Bubble nucleation in H 2 OCO 2 bearing

Bubble nucleation in H 2 O-CO 2 -bearing basaltic melts: results of high temperature decompression experiments Nolwenn Le Gall 1, Michel Pichavant 1 1 ISTO/CNRS, Orléans, France VUELCO conference Barcelona
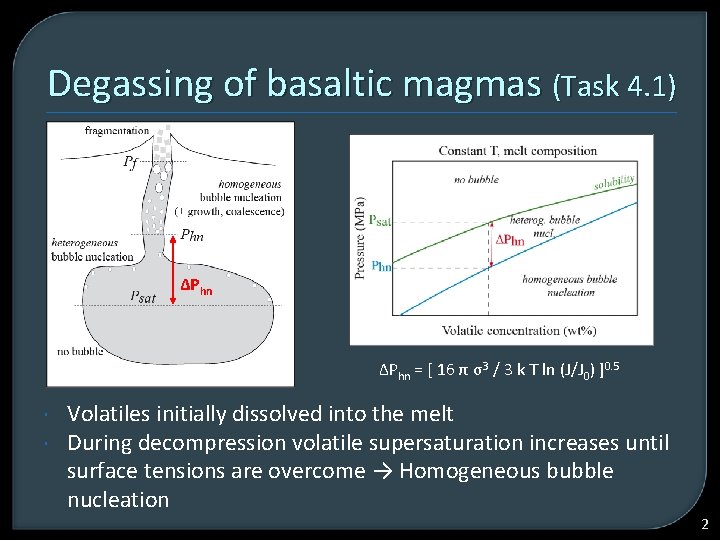
Degassing of basaltic magmas (Task 4. 1) ΔPhn = [ 16 π σ3 / 3 k T ln (J/J 0) ]0. 5 Volatiles initially dissolved into the melt During decompression volatile supersaturation increases until surface tensions are overcome → Homogeneous bubble nucleation 2
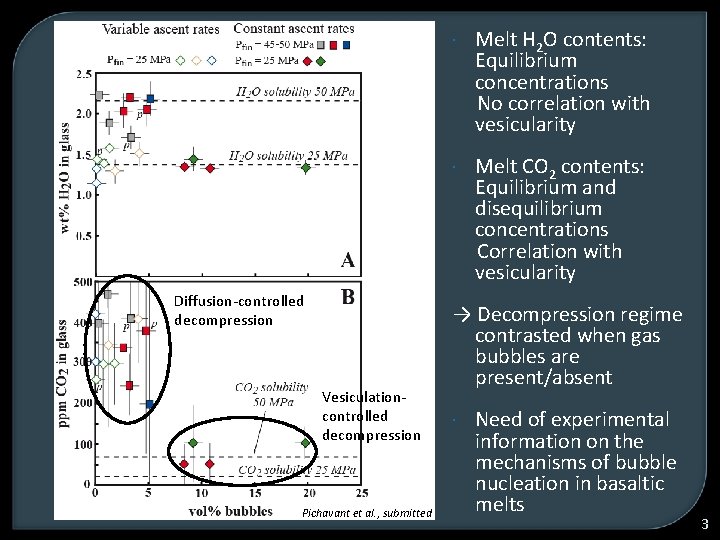
Diffusion-controlled decompression Vesiculationcontrolled decompression Pichavant et al. , submitted Melt H 2 O contents: Equilibrium concentrations No correlation with vesicularity Melt CO 2 contents: Equilibrium and disequilibrium concentrations Correlation with vesicularity → Decompression regime contrasted when gas bubbles are present/absent Need of experimental information on the mechanisms of bubble nucleation in basaltic melts 3
Experimental simulation of basaltic magma ascent and vesiculation… → To simulate the behaviour of H 2 O and CO 2 during degassing → To obtain information on the conditions of homogeneous bubble nucleation (=f(P, ascent rate, dissolved H 2 O/CO 2)) → To test equilibrium vs. disequilibrium degassing (interpretation of gas data) and explore the implications for the generation of explosive eruptions Times series of volcanic gas amounts and radiating source temperature measured during quiescent and explosive degassing on Stromboli (9 april 2002) Burton et al. , 2007 4

