Building blocks of rocks MINERALS MINERALS A mineral

“Building blocks” of rocks MINERALS

MINERALS �A mineral is a � Naturally occuring � Inorganic � Solid with a � Definite chemical composition and � Crystalline structure �A substance must satisfy ALL FIVE characteristics in order to be classified as a mineral.

MINERALS � Naturally � Must occuring be formed through natural geologic processes � Crystallization from magma � Precipitation (from solutions – not rain) � High temperature and pressure � Hydrothermal solutions � CANNOT be man-made or synthetic � Inorganic � Cannot come from living things (there a few exceptions)

MINERALS � Solid � � Must exist in the solid state under NORMAL temperatures on the Earth’s surface Definite chemical composition Must be either an element or a compound � Cannot be a mixture (mixtures are rocks) � � Crystalline structure Atoms must be arranged in a repeating geometric pattern � There are 7 crystal groups. �

MINERALS � There are SIX major groups of Minerals. � Groups are determined by the mineral’s chemical composition (formula) 1. 2. 3. 4. 5. 6. Silicates Carbonates Oxides Sulfates/Sulfides Halides Natural Elements

SILICATES � Silicon and oxygen combine to form a structure called the silicon-oxygen tetrahedron. � Most common group of minerals � Examples: � Quartz – most common � Olivine � Augite � Mica � Feldspar

CARBONATES � Minerals that contain the elements carbon, oxygen, and one or more other metallic elements � Examples: � Calcite - most common � dolomite

OXIDES � Minerals that contain oxygen and one or more other elements, which are usually metals � Examples: � Rutile � Corundum � Hematite

SULFATES / SULFIDES � Minerals that contain the element sulfur � Examples: � Anhydrite � Gypsum � Galena � Pyrite (Fool’s Gold)

HALIDES � Minerals that contain a halogen ion plus one or more other elements � Halogens are elements in Group 17 on the Periodic Table � Examples: � Halite – where we get table salt � Fluorite

NATURAL ELEMENTS � Minerals that exist in relatively pure form � Elements – not compounds � Examples: � Gold � Silver � Copper � Sulfur � Carbon – graphite and diamond

PROPERTIES OF MINERALS

PROPERTIES � Color Results from elements in mineral � Impurities can change the color of a mineral � � Streak Color of the mineral in powdered form � Sometimes same as mineral color; sometimes different � � Luster Way light reflects off of the mineral � Metallic, glassy, dull, silky, etc. �

PROPERTIES � Hardness � Resistance to being scratched � Value determined using Moh’s Hardness Scale

PROPERTIES � Crystal form � Arrangement � Cleavage � Way of atoms in a geometric pattern vs. Fracture a mineral breaks � Even = Cleavage � Uneven = Fracture

PROPERTIES � Density � Mass per unit volume � D = M/V � Distinctive Properties exhibited by some minerals � Magnetic � Texture/Feel – greasy, soft, soapy, etc. � Smell � Double Refraction

Chapter 3 ROCKS

ROCKS � Any solid mass of mineral or mineral-like matter that occurs naturally � Most rocks are MIXTURES of minerals � Some rocks contain nonmineral material (shells, plants, bones, etc. ) � Some rocks do not have crystalline structures

CLASSIFICATION OF ROCKS � Rocks are classified based on how they were formed. � Three classifications � Igneous – form when magma or lava cools and hardens � Sedimentary – form when sediments are compacted and cemented � Metamorphic – form when rocks are changed by extreme heat and pressure

ROCK CYCLE � Shows relationships between different rock forms � All rocks can be changed from one kind to another � Ex. Igneous rocks can become sedimentary rocks through weathering and erosion � The Rock Cycle shows how these changes can take place.
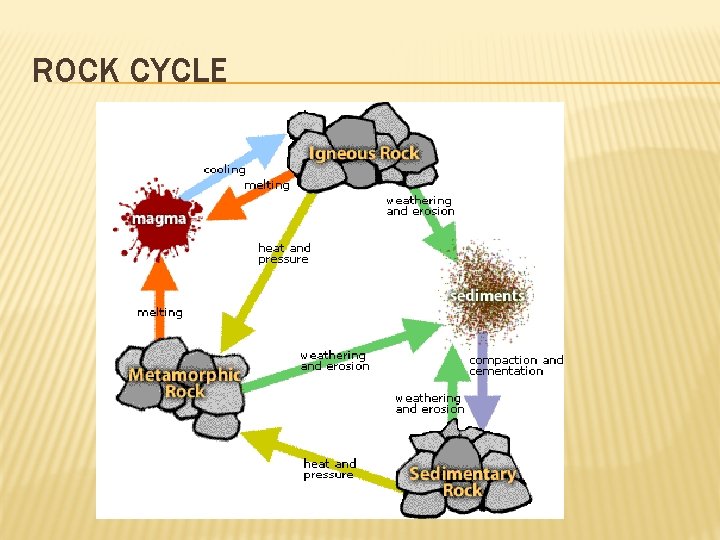
ROCK CYCLE
- Slides: 21