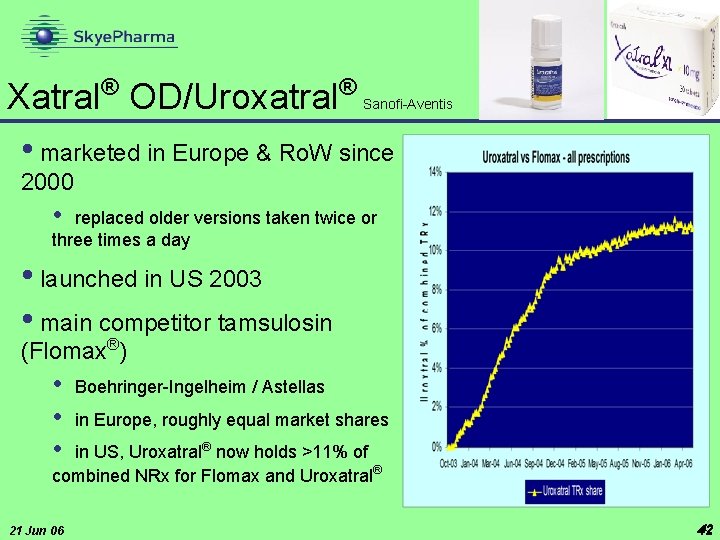
® ® Xatral OD/Uroxatral • marketed in Europe & Ro. W since Sanofi-Aventis 2000 • replaced older versions taken twice or three times a day • launched in US 2003 • main competitor tamsulosin (Flomax®) • • • Boehringer-Ingelheim / Astellas in Europe, roughly equal market shares in US, Uroxatral® now holds >11% of combined NRx for Flomax and Uroxatral® 21 Jun 06 42 42
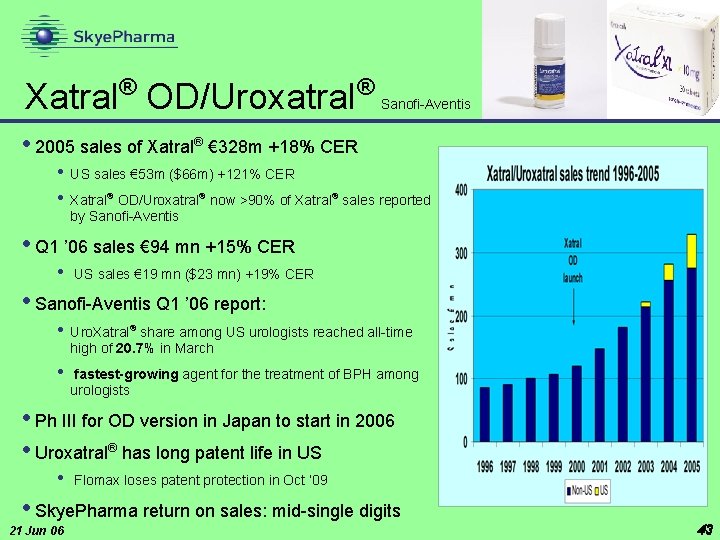
® Xatral OD/Uroxatral ® Sanofi-Aventis • 2005 sales of Xatral® € 328 m +18% CER • US sales € 53 m ($66 m) +121% CER • Xatral® OD/Uroxatral® now >90% of Xatral® sales reported by Sanofi-Aventis • Q 1 ’ 06 sales € 94 mn +15% CER • US sales € 19 mn ($23 mn) +19% CER • Sanofi-Aventis Q 1 ’ 06 report: • Uro. Xatral® share among US urologists reached all-time high of 20. 7% in March • fastest-growing agent for the treatment of BPH among urologists • Ph III for OD version in Japan to start in 2006 • Uroxatral® has long patent life in US • Flomax loses patent protection in Oct ‘ 09 • Skye. Pharma return on sales: mid-single digits 21 Jun 06 43 43

® Xatral OD/Uroxatral ® Sanofi-Aventis Conclusions • ongoing successful sales effort through Sanofi. Aventis • excellent sales development - particularly in the USA through covering urologists and high potential GPs • continued growth in Europe 45

Solaraze ® Bradley / Shire • topical gel formulation of diclofenac for actinic keratosis (AK) • AK is early form of squamous cell carcinoma • caused by over-exposure to sunlight • especially common in fair-skinned individuals • typically presents in middle age • increasingly common • AK lesions usually found in clusters on scalp, forehead or backs of hands • lesions gradually progress to metastatic stage so need to be treated • alternative treatments for AK are effective… • cryosurgery (liquid nitrogen) • topical 5 -FU • photodynamic therapy • …but painful and disfiguring • scarring and reddening very visible and cosmetically unacceptable 21 Jun 06 46 46
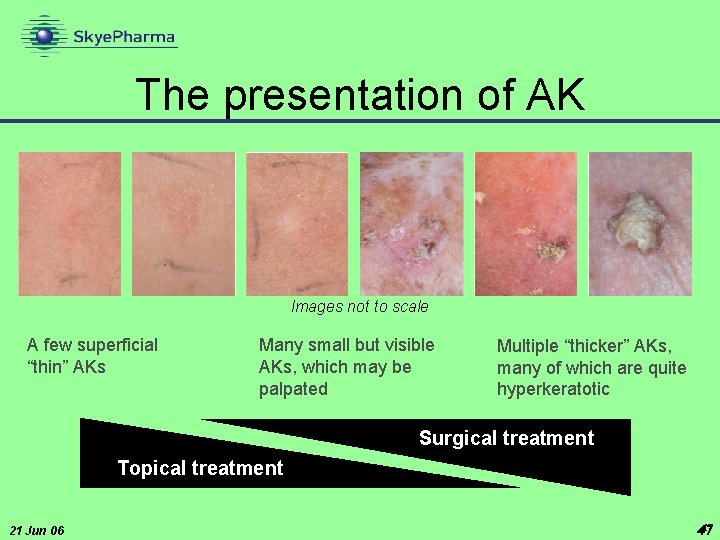
The presentation of AK Images not to scale A few superficial “thin” AKs Many small but visible AKs, which may be palpated Multiple “thicker” AKs, many of which are quite hyperkeratotic Surgical treatment Topical treatment 21 Jun 06 47 47

Solaraze ® Bradley / Shire • Solaraze® is a topical formulation of 3% diclofenac in a proprietary hyaluronic acid (HA) gel formulation • mode of action unclear but HA formulation retains high concentration of diclofenac in upper skin layers • other gel formulations of diclofenac ineffective in AK • applied topically twice a day for 6090 days • particularly suitable for treating clusters of lesions • generally well-tolerated and invisible 21 Jun 06 48 48

Solaraze ® Bradley / Shire • 2005 global in-market sales $27. 5 mn (+78%) • Bradley (North America) • Doak Dermatologics unit has ~135 reps • 2005 sales $10 m in 9 M ‘ 05 (latest data reported by Bradley) • • FY ‘ 05 est $15 m vs $6 m in 2004 (for 4 months) • Bradley estimate Solaraze has 15% of the US market for topical AK treatments • Shire (Europe / Australasia) • 2005 sales $12. 5 m (+32%) • Q 1 ’ 06 $3. 3 mn (+38%) • Australian regulatory review ongoing • Skye. Pharma royalty: double digit 21 Jun 06 51 51

® Solaraze - raising awareness Joint Doak / Skin Cancer Foundation campaign (Mar-Sep ’ 06): “We’re Going On The Road” To raise US public awareness about skin cancer and the importance of early detection and treatment The Goal: http: //www. skincancerscreeningtour. com/ Encourage people to see their local dermatologists for testing and treatment Campaign expected to contact 20 mn people in 2006 Guidelines for the Management of ACTINIC KERATOSES Developed by the Guideline Subcommittee May 2006 • Diclofenac in hyaluronic acid gel… Adverse effects were skin related and mild to moderate in severity 52

Solaraze ® Bradley / Shire Conclusions • good sales growth in a market driven by epidemiology, awareness and prevention • both partners actively promote product and invest in campaigns to raise profile of disease and treatment options • activities ongoing to expand territories 21 Jun 06 53 53

™ Triglide Sciele (formerly First Horizon) • Skye. Pharma oral formulation of fenofibrate • fenofibrate used to treat lipid disorders • lowers LDL and TG but raises HDL • potential synergy with statins • uses Insoluble Drug Delivery – Microparticle (IDD-P™) technology • can be taken with or without food • launched in US July ’ 05 by Sciele (formerly First Horizon) • Skye. Pharma receives up to $50 m in milestones and 25% share of sales • Skye. Pharma manufactures in Lyon (and bears COGS) 54

nisoldipine CR ® • Geomatrix™ technology applied to nisoldipine (Sular®) • calcium channel blocker for treating hypertension • 3 goals: • • • improve bioavailability reduce therapeutic dose optimize side-effect profile • target filing: mid-07 • target US launch: 2008 • partnered with Sciele Pharma (formerly First Horizon) • mid-single digit royalty + option to manufacture for Sciele 56

