Buffered Solutions Chem 101 Dr Prem D Sattsangi

Buffered Solutions Chem 101 Dr. Prem D. Sattsangi © 2010 - and Christopher L. Byers (programmer)

Acids and Bases Definitions Acid Base Arrhenius: produce H+ produce OH- Bronsted-Lowry: H+ donor H+ acceptor Lewis: electron pair acceptor electron pair donor
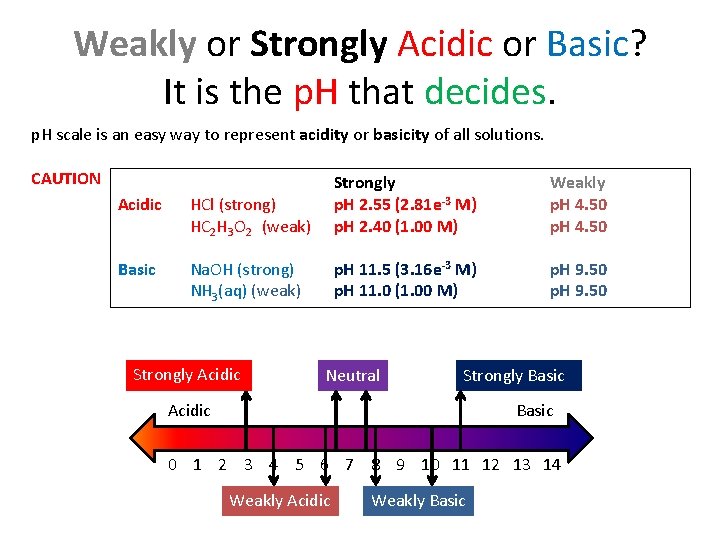
Weakly or Strongly Acidic or Basic? It is the p. H that decides. p. H scale is an easy way to represent acidity or basicity of all solutions. CAUTION Acidic HCl (strong) HC 2 H 3 O 2 (weak) Strongly p. H 2. 55 (2. 81 e-3 M) p. H 2. 40 (1. 00 M) Basic Na. OH (strong) NH 3(aq) (weak) p. H 11. 5 (3. 16 e-3 M) p. H 11. 0 (1. 00 M) Strongly Acidic Neutral Weakly p. H 4. 50 p. H 9. 50 Strongly Basic Acidic Basic 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 Weakly Acidic Weakly Basic

Application of p. H Scale • • SCE: Classify the following as weakly/strongly acidic, basic, or neutral. Weakly Acidic Black Coffee p. H ≈ 4. 9 Weakly Basic Baking Soda p. H ≈ 8. 2 Strongly Acidic Cola p. H ≈ 2. 9 Neutral Milk p. H ≈ 6. 7 Weakly Basic Lime Water p. H ≈ 10. 4 Use p. H to figure it out. Table: p. H scale 0 -3 Strongly Acidic 4 -6 Weakly Acidic 6. 5 -7. 5 Neutral 8 -10 Weakly Basic 11 -14 Strongly Basic
![Concentration and p. H Table: [H+] 10 x decrease vs. p. H +1 increase Concentration and p. H Table: [H+] 10 x decrease vs. p. H +1 increase](http://slidetodoc.com/presentation_image_h2/a95ea8110e3b974686127b0e6466c2d0/image-5.jpg)
Concentration and p. H Table: [H+] 10 x decrease vs. p. H +1 increase [H+] p. H 1. 0 x 10 -1 1. 00 1. 0 x 10 -2 2. 00 1. 0 x 10 -3 3. 00 1. 0 x 10 -14 14. 00 • Concentration decreases by 10 x, the p. H increases by 1 unit. • p. H scale is from 0 -14. • p. H + p. OH = 14
![p. H calculation • • • For strong acids M of acid = [H+] p. H calculation • • • For strong acids M of acid = [H+]](http://slidetodoc.com/presentation_image_h2/a95ea8110e3b974686127b0e6466c2d0/image-6.jpg)
p. H calculation • • • For strong acids M of acid = [H+] HCl(aq) H+(aq) + Cl-(aq) (completely ionized) For Example, 0. 0253 M HCl = 0. 0253 M H+ p. H = -log[H+] = [0. 0253 M] p. H of 0. 0253 M HCl = -log[0. 0253] = 1. 5969 = 1. 597 (3 sig. digits) • To find p. H on calculator… (3 decimal places) 1. press (-) 2. press “log” 3. enter the [H+] • E. g. If [H+] = 1. 25 x 10 -5 M p. H = 4. 903 (-) log 1. 25 e-5 =
![p. OH calculation • • • For strong bases M of base = [OH-] p. OH calculation • • • For strong bases M of base = [OH-]](http://slidetodoc.com/presentation_image_h2/a95ea8110e3b974686127b0e6466c2d0/image-7.jpg)
p. OH calculation • • • For strong bases M of base = [OH-] Na. OH(aq) Na+(aq) + OH-(aq) (completely ionized) For Example, 0. 0253 M Na. OH = 0. 0253 M OHp. OH = -log[OH-] = [0. 0253 M] p. OH of 0. 0253 M Na. OH = -log[0. 0253] = 1. 5969 = 1. 597 3 sig. digits 3 dec. places • To find p. OH on calculator… 1. press (-) 2. press “log” 3. enter the [OH-] • E. g. If [OH-] = 1. 25 x 10 -5 M p. OH = 4. 903 (-) log 1. 25 e-5 =
![Finding p. H of a strong base [OH-] p. OH p. H • Eg. Finding p. H of a strong base [OH-] p. OH p. H • Eg.](http://slidetodoc.com/presentation_image_h2/a95ea8110e3b974686127b0e6466c2d0/image-8.jpg)
Finding p. H of a strong base [OH-] p. OH p. H • Eg. 1. 25 x 10 -5 M OH- p. OH = -log[1. 25 e-5] = 4. 903 • p. H + p. OH = 14 p. H = 14 – p. OH • p. H = 14 – 4. 903 • p. H = 9. 097 • SCE: [OH-] = 1. 25 e-3, Calculate p. OH _____ • Calculate p. H _____ • p. OH = 2. 903, p. H = 11. 097
![Calculating H+ or OH- from p. H • Inverse log[-p. H] = [H+] • Calculating H+ or OH- from p. H • Inverse log[-p. H] = [H+] •](http://slidetodoc.com/presentation_image_h2/a95ea8110e3b974686127b0e6466c2d0/image-9.jpg)
Calculating H+ or OH- from p. H • Inverse log[-p. H] = [H+] • On calculator… 1. press 10 X (Usually 2 nd log) 2. press (-) 3. enter [p. H] E. g. 10 X [-2. 90] = 1. 26 e-3 10 x (-) 2. 90 • Calculating OH- from p. H • p. H p. OH [OH-] • SCE: What is OH- if p. H = 10. 325 p. H p. OH: 14 - 10. 325 = 3. 675 p. OH [OH-]: Inverse log[-3. 675] = 2. 11 e-4 =

