Bullseye Activity Did the pennies consistently drop in
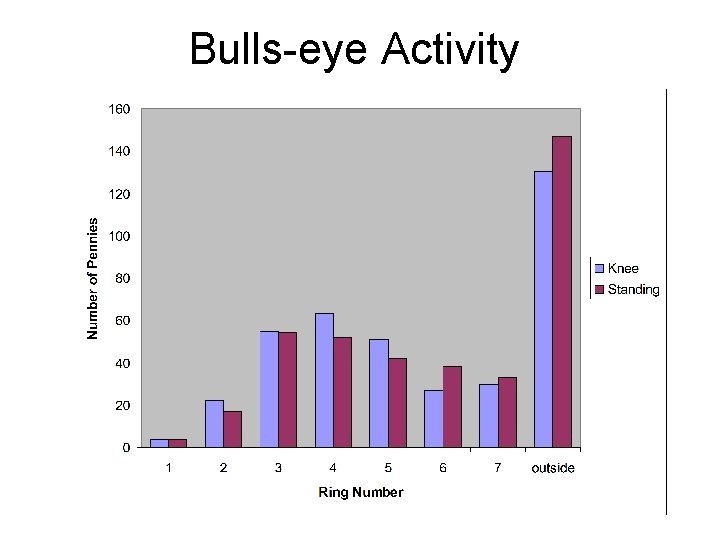
Bulls-eye Activity

• Did the pennies consistently drop in the same ring? • Can we use our empirical evidence to predict exactly where a penny would land?

Quantum Mechanical Model of the Atom
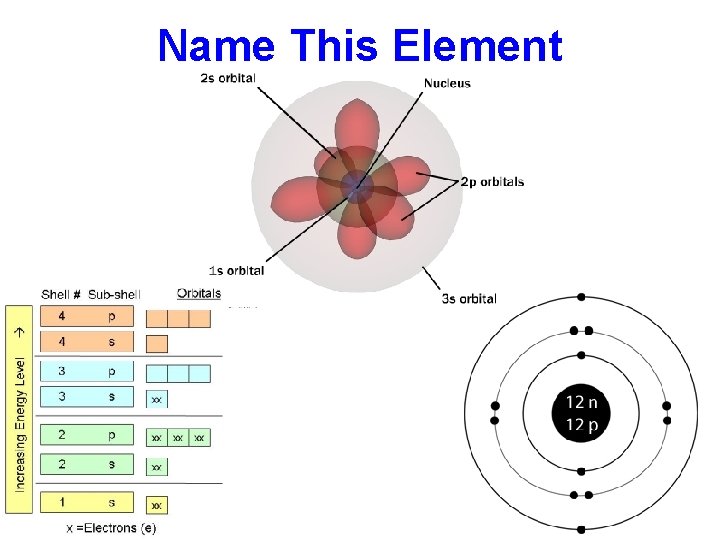
Name This Element
Building on Bohr • The simple Bohr model was unable to explain properties of complex atoms • Only worked for hydrogen • A more complex model was needed… • Our understanding of physics needed to “catch up”

Wave Particle Duality • Experimentally, De. Broglie found that light had both wave and particle properties • Therefore De. Broglie assumed that any particle (including electrons) could also travel in waves • Wavelengths must be quantized or they would cancel out • Applied his theory to Bohr’s model and it fit, but still only for hydrogen • Video

Heisenberg’s Uncertainty Principle • Due to the wave and particle nature of matter, it is impossible to precisely predict both the position and momentum of an electron • Δposition • Δmv = a constant • We can only describe a region of probability of finding an electron • Video

SchrÖdinger’s Wave Equation • SchrÖdinger’s developed a mathematical equation that could be used to determine a region of probability for finding an electron • Substitute in a series of quantum numbers to solve the wave function • His equation matched Bohr’s calculations including multi-electron atoms.


