Buffer calculations 0 842 g of propanoic acid

Buffer calculations

0. 842 g of propanoic acid is dissolved in water and mixed with 25. 0 m. L of 0. 108 mol L– 1 Na. OH. The final solution is made up to 100. 0 m. L with distilled water. M(CH 3 CH 2 COOH) = 74. 1 g mol– 1, p. Ka = 4. 87 a Calculate the p. H of the solution. b Calculate the new p. H if 1. 00 m. L of 1. 00 mol L– 1 Na. OH is added to the solution made above. Find the amount, in moles, of propanoic acid and Na. OH added to the mixture.
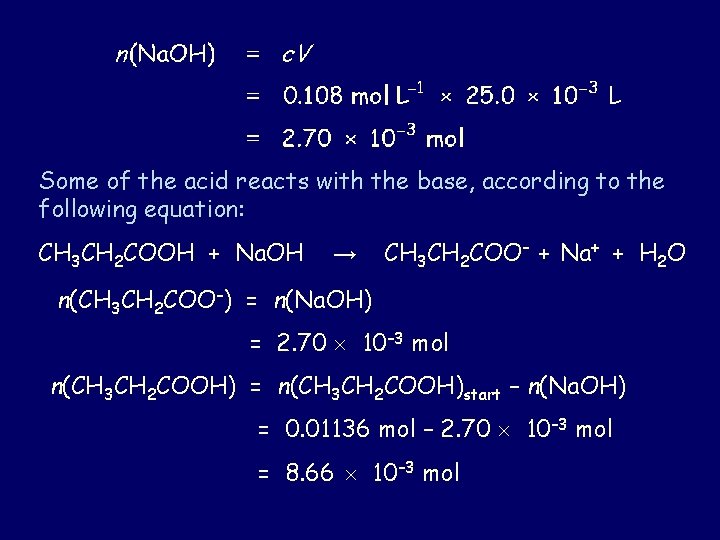
Some of the acid reacts with the base, according to the following equation: CH 3 CH 2 COOH + Na. OH → CH 3 CH 2 COO– + Na+ + H 2 O n(CH 3 CH 2 COO–) = n(Na. OH) = 2. 70 10– 3 mol n(CH 3 CH 2 COOH) = n(CH 3 CH 2 COOH)start – n(Na. OH) = 0. 01136 mol – 2. 70 10– 3 mol = 8. 66 10– 3 mol

To calculate the p. H we will need to derive the buffer formula: CH 3 CH 2 COOH + H 2 O CH 3 CH 2 COO– + H 3 O+

To convert the amounts (in moles) of acid and base present in the mixture into concentrations, we divide by the volume of liquid present. The volumes cancel — so we can ignore them and just work with the amounts of acid and base present.

Now to find the p. H when 1. 00 m. L of 1. 00 mol L– 1 Na. OH added. n(Na. OH)added = c. V = 1. 00 mol L– 1 1. 00 10– 3 L = 1. 00 10– 3 mol The Na. OH will convert CH 3 CH 2 COOH into CH 3 CH 2 COO–: n(CH 3 CH 2 COO–) = 2. 70 10– 3 mol + 1. 00 10– 3 mol = 3. 70 10– 3 mol n(CH 3 CH 2 COOH) = 8. 66 10– 3 mol – 1. 00 10– 3 mol = 7. 66 10– 3 mol

The volume of the solution is now 101. 0 m. L, but, as we have already seen, we can ignore the volume and simply work with the amounts of acid and base present.

Calculate for an ammonia buffer solution with a p. H of 10. 2. p. Ka(NH 4+) = 9. 24. Write the expression for Ka: NH 4+ + H 2 O NH 3 + H 2 O Rearrange it to form the required ratio:
![Calculate Ka and [H 3 O+] and hence calculate the ratio. Calculate Ka and [H 3 O+] and hence calculate the ratio.](http://slidetodoc.com/presentation_image_h/deb593fab8d9310086153d2a6d00d8d8/image-9.jpg)
Calculate Ka and [H 3 O+] and hence calculate the ratio.
- Slides: 9