Introducing Patrick Fourteau CEO Sciele Pharma (the new name for First Horizon Pharmaceuticals) Alpharetta, GA, USA 57

58

SCIELE PHARMA – WHO WE ARE • Revenue of $216 million for full-year 2005 • Specialty pharmaceuticals with 70% of sales derived from cardiovascular/ metabolic • Sales force of 525 representatives • Unique business model – pay for performance 59

Importance • Fast growing market • Large market – over $1 billion • One dominant player • Synergy within our cardiovascular/ metabolic portfolio

PRODUCT PORTFOLIO SYNERGY Products address multiple risk factors in cardiovascular and metabolic diseases • Hypertension • Hypercholesterolemia • High triglycerides • Type 2 Diabetes 61

Strategy • Managed care – cost savings • 450 sales representatives/11 calls per day • Calling on primary care physicians • Savings card
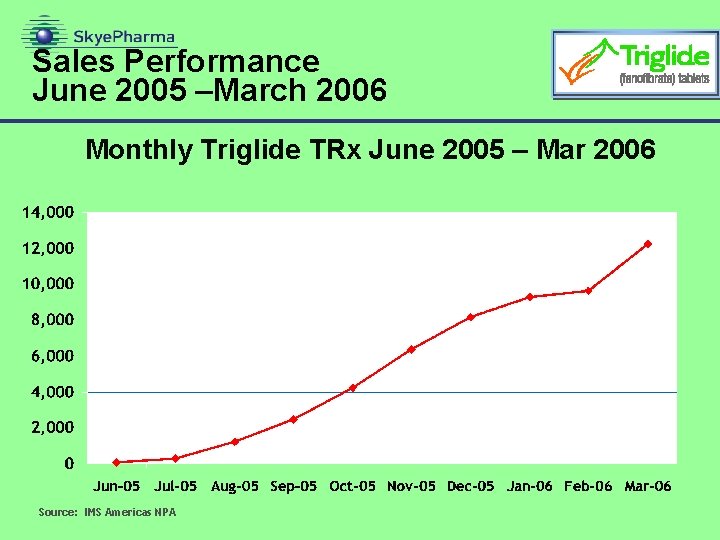
Sales Performance June 2005 –March 2006 Monthly Triglide TRx June 2005 – Mar 2006 Source: IMS Americas NPA
Formulation development project • Sular is Sciele's flagship product • New CR formulation is a priority for the company • Sular has achieved excellent growth • New Rx’s increased 32% in 1 Q 06 versus 1 Q 05 • Total Rx’s increased 17% in 1 Q 06 versus 1 Q 05

Development timeline • Initiate pivotal clinical trial in second half 2006 • FDA filing in first half of 2007 • FDA approval expected in 2008
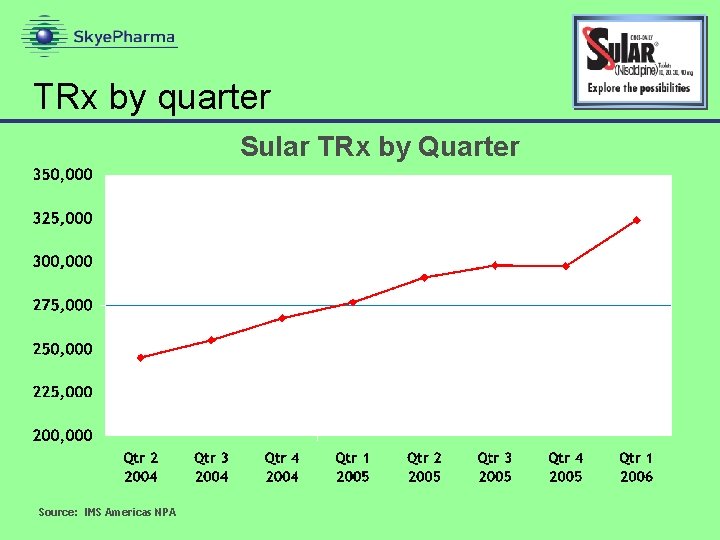
TRx by quarter Sular TRx by Quarter Source: IMS Americas NPA

NASDAQ Symbol: SCRX 67

Business Review Day 2006 Near-term pipeline and pre-approval products Dr Francesco Patalano Managing Director Europe Dr Geraldine Venthoye Head of Inhalation Business Unit 21 June 2006 NASDAQ: SKYE LSE: SKP www. skyepharma. com UK tel: +44 (0)207 491 1777 US tel: +1 (212) 753 5780

Key near-term pipeline products Oral / inhalation: Pulmicort® HFA Requip Once-a-day zileuton CR Lodotra™ Foradil® Certihaler™ + nisoldipine CR – already presented Flutiform ™ – to be presented by Ken Cunningham 69

Pulmicort® HFA-MDI Astra. Zeneca (for Europe) • Pulmicort® (budesonide) in CFC-free MDI • inhaled steroid for asthma • will allow AZN to withdraw CFC-MDI version (Montreal protocol requirement) • Skye. Pharma developed formulation and conducted clinical development for AZN • development started Jan ′ 02 • filed on country-by-country basis in Europe mid ′ 05 • first European approval (Finland) Feb ′ 06 • development experience underwrites Flutiform™ development • double digit royalty 70

Requip Once-a-day Glaxo. Smith. Kline • once-daily oral dosage formulation of Requip® (ropinirole) • dopamine agonist for Parkinson’s disease • dopamine agonists are used as as first-line therapy for Parkinson’s disease • Requip® is dosed three times a day • once-daily version provides patient convenience in dosage titration • filed by GSK Dec ′ 05 • filed in Europe and New Zealand • US filing to be resubmitted Q 4 ′ 06 • GSK’s 2005 sales of Requip® $284 m (+34%) • approx two-thirds of sales currently for RLS indication 71

zileuton CR Critical Therapeutics • highly potent oral anti-inflammatory • 5 -lipoxygenase inhibitor • blocks both branches of inflammatory cascade • primary indication: severe asthma • reduce or eliminate need for oral steroids • current version (Zyflo ) has to be taken four times a day • zileuton CR is twice daily • Ph III trial of zileuton CR completed - filing expected Q 3’ 06 • high single digit royalty rate + option to manufacture • peak sales estimates in region of $150 mn 72
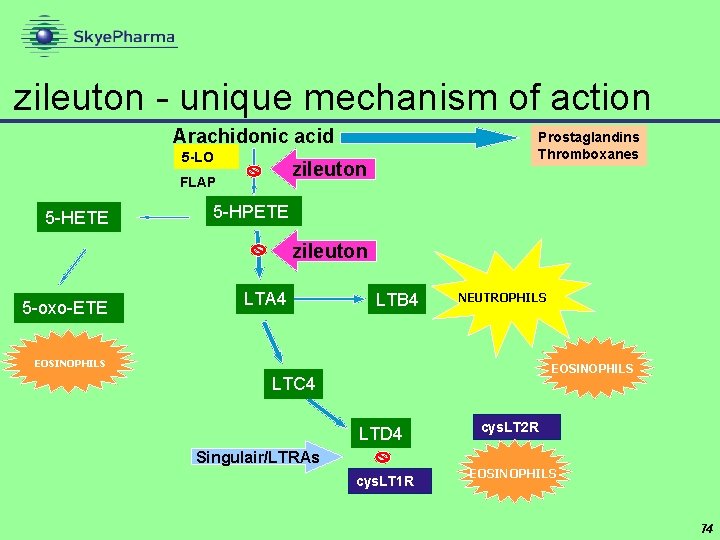
zileuton - unique mechanism of action Arachidonic acid 5 -LO zileuton FLAP 5 -HETE Prostaglandins Thromboxanes 5 -HPETE zileuton 5 -oxo-ETE LTA 4 LTB 4 NEUTROPHILS EOSINOPHILS LTC 4 LTD 4 Singulair/LTRAs cys. LT 1 R cys. LT 2 R EOSINOPHILS 74
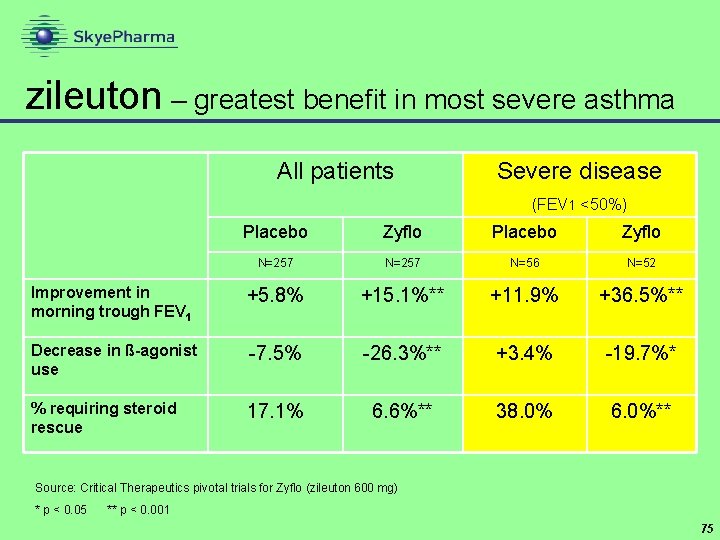
zileuton – greatest benefit in most severe asthma All patients Severe disease (FEV 1 <50%) Placebo Zyflo N=257 N=56 N=52 Improvement in morning trough FEV 1 +5. 8% +15. 1%** +11. 9% +36. 5%** Decrease in ß-agonist use -7. 5% -26. 3%** +3. 4% -19. 7%* % requiring steroid rescue 17. 1% 6. 6%** 38. 0% 6. 0%** Source: Critical Therapeutics pivotal trials for Zyflo (zileuton 600 mg) * p < 0. 05 ** p < 0. 001 75

