Building Materials Metals Metals Useful building material Exist

Building Materials Metals

Metals Useful building material Exist in nature as compounds like oxides, carbonates, sulphides and phosphates Ores → Remove the impurities → metals Ferrous metals – iron is the main constituent eg. Cast iron, wrought iron, steel Non Ferrous metals – iron is not the main constituent eg. Aluminium, zinc, lead, tin

Ferrous Metals Iron Available abundantly but not freely. It is found in ores Eg. Magnetite – Fe 3 O 4 - 70 -75% iron Haematite – Fe 2 O 3 – 70% iron Limonite – 2 Fe 3 O 4. 3 H 2 O – 60% iron Iron pyrite – Fe. S 3 – 47% iron Haematite
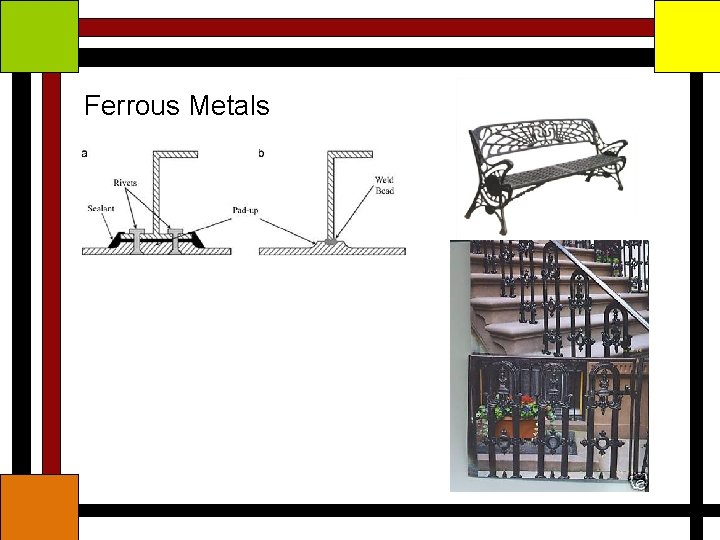
Ferrous Metals
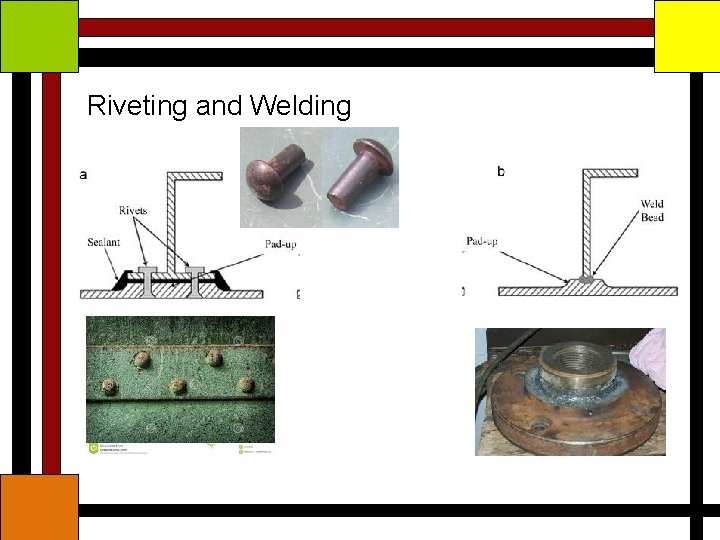
Riveting and Welding

Ferrous Metals Steel and iron 1. Iron is an element while steel is an alloy of iron and carbon. 2. Iron has less carbon than steel. 3. Steel is used in construction because it is stronger than iron and has a superior tensile strength. 4. Iron was known to the humans from the beginning of civilization while steel was discovered much later.

Ferrous Metals Steel The most suitable among metallic building material Because of wide range of physical and mechanical properties By controlling the carbon content, alloying elements and heat treatment, a desired combination of hardness, strength and ductility can be obtained in steel. . Composition of steel Iron, Carbon, Manganese, Aluminum, Chromium, Columbium, Copper, Molybdenum, Nickel, Silicon, Vanadium

Ferrous Metals Manufacture of Steel Bessemer process Open Hearth process Electric process Blast of hot air is pressurized to oxidise the carbon Carbon and Manganese are added and hot air is blasted again Then the molten materials is pored into moulds to form ingots