Quantum Mechanics • Uses mathematical equations to describe the wave properties of subatomic particles • It’s impossible to know both the exact position, momemtum of an electron (Heisenberg Uncertainty Principle) • So Bohr’s “orbits” were replaced by orbitals – A wave function that predicts an electron’s energy and location within an atom – A probability cloud in which an electron is most likely to be found
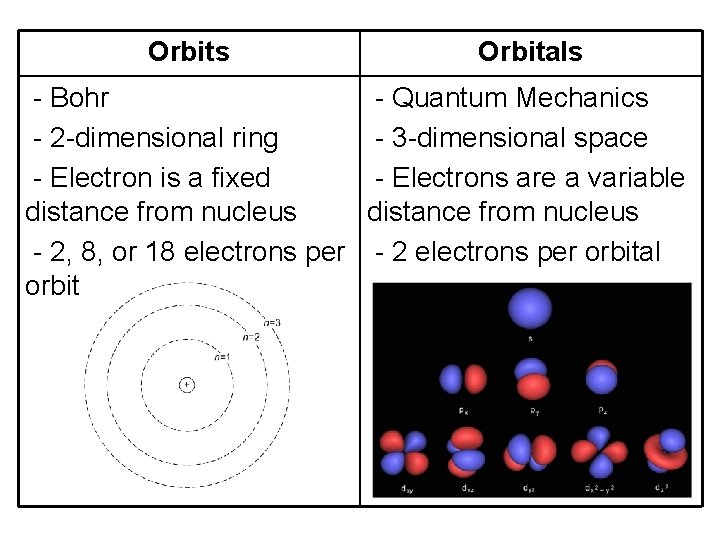
Orbits Orbitals - Bohr - Quantum Mechanics - 2 -dimensional ring - 3 -dimensional space - Electron is a fixed - Electrons are a variable distance from nucleus - 2, 8, or 18 electrons per - 2 electrons per orbital orbit

Practice Problems • Read p. 199 -202 • P. 202 #1 -5 • “Empirical Evidence supports theoretical knowledge”

Quantum Numbers • Four numbers used to describe a specific electron in an atom • Each electron has its own specific set of quantum numbers • Recall: Describes orbitals (probability clouds)

The Principal Quantum Number “n” • Indicates the average distance (size) of the orbital from the nucleus (same as Bohr’s energy levels) • Higher n = greater distance from nucleus = greater energy • n = integers > 1 (1, 2, 3…) • The greatest number of electrons possible in each energy level is 2 n 2
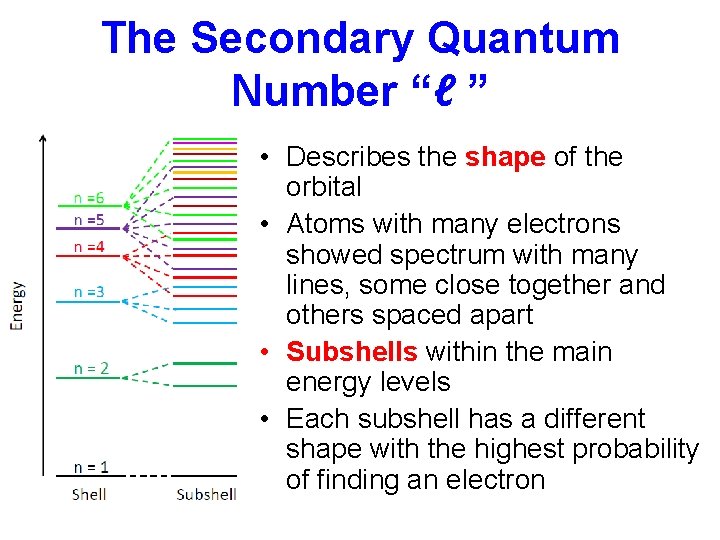
The Secondary Quantum Number “ℓ ” • Describes the shape of the orbital • Atoms with many electrons showed spectrum with many lines, some close together and others spaced apart • Subshells within the main energy levels • Each subshell has a different shape with the highest probability of finding an electron