™ Lodotra Nitec • controlled release formulation of oral anti-inflammatory • active not yet disclosed • indication: rheumatoid arthritis • Ph III recently completed • data not yet available • Nitec has licensed rights for Germany and Austria to Merck KGa. A • negotiations for other territories ongoing • mid-single digit royalty and manufacture for Nitec 77
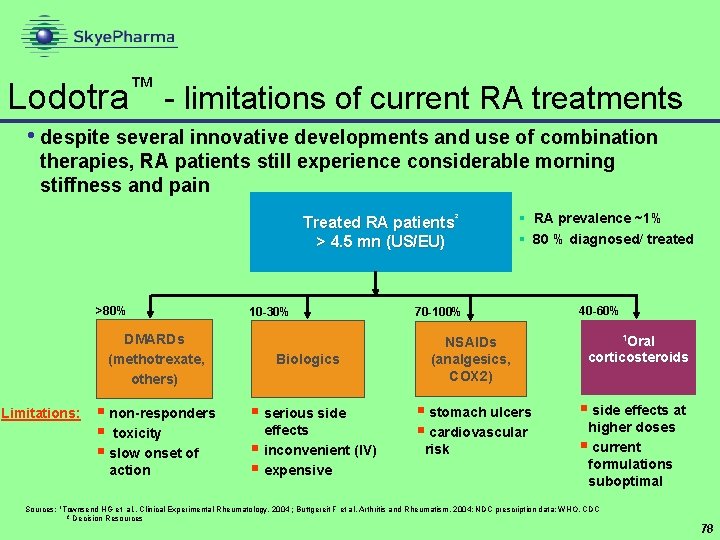
Lodotra - limitations of current RA treatments ™ • despite several innovative developments and use of combination therapies, RA patients still experience considerable morning stiffness and pain Treated RA patients² > 4. 5 mn (US/EU) >80% 10 -30% DMARDs (methotrexate, others) Limitations: § non-responders § toxicity § slow onset of action Biologics § serious side § § effects inconvenient (IV) expensive § RA prevalence ~1% § 80 % diagnosed/ treated 70 -100% NSAIDs (analgesics, COX 2) § stomach ulcers § cardiovascular risk 40 -60% 1 Oral corticosteroids § side effects at higher doses § current formulations suboptimal Sources: 1 Townsend HG et al. , Clinical Experimental Rheumatology, 2004 ; Buttgereit F et al, Arthritis and Rheumatism, 2004; NDC prescription data; WHO, CDC ² Decision Resources 78
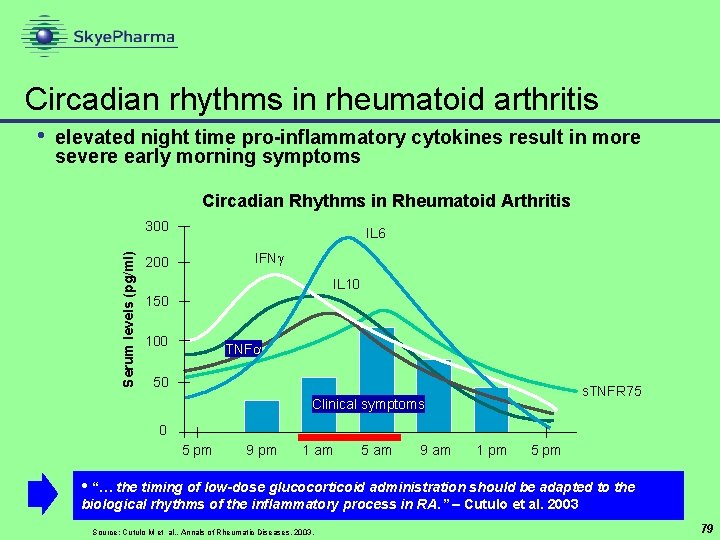
Circadian rhythms in rheumatoid arthritis elevated night time pro-inflammatory cytokines result in more severe early morning symptoms Circadian Rhythms in Rheumatoid Arthritis 300 Serum levels (pg/ml) • IL 6 IFN 200 IL 10 150 100 TNF 50 s. TNFR 75 Clinical symptoms 0 5 pm 9 pm 1 am 5 am 9 am 1 pm 5 pm • “… the timing of low-dose glucocorticoid administration should be adapted to the biological rhythms of the inflammatory process in RA. ” – Cutulo et al. 2003 Source: Cutulo M et al, . Annals of Rheumatic Diseases, 2003. 79
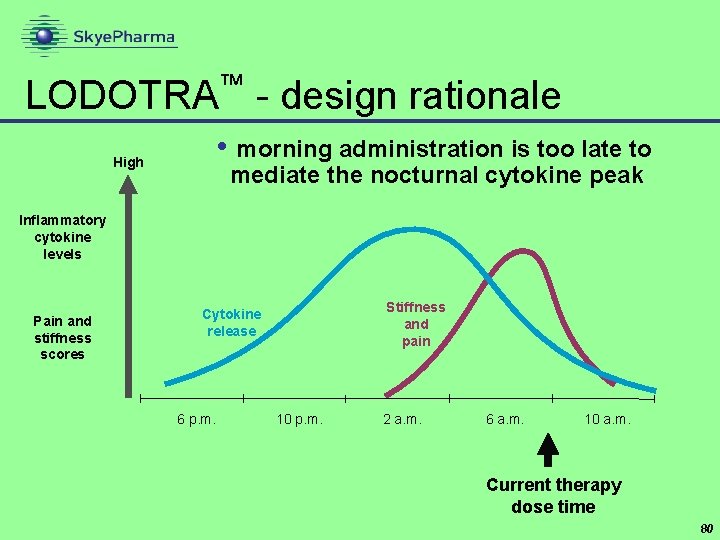
™ LODOTRA - design rationale • morning administration is too late to High mediate the nocturnal cytokine peak Inflammatory cytokine levels Pain and stiffness scores Stiffness and pain Cytokine release 6 p. m. 10 p. m. 2 a. m. 6 a. m. 10 a. m. Current therapy dose time 80
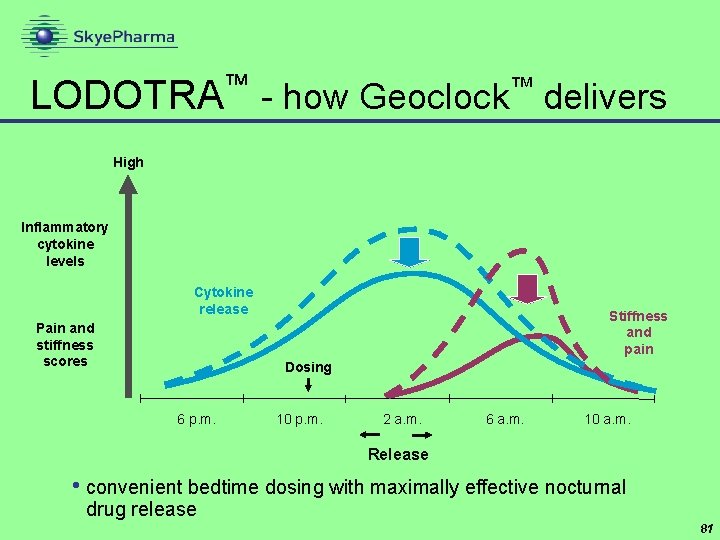
™ LODOTRA - how Geoclock delivers ™ High Inflammatory cytokine levels Cytokine release Pain and stiffness scores Stiffness and pain Dosing 6 p. m. 10 p. m. 2 a. m. 6 a. m. 10 a. m. Release • convenient bedtime dosing with maximally effective nocturnal drug release 81