Ferrous Metals Properties and uses Mild steel – low carbon or soft steel Rolled section, reinforcing bars, roof coverings, railway tracks High carbon steel – 0. 61. 5%carbon - hard steel – RCC and pre-stressed concrete members High tensile steel – 0. 6 -0. 8% carbon – prestressed concrete construction
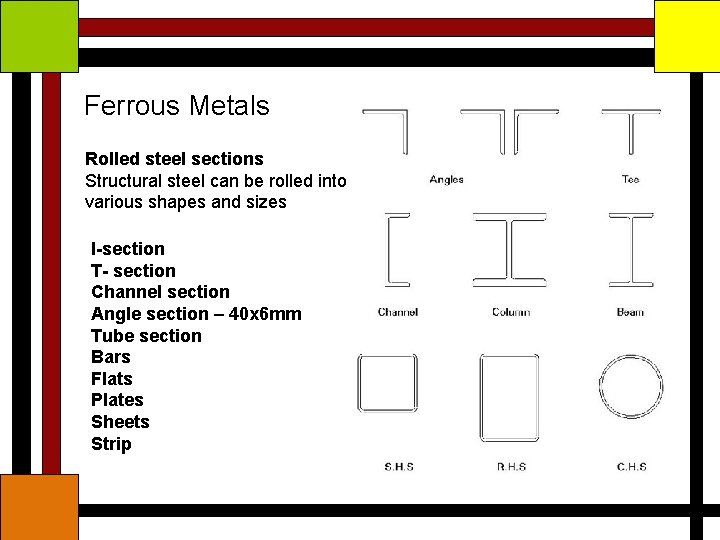
Ferrous Metals Rolled steel sections Structural steel can be rolled into various shapes and sizes I-section T- section Channel section Angle section – 40 x 6 mm Tube section Bars Flats Plates Sheets Strip

Ferrous Metals Rolled steel sections Structural steel can be rolled into various shapes and sizes I-section T- section Channel section Angle section Tube section Bars Flats Plates Sheets Strip Bars Plates 2000 x 1000 x 8 mm Flats 30 x 10 mm

Ferrous Metals Rolled steel sections Structural steel can be rolled into various shapes and sizes I-section T- section Channel section Angle section Tube section Bars Flats Plates Sheets Strip Sheets strips Sheets 2000 x 600 x 4 mm

Ferrous Metals Reinforcing steel bars Concrete being weak in tension requires reinforcement which is invariably steel Available in the form of bars of different diameters and of different chemical composition – mild steel, high tensile steel and surface characteristics – plain or deformed

Ferrous Metals Rusting and Corrosion When steel is exposed to atmosphere, the humid air causes rusting and along with rain produces oxidation and corrosion

Ferrous Metals To safe guard from rusting and corrosion, some of the prevalent methods are 1. enameling; 2. applying metal coatings – galvanizing, electro plating, tin plating 3. Applying organic coatings – painting and coal tarring The glass powder melts, flows, and then hardens to a smooth, durable vitreous coating on metal - enameling

Ferrous Metals applying metal coatings – galvanizing, electro plating, tin plating add a thin layer of another metal to an item made of steel, in order to prevent rusting - galvanising

Ferrous Metals applying metal coatings – galvanizing, electro plating, tin In industry electroplating is often the plating process used to galvanise steel products as it is more efficient and more cost effective than the hot dipping process. The thickness of the coating can be determined with great accuracy.