Weak Acids and Conjugate Bases (difference is H+) HA(aq) + H 2 O(l) H 3 O+(aq) + A-(aq) Acid Base Conjugate Acid Conjugate Base (difference is H+) Eg. HC 2 H 3 O 2(aq) + H 2 O(l) H 3 O+(aq) + C 2 H 3 O 2 -(aq) Acid Base Conjugate Acid Conjugate Base Water (H 2 O) is amphoteric, it can function as an acid or a base. H 2 O(l) + H 2 O(l) Acid Base H 3 O+(aq) + OH-(aq) Conjugate Acid Conjugate Base

What is a Buffer and How does it Work? • A buffered solution is a mixture of a weak acid (HC 2 H 3 O 2), and its conjugate base (Na. C 2 H 3 O 2). • In a buffered solution of HC 2 H 3 O 2 and Na. C 2 H 3 O 2 adding a strong acid or base would react as follows… • HC 2 H 3 O 2 + OH- (from base) C 2 H 3 O 2 - + H 2 O or • C 2 H 3 O 2 - + H+ (from acid) HC 2 H 3 O 2 Neutralization
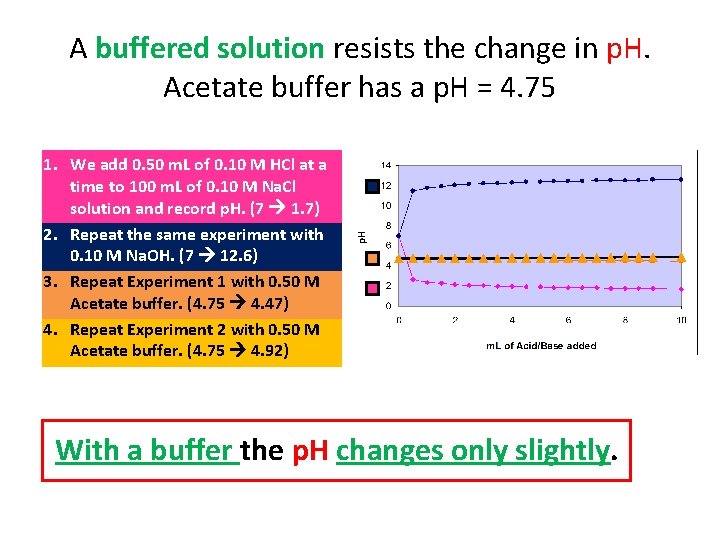
A buffered solution resists the change in p. H. Acetate buffer has a p. H = 4. 75 1. We add 0. 50 m. L of 0. 10 M HCl at a time to 100 m. L of 0. 10 M Na. Cl solution and record p. H. (7 1. 7) 2. Repeat the same experiment with 0. 10 M Na. OH. (7 12. 6) 3. Repeat Experiment 1 with 0. 50 M Acetate buffer. (4. 75 4. 47) 4. Repeat Experiment 2 with 0. 50 M Acetate buffer. (4. 75 4. 92) With a buffer the p. H changes only slightly.

Buffers in Biology • Blood has a normal p. H range of 7. 35 – 7. 45. • If blood falls below 6. 8 or rises above 7. 8 death may result. • Blood has a carbonic acid – bicarbonate conjugate pair buffer system. • Without a natural buffer, even eating grapefruit or lime (strongly acidic things) could be harmful to health.
- Slides: 13