Foradil Certihaler Novartis / Schering-Plough • formoterol (fast-onset, long-acting bronchodilator) for asthma / COPD • uses Skye. Pharma’s Skye. Haler™ multi-dose dry-powder inhaler (DPI) • Skye. Pharma developed both device and formulation • formulation keeps powder dry, ensures accurate consistent dose • Schering-Plough to market in US market, Novartis elsewhere • 2005 sales of Foradil in single-dose DPI: $332 mn • includes Novartis sales to Schering-Plough but no US end-user sales • mid-single digit royalty + manufacturing return 82

Foradil Certihaler ™ Novartis / Schering-Plough • Foradil Certihaler™ now approved in 24 markets (Europe, Mid-East, S America…) • launched in Germany Sep ′ 05 and Switzerland Oct ′ 05 but batches withdrawn by Novartis from both markets Jan ′ 06 • a few patients mishandled the device and received an incorrect dose • Skye. Pharma working with Novartis to investigate cause and correct • FDA “approvable” letter Apr ′ 06 – device modifications will be required for final approval 83
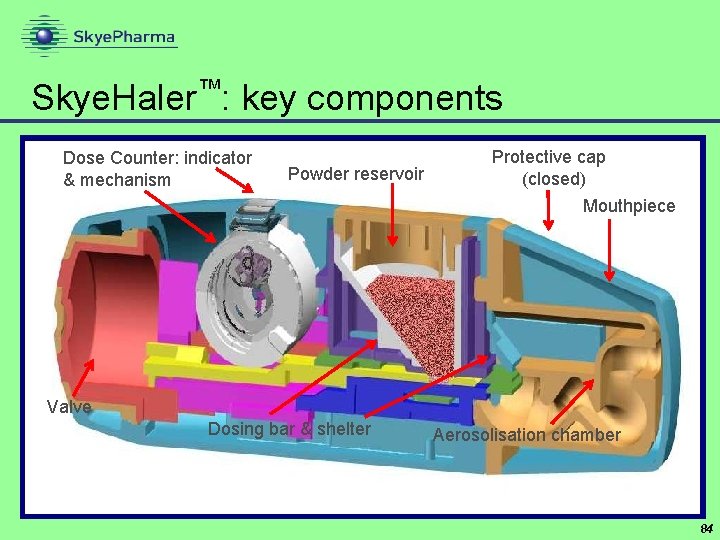
™ Skye. Haler : key components Dose Counter: indicator & mechanism Powder reservoir Protective cap (closed) Mouthpiece Valve Dosing bar & shelter Aerosolisation chamber 84

Skye. Haler™: operation Horizontal start position 85
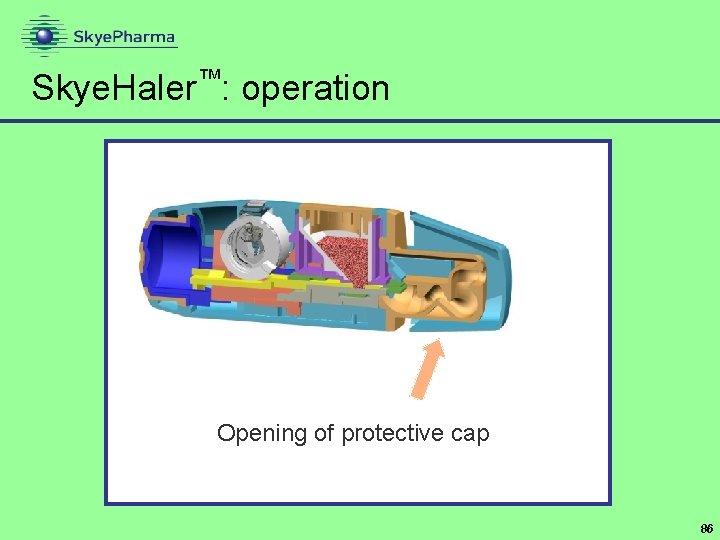
Skye. Haler™: operation Opening of protective cap 86

Skye. Haler™: operation Powder dispensed into dosing chamber 87

Skye. Haler™: operation Powder dose slides towards mouthpiece 88

Skye. Haler™: operation Powder dose slides towards mouthpiece 89

Skye. Haler™: operation Protective cap opened to 90 o position 90

Skye. Haler™: operation Powder in dispense position 91

Skye. Haler™: operation Device ready for inhalation 92

Skye. Haler™: operation Inhalation: powder dispersed through mouthpiece 93

Skye. Haler™: operation Inhalation: valve moves forward 94

Skye. Haler™: operation Closing of protective cap 95

Skye. Haler™: operation Closing of protective cap 96

Skye. Haler™: operation Closing of protective cap 97

Skye. Haler™: operation Counter advances 98
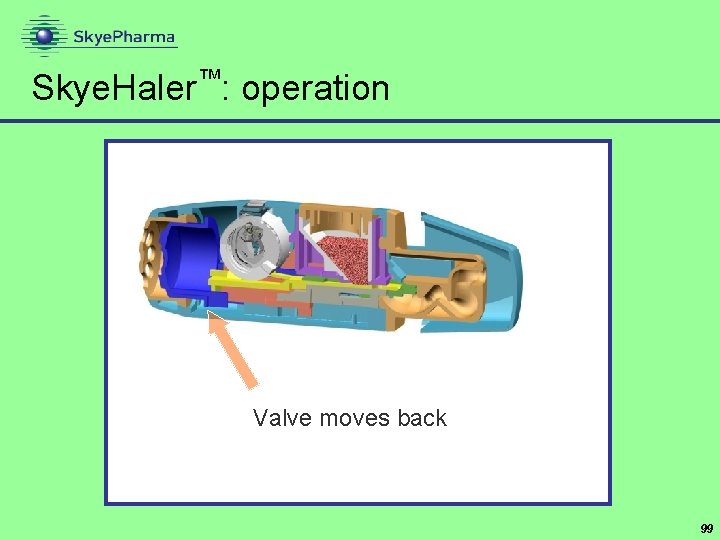
Skye. Haler™: operation Valve moves back 99

Skye. Haler™: operation Device ready for next actuation 100

™ Skyehaler – current status • in collaboration with Novartis, the Skyehaler™ has now been successfully modified • the adapted device provides equivalent performance to the previous device version • testing of the adapted device is complete • DMF (Drug Master File) updated by the device manufacturer Riwisa and data submitted to the FDA 101

Business Review Day 2006 Flutiform™ Dr Ken Cunningham Chief Operating Officer Adrian Adams CEO, Kos Pharmaceuticals 21 June 2006 NASDAQ: SKYE LSE: SKP www. skyepharma. com UK tel: +44 (0)207 491 1777 US tel: +1 (212) 753 5780

™ Flutiform – product features 103

Asthma therapy 1. 01 • asthma is an inflammatory condition - the airways become hyper-reactive to external stimuli • in an asthma attack, the airways narrow (“bronchoconstriction”), causing breathing difficulties Two complementary types of treatment are needed for asthma: • controller • an anti-inflammatory to control the underlying cause of asthma • most commonly an inhaled corticosteroid (“ICS”) • reliever • a beta-agonist bronchodilator to open the airways and make breathing easier • a short-acting beta-agonist is useful for acute relief from an asthma attack • a long-acting beta-agonist (“LABA”) provides 12 hours of relief • very important in avoiding night-time asthma attacks • “combination” treatments have an ICS and a LABA in the same container • convenient for patient better compliance • synergistic – the two agents work better together than on their own • delivery: metered-dose aerosol inhaler (“MDI”) or dry-powder inhaler (“DPI”) • each type has merits and drawbacks – no dominant advantage for either type 104

™ Flutiform product details • formoterol (LABA) and fluticasone (ICS) in a fixed-dose combination • formoterol 5 μg with fluticasone 50 μg or 125 μg per actuation (“puff”) • two actuations per dose • dosed twice a day (morning and night) • proprietary HFA-powered MDI with dose counter • covered by Skye. Dry™ patent (2019 expiration) • unique proprietary formulation technology • uses DSCG (cromolyn sodium, Intal) at sub-therapeutic dose as excipient to stabilize formoterol • maintains dose-to-dose consistency • target indication: asthma in adults and adolescents ≥ 12 • COPD and paediatric asthma to follow 105

Skye. Pharma’s proprietary MDI • • • active dispersion of drug to the airways dose is independent of patient’s inspiration flow-rate largely avoids common DPI problem of deposition of steroid in the throat and consequent hoarseness • • “steroid-induced laryngeal candidiasis” requires patient to have a drink of water after each DPI dose HFA propellant (non-CFC) dose counter indicates number of doses remaining • new FDA requirement for MDIs • perceived faster onset of effect with an MDI than with a DPI • Skye. Pharma’s MDI is small, portable, discrete and easy to use • no need to train patients in a new device 106