Non Ferrous Metals Production of non ferrous metals is small when compared to iron but they are used as building material despite their high cost Aluminium, copper, tin, zinc, lead Aluminium and manganese zinc tin copper
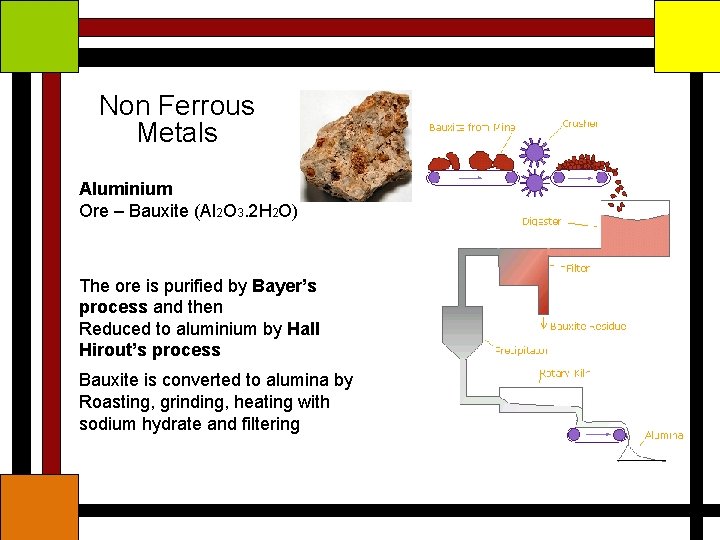
Non Ferrous Metals Aluminium Ore – Bauxite (Al 2 O 3. 2 H 2 O) The ore is purified by Bayer’s process and then Reduced to aluminium by Hall Hirout’s process Bauxite is converted to alumina by Roasting, grinding, heating with sodium hydrate and filtering
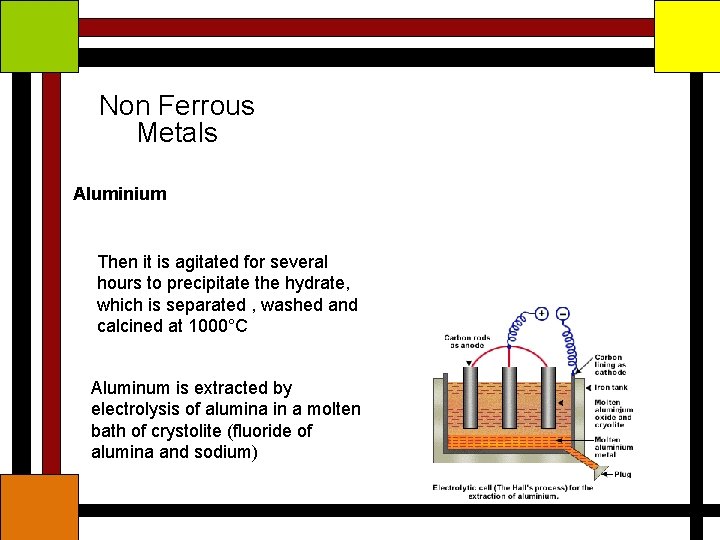
Non Ferrous Metals Aluminium Then it is agitated for several hours to precipitate the hydrate, which is separated , washed and calcined at 1000°C Aluminum is extracted by electrolysis of alumina in a molten bath of crystolite (fluoride of alumina and sodium)

Non Ferrous Metals Properties of Aluminium Silver white in colour with a metallic lustre Malleable, less ductile than copper Very light, soft, strong and durable, low thermal conductivity, good conductor of electricity

Non Ferrous Metals Uses of aluminium Pure aluminium is soft and is not suitable for structural purposes But satisfactory properties are achieved by alloying copper, manganese, zinc, silicon, nickel with aluminium. Make door and window frames Railings Corrugated sheets Aluminium sheets are used over doors in bathrooms Aluminium powder is used to make paint Parts of engine, airplanes, utensils, etc Aluminium alloys used in the manufacture of rolled sections – angles, channels, Isections, round and rectangular pipes, rivets and bolts
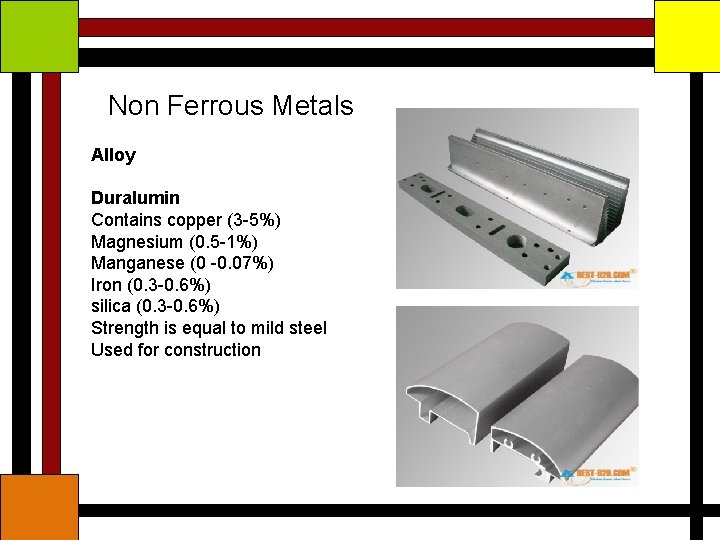
Non Ferrous Metals Alloy Duralumin Contains copper (3 -5%) Magnesium (0. 5 -1%) Manganese (0 -0. 07%) Iron (0. 3 -0. 6%) silica (0. 3 -0. 6%) Strength is equal to mild steel Used for construction
- Slides: 23