The Secondary Quantum Number “ℓ ” • Positive integers ranging from 0 -3 • Maximum value of n-1 – ℓ = 0 (s orbital) – ℓ = 1 (p orbital) – ℓ = 2 (d orbital) – ℓ = 3 (f orbital) • Total number of sublevels = n
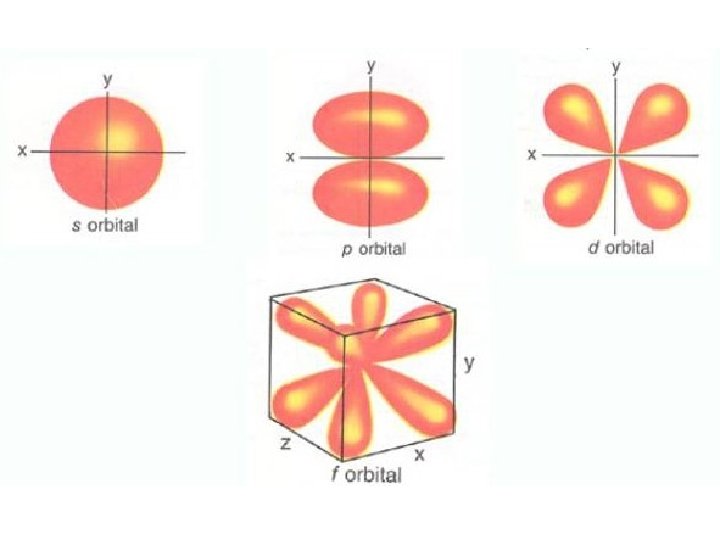

The Magnetic Quantum Number “mℓ” • Describes orientation of the orbital • mℓ = integers from -ℓ to +ℓ • Maximum number of orientations = n 2
Animation
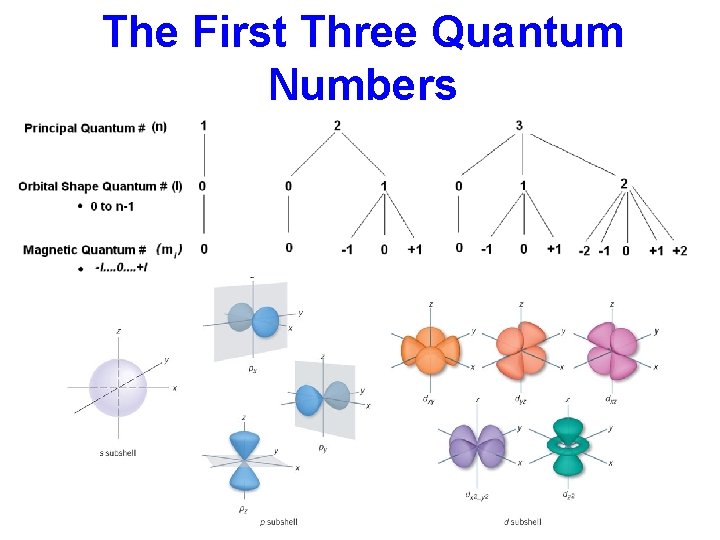
The First Three Quantum Numbers

The Spin Quantum Number “ms” • Describes the direction an electron is spinning in a magnetic field (up or down) • Only two electrons per orbital • ms = + 1/2 or - 1/2

Letter Analogy Miss Smith 4 The Parkway Kanata ON n= 3 ℓ=1 mℓ = -1 ms = +1/2

Quantum Numbers Summary Chart Name Symbol Allowed Values Property n positive integers 1, 2, 3… Orbital size and energy level Secondary ℓ Integers from 0 to (n-1) Orbital shape (sublevels/subshells) Magnetic mℓ Integers –ℓ to +ℓ Orbital orientation +½ or –½ Electron spin Direction Principal Spin ms

Practice! • • Read p. 181 -184 Quantum number handout p. 182 #3 -5 p. 184 #3 -7
- Slides: 24