Flutiform - product profile • formoterol LABA (long-acting beta-2 agonist) • 12 hours bronchodilation = twice-daily dosing • faster onset of action (1 -3 mins) than salmeterol (30 -45 mins) • gives rapid relief from common symptom of wheeziness on waking • • • patient confidence that medication is working enhances compliance less risk of over-dosage • fluticasone ICS (inhaled corticosteroid) • low level of systemic uptake • physician-preferred ICS in the US • US physicians in particular are concerned by the risk of systemic uptake of inhaled steroids Source: TVG market research 2004 (commissioned by Skye. Pharma) 107

108

Flutiform™ – clinical trials 109

™ Fluti. Form - Phase II trial design • double-blind randomised 6 -way crossover study • • • Fluti. Form™ HFA p. MDI 100/10 g Fluti. Form™ HFA p. MDI 250/10 g fluticasone (Flixotide) HFA p. MDI 250 g + formoterol (Foradil) DPI 12 g fluticasone (Flixotide) HFA p. MDI 250 g formoterol (Foradil) DPI 12 g placebo • each patient received four of six treatments • N=64 (7 treatment centres in UK) 110

™ Fluti. Form - Phase II efficacy results • at both doses, Flutiform™ behaves exactly the same as the two component actives given separately • rapid onset (<5 min) of bronchodilation with both doses of Fluti. Form™ • 100/10 g and 250/10 g • bronchodilator effect maintained for ~12 hours • supports proposed twice-daily dosing • no interference or other drug/drug interactions • no safety concerns 111
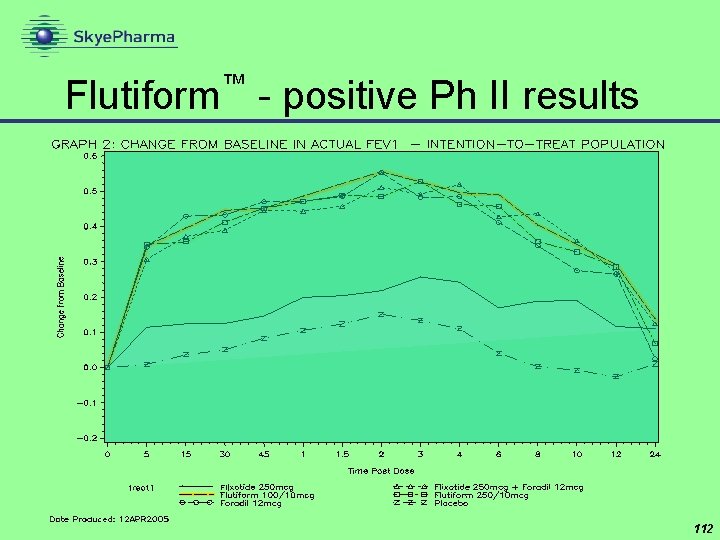
™ Flutiform - positive Ph II results 112

™ Fluti. Form - Phase III trials • design approved by FDA • 3 double-blind pivotal trials • primary end-point : FEV 1 • patient population: mild-moderate / moderate-severe asthmatics age >12 • duration: 12 weeks • one open-label 12 -month safety study • conducted in parallel with pivotal trials • total number of patients N = 1700 • ~300 treatment centres in Europe and North America • started as planned in Feb ’ 06 • target filing H 2 ‘ 07 113

™ Flutiform – commercial potential 114
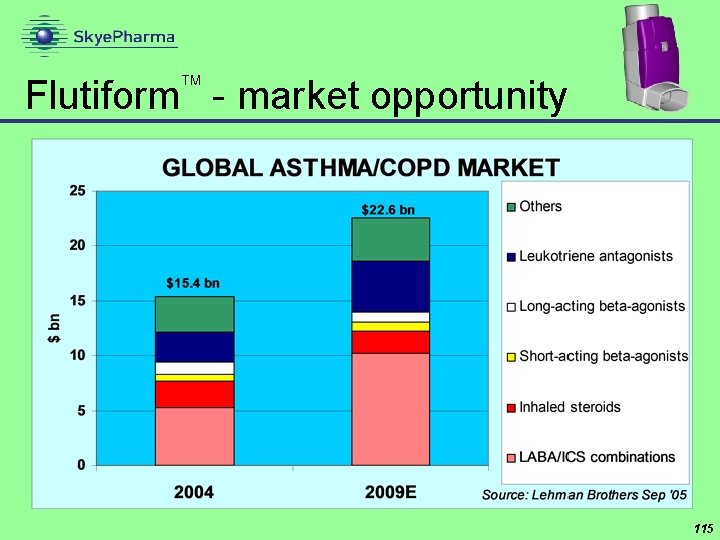
Flutiform - market opportunity 115
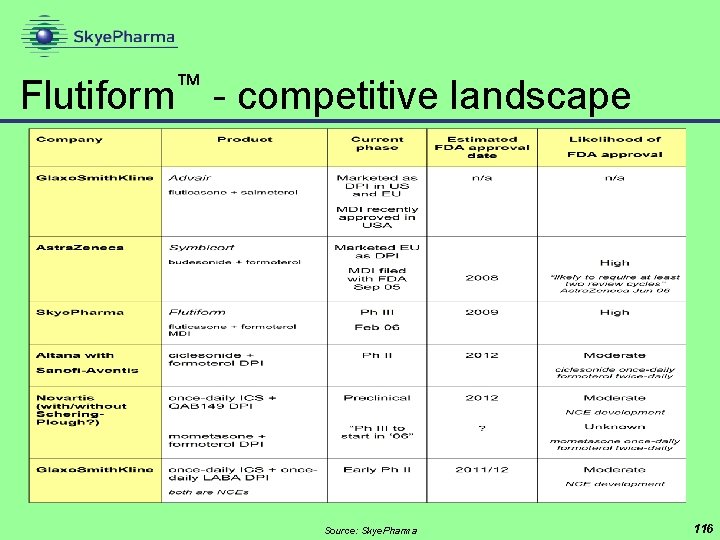
™ Flutiform - competitive landscape Source: Skye. Pharma 116
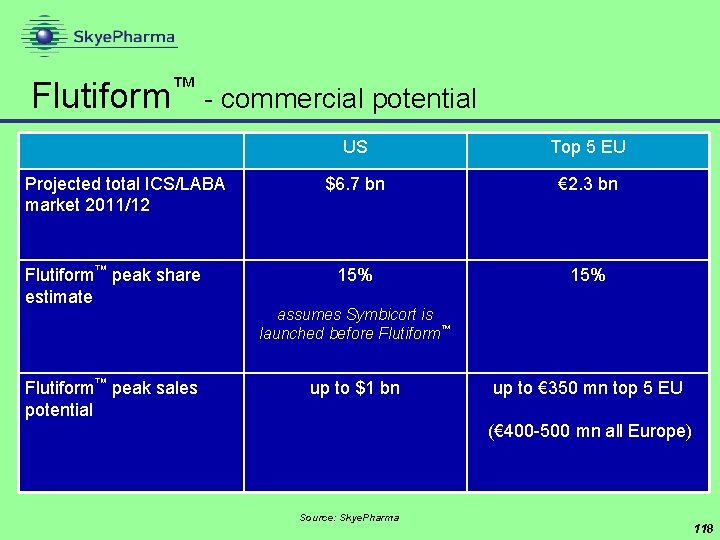
Flutiform™ - commercial potential Projected total ICS/LABA market 2011/12 Flutiform™ peak share estimate Flutiform™ peak sales potential US Top 5 EU $6. 7 bn € 2. 3 bn 15% assumes Symbicort is launched before Flutiform™ up to $1 bn up to € 350 mn top 5 EU (€ 400 -500 mn all Europe) Source: Skye. Pharma 118

Flutiform™ – US marketing partner 119

™ Flutiform US licence – Kos (May 2006) • Kos has exclusive marketing rights for Flutiform™ in US • right of first negotiation for Canada • Skye. Pharma receives up to $165 mn in milestone payments on regulatory & sales targets • includes $25 mn upfront • Skye. Pharma royalty rate starts in mid-teens • rate escalates on sales targets • Skye. Pharma and Kos share development of Flutiform™ for asthma and COPD • Skye. Pharma funds trials needed for approval in adult asthma • Kos funds trials for all other indications and marketing/post-approval studies • Skye. Pharma supplies product to Kos 120

™ US partner for Flutiform – why Kos? • successful, fully integrated US Specialty Pharmaceutical company • strong financial position, no debt, excellent long-term outlook • strong commercial capabilities, proven track record • 750 person sales force (>1000 by Flutiform™ launch), broad managed care, medical affairs and customer services functions • • • established presence in cardiovascular & asthma areas track record of respiratory market performance with Azmacort proven success in creating and building markets supported by quality medical and patient education • excellent, therapeutically aligned R&D capabilities • clinical, regulatory, safety and surveillance strengths in CV, metabolic and respiratory disease areas • both partners see high potential in the product • • • superior product concept differentiated from competing combinations clear window of opportunity 121

Introducing Adrian Adams President and Chief Executive Officer Kos Pharmaceuticals Cranbury, NJ, USA 122

Overview of Kos Pharmaceuticals • founded in 1988, IPO in 1997 (Nasdaq: KOSP) • named after Greek island of Kos – birthplace of Hippocrates • fully integrated Specialty Pharmaceutical company • core therapeutic areas: cardiovascular, respiratory, diabetes • key marketed brands: • 1400 associates (sales: 750, R&D: 225) – sales force increased to 900 by end ‘ 06 • headquarters in Cranbury, New Jersey – additional locations in NJ (Edison), FL (Weston and Hollywood) and NC (Raleigh) 123
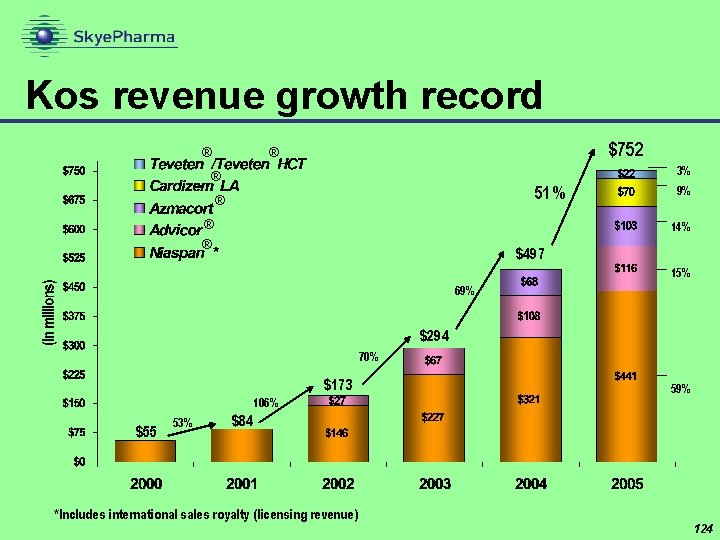
Kos revenue growth record ® $752 ® 3% ® 51% ® ® 9% 14% ® $497 15% 69% $294 70% $173 106% $55 53% 59% $84 *Includes international sales royalty (licensing revenue) 124

Kos - successful amongst “Big Pharma” • HDL market creation and development • launched Niaspan into LDL-dominated market - HDL awareness very low • 90 territory managers vs LDL “thousands” • Kos commercial philosophy • SMART sales and marketing with quality medical education • recruit experience and success, pay in top quartile • invest into success • successful and highly differentiated position • cholesterol franchise grown to around $600 mn – on way to billion dollars • dominant managed care position – over 95% lives covered • HDL drug of choice among cardiologists and primary care physicians • most big pharma companies now attempting access into “Kos market” 125

Kos - inhalation history • • 1993 • 1999 Kos acquires IEP Group, Inc. , a design and engineering company focused on proprietary inhalation devices • 2003 water as Kos acquires Aeropharm Technology inhalation platform 1997 Kos becomes a full member of IPACT-1, granting the Company necessary data for environmentally safe HFA-134 a • • Kos secures key HFA patent - involves innovative use of a stabilizing excipient scientific infrastructure and broad formulation and delivery device IP portfolio basis of inhaled insulin development – phase III ready in 2007 • 2004 Kos conducts strategic acquisition of global Azmacort franchise - represents first commercial entry into the respiratory market • 2006 Kos and Skyepharma announce exclusive licence agreement for Flutiform in the USA 126
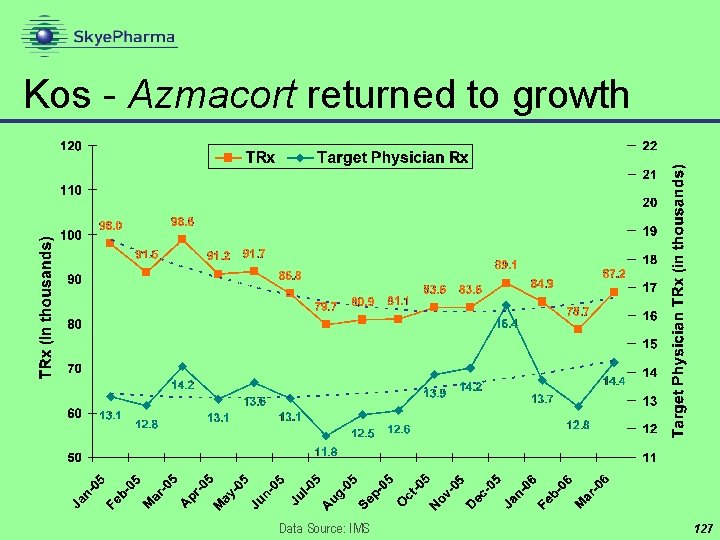
Kos - Azmacort returned to growth Data Source: IMS 127

™ Kos - rationale for Flutiform licence • strong strategic rationale • • • good fit with Azmacort, coverage of mono and combo market growth opportunity beginning 2008/9 2 projected new products to launch a year during 2007 -2009 strong financial returns • • • broadens Kos’ established presence in asthma/respiratory market win-win financial terms potential blockbuster with $500 MM plus anticipated peak year sales synergistic partnership 128

Kos – delivering value on many levels • focussed and successful specialty pharmaceuticals business model • • Focus in R&D - measured investments in “R” of R&D highly differentiated and sustainable cholesterol franchise • • • SMART targeted sales and marketing approach – enhanced productivity, eliminates “waste” increasing awareness of benefits of HDL, move towards combination therapy Niaspan & Advicor - most effective HDL therapies available - Simcor™ opportunity financial strength & expanding commercial brands • • strong cash position, no debt, strong earnings outlook in 2007 and beyond resources to fully exploit product franchises in CV, Metabolic and Respiratory Diseases robust Research & Development pipeline • • • one to two new products per year 2007 to 2009 – major potential with Simcor™ and Flutiform™ Kos’ inhaled insulin opportunity in high potential market progress in “measured” HDL and atherosclerosis NCE development 129

Business Review Day 2006 Skye. Pharma oral and inhalation delivery technologies Dr Guy Vergnault Head of Oral Business Unit Dr Geraldine Venthoye Head of Inhalation Business Unit NASDAQ: SKYE LSE: SKP www. skyepharma. com 21 June 2006 UK tel: +44 (0)207 491 1777 US tel: +1 (212) 753 5780

Skye. Pharma delivery technologies • Oral • most widely-used drug delivery route (~50% of all drugs taken orally) • Inhalation • • cost-effective metered-dose liquid aerosol and dry-powder inhalers enabling formulation technologies to ensure dose-to-dose consistency and extended shelf-life at room temperature • Solubilization • multiple technologies to improve bioavailability • Sustained-release injectable (unit to be divested) • Depo. Foam : active dissolved in water within multivesicular liposome particles • Biosphere : active dispersed in dry starch core within PLG shell • Topical • multiple transdermal technologies - now licensed to Trigenesis (Dr Reddy’s) 131

Geomatrix™ - a family of oral DDS, not a single DDS • why do we have more than one DDS ? • physicochemical characteristics of the API • pharmacological characteristics of the API • answer to specific therapeutic needs • zero order release cannot satisfy all needs for extended drug delivery • recent developments in oral portfolio • Geoclock™ press coated tablets • chronotherapy • colonic delivery • pulse delivery 132

™ Chronotherapy Geoclock Ischemic heart diseases THE BODY CLOCK Disorders displaying circadian rhythm modified Smolensky & Labrecque by Veski 133
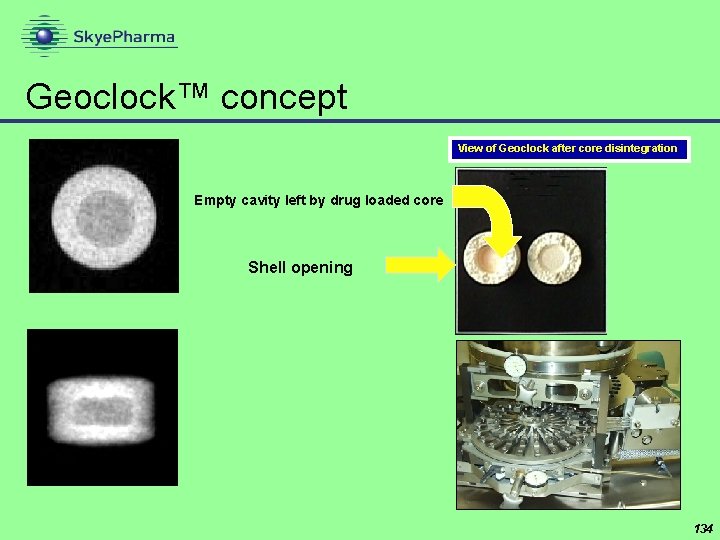
Geoclock™ concept View of Geoclock after core disintegration Empty cavity left by drug loaded core Shell opening 134
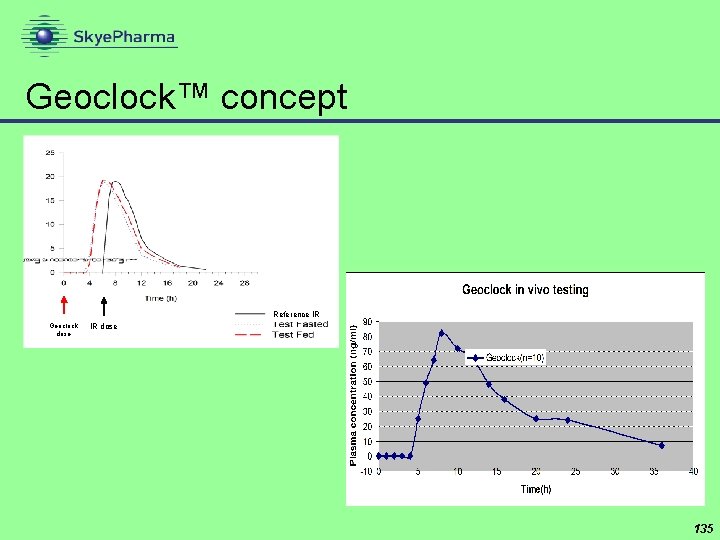
Geoclock™ concept Reference IR Geoclock dose IR dose 135
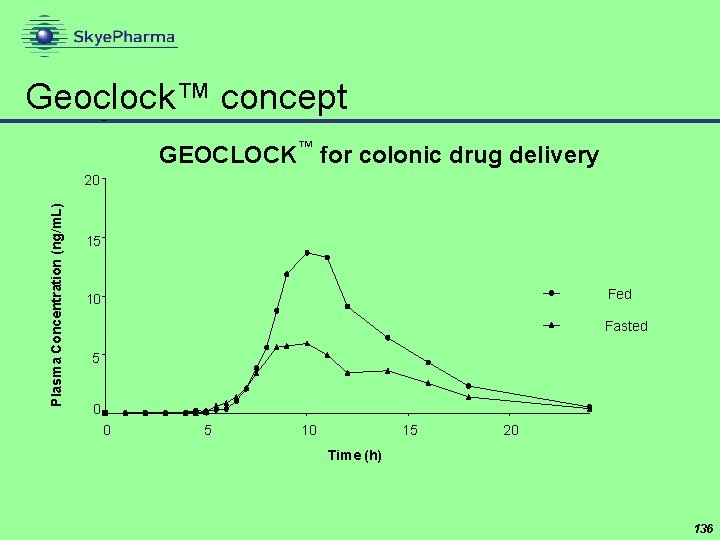
Geoclock™ concept GEOCLOCK™ for colonic drug delivery Plasma Concentration (ng/m. L) 20 15 Fed 10 Fasted 5 0 0 5 10 15 20 Time (h) 136
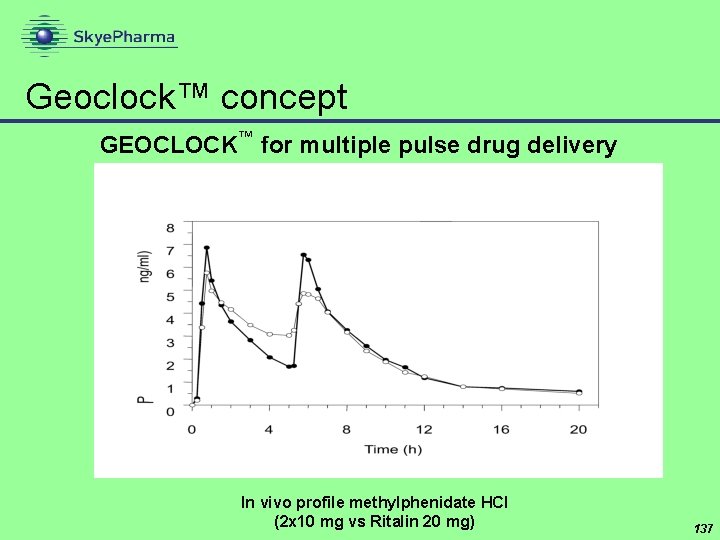
Geoclock™ concept GEOCLOCK™ for multiple pulse drug delivery In vivo profile methylphenidate HCl (2 x 10 mg vs Ritalin 20 mg) 137

Geoclock™ concept • proof of Concept in vivo trials have shown reliable performance of the Geoclock™ DDS • • industrial scale expertise • one product in late-stage clinical development many opportunities for specific timed or targetted drug delivery • Nitec product • supportive patent applications should provide protection beyond 2023 138

Solubilization technologies for oral route • increasing number of APIs have poor solubility • formulation issues • bioavailability issues • food effect • can result in under- or over-dosing • poor dose proportionality • Skye. Pharma technology solutions • Dissocubes® • SLN® (Solid Lipid Nanoparticles) • IDD® (Insoluble Drug Delivery) 139

Solubilization technologies for oral route • right combination of particle size and stabilization system • challenge of solid dosage formulation • conflicting targets • requires specific “savoir faire” • Triglide™ - first approved product based on IDD -P • commercial scale – 500 litre suspension • manufactured in Skye. Pharma’s Lyon plant 140

Solubilization technologies for oral route 141

Inhalation technologies - devices • Skye. Haler™ – “next generation” multi-dose breathactuated dry powder inhaler • accurate, robust and simple to use • medium-resistance device suitable for all types of patient • available at commercial scale manufacture volumes • HFA powered metered-dose aerosol inhaler • developed to meet stringent new regulatory guidelines • proprietary design incorporating dose counter • supplied for Ph III clinical programs from the commercial scale manufacture site 142

Inhalation technologies - formulation The use of a range of novel excipients to optimise dose-to-dose repeatability - a major hurdle for regulatory approval of inhalation products • Skye. Protect™ and Skye. Dry™ • “intrinsically” protect a powder or liquid suspension from the detrimental effects of moisture ingress and thereby stabilise labile compounds in the formulation • ensure accurate, consistent and reproducible dosing • Skye. Stabe™ • • • protects aerosol valve gaskets from abrasion improves inhaler functionality throughout can life and shelf life better suspension homogeneity provides superior dose uniformity • Skye. Fine™ • maximises the fine particle fraction (and therefore the dose delivered to the deep lung) in the dose emitted by a metered-dose aerosol inhaler 143

Business Review Day 2006 Early-stage pipeline Dr Ken Cunningham Chief Operating Officer NASDAQ: SKYE LSE: SKP www. skyepharma. com 21 June 2006 UK tel: +44 (0)207 491 1777 US tel: +1 (212) 753 5780
Early-stage pipeline • SKP-1032 : opioid-NSAID combo BID • SKP-1041 : non bzd hypnotic TRT • SKP-6005 : PPARα /PPARγ combo OD • SKP-6006 : ARB CR OD • only SKP-1032 and SKP-1041 will be presented today 145

SKP-1032 Opioid – NSAID combo Short-term treatment of moderate-to-severe acute pain 146

SKP-1032 – clinical advantages • combines an anti-inflammatory dose of an NSAID with the potency of an opioid analgesic • • only combination of an opioid with this NSAID • • selected NSAID is preferred by physicians selected NSAID perceived as more effective than ibuprofen greater potentiation of opioid effect with the selected NSAID compared with ibuprofen 12 -hour pain relief with rapid onset • • useful for orthopaedic surgeries, sprains, fractures avoids peaks and troughs and breakthrough pain Skye. Pharma product will be Schedule III 147
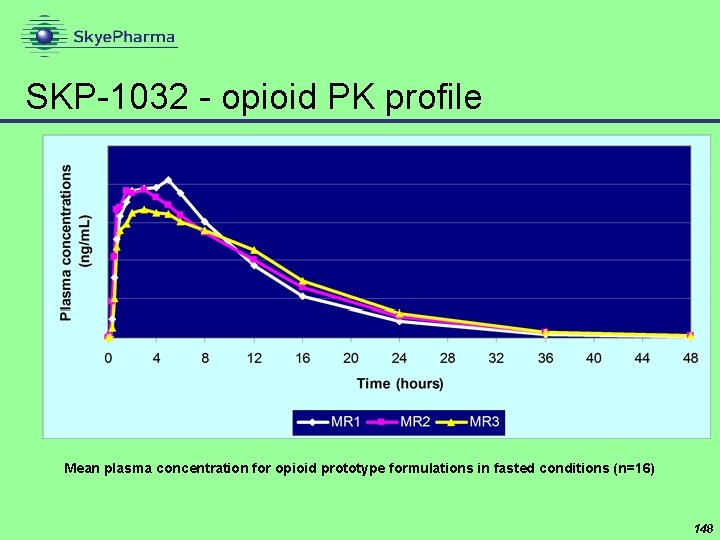
SKP-1032 - opioid PK profile Mean plasma concentration for opioid prototype formulations in fasted conditions (n=16) 148
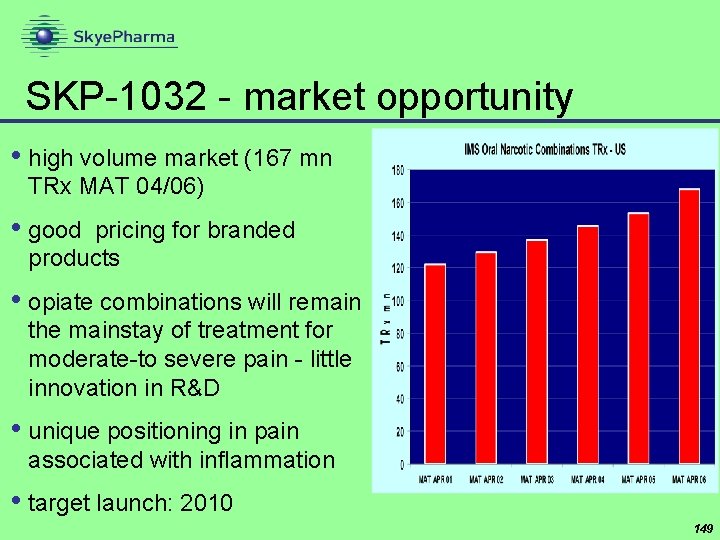
SKP-1032 - market opportunity • high volume market (167 mn TRx MAT 04/06) • good pricing for branded products • opiate combinations will remain the mainstay of treatment for moderate-to severe pain - little innovation in R&D • unique positioning in pain associated with inflammation • target launch: 2010 149

SKP-1041 Non-benzodiazepine hypnotic Sleep maintenance Geo. Clock™ tablets ensure optimum release profile
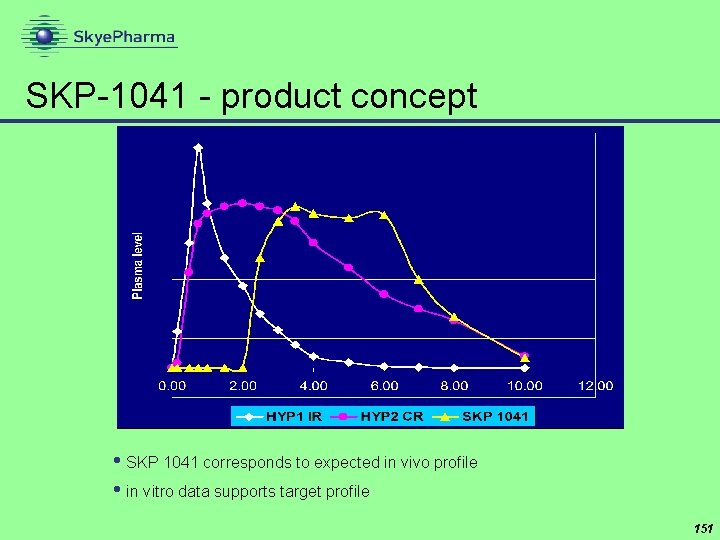
SKP-1041 - product concept • SKP 1041 corresponds to expected in vivo profile • in vitro data supports target profile 151

SKP-1041 – unique clinical advantage • non-benzodiazepine hypnotic • first hypnotic product specifically designed for patients who have difficulty with sleep maintenance • lowest effective hypnotic dose • no drug on board when not needed = “natural” sleep onset • very short half-life, resulting in fast clear-headed awakening 152
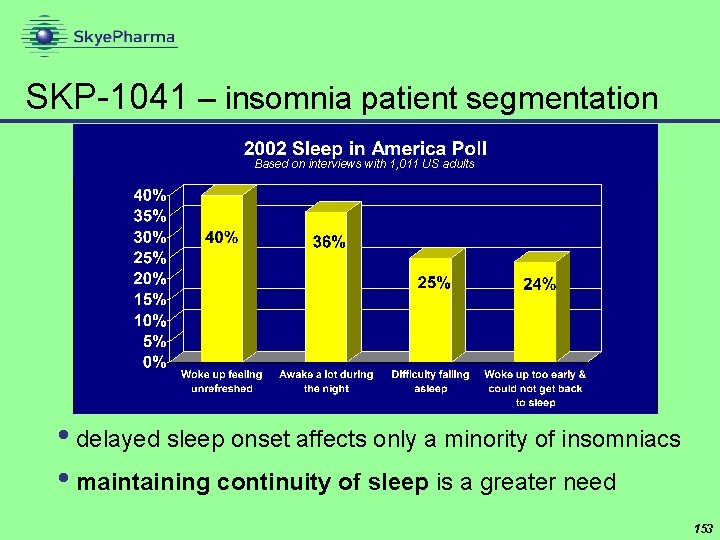
SKP-1041 – insomnia patient segmentation Based on interviews with 1, 011 US adults • delayed sleep onset affects only a minority of insomniacs • maintaining continuity of sleep is a greater need 153
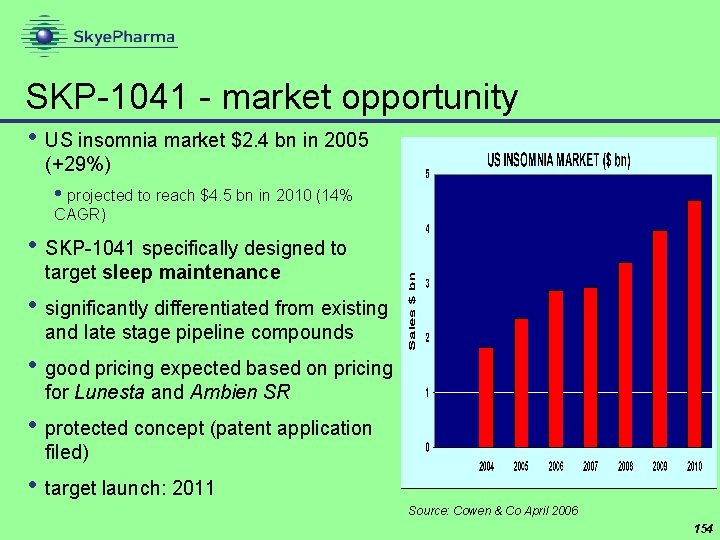
SKP-1041 - market opportunity • US insomnia market $2. 4 bn in 2005 (+29%) • projected to reach $4. 5 bn in 2010 (14% CAGR) • SKP-1041 specifically designed to target sleep maintenance • significantly differentiated from existing and late stage pipeline compounds • good pricing expected based on pricing for Lunesta and Ambien SR • protected concept (patent application filed) • target launch: 2011 Source: Cowen & Co April 2006 154

Fast track from technologies to marketed products The 505 (b) 2 approval process 155

The 505(b)(2) approval process • Hatch-Waxman amendment to the Federal Food, Drug & Cosmetics Act to allow an NDA based in part on data NOT generated by the applicant • scientific literature and/or • data used to support the filing of a product previously approved by the FDA • examples: • a new dosage form or formulation of an approved product • a new indication of an approved product • a new route of administration of an approved product • substitution of an active in an approved combination product • a pro-drug of an approved product • 505(b)(2) applicants must certify non-infringement of patents • 505(b)(2) approval earns 3 years of market exclusivity • 5 years for an NCE • does not affect protection arising from specific IP 156

Business Review Day 2006 Summary Dr Ken Cunningham Chief Operating Officer 21 June 2006 NASDAQ: SKYE LSE: SKP www. skyepharma. com UK tel: +44 (0)207 491 1777 US tel: +1 (212) 753 5780

Major objectives 2006 • divest injectable unit • outlicense Flutiform™ • negotiations for non-US territories ongoing • complete modifications to Certihaler™ • expand oral / inhalation pipeline • work with licensing partners to drive revenues of marketed products 158

The new Skye. Pharma • new leadership • drive for sustainable profitability • accelerated by sale of injectables unit • core business is oral and inhalation • potential blockbuster in Flutiform™ • strong cashflow from existing royalties 159

Q&A This presentation is being webcast Please give your name and affiliation when asking questions 160

Investor relations contacts London Peter Laing, Director of Corporate Communications 44 -(0)207 -491 -1777 plaing@skyepharma. co. uk New York Sandra Haughton, US Investor Relations Manager 1 -212 -753 -5780 shaughton@skyepharma. com …and please visit our website NASDAQ: SKYE LSE: SKP www. skyepharma. com

Business Review Day 2006 Back-up slides These slides will not be shown 21 June 2006 NASDAQ: SKYE LSE: SKP www. skyepharma. com UK tel: +44 (0)207 491 1777 US tel: +1 (212) 753 5780
- Slides: 144