Experimental methodology: starting material preparation Starting material: natural basaltic pumice from Stromboli → Fusion: 3 h - 1 atm 1400°C → Glass coring: ø 2. 5 mm, l up to 10 mm 5

Experimental methodology: equipment and run procedure Synthesis of the H 2 O-CO 2 bearing basaltic glasses - Use of Au. Pd capsules - 3 different dissolved H 2 O/CO 2: XH 2 Oin. = 1 (#1) XH 2 Oin. = 0. 55 (#2) XH 2 Oin. = 0 (#3) Glass Distilled H 2 O Ag 2 C 2 O 4 (XH 2 Oin. = H 2 O / (H 2 O + CO 2)Charge) - > 40 h - 200 MPa - 1200°C 6
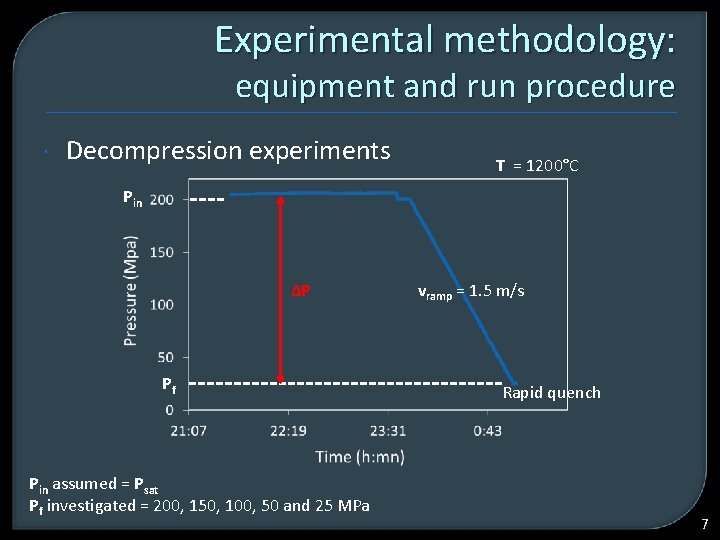
Experimental methodology: equipment and run procedure Decompression experiments T = 1200°C Pin ∆P Pf Pin assumed = Psat Pf investigated = 200, 150, 100, 50 and 25 MPa vramp = 1. 5 m/s Rapid quench 7
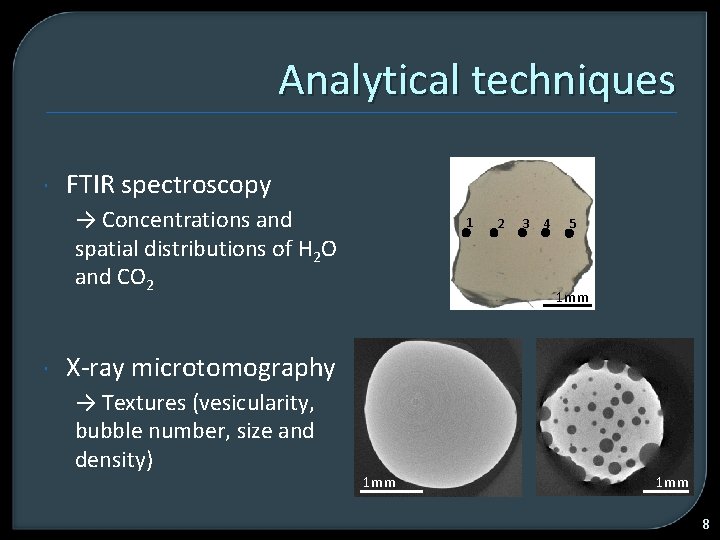
Analytical techniques FTIR spectroscopy → Concentrations and spatial distributions of H 2 O and CO 2 1 2 3 4 5 1 mm X-ray microtomography → Textures (vesicularity, bubble number, size and density) 1 mm 8

Experimental results: H 2 O and CO 2 concentrations of pre-decompression glasses Group #1 Group #2 5. 07 wt% 5. 03 wt% 5. 06 wt% 0 ppm P = 200 MPa T = 1200°C t = 50 h Group #3 0. 65 wt% 1033 ppm 2. 37 wt% 805 ppm 2. 36 wt% 886 ppm 2. 37 wt% 772 ppm 0. 65 wt% 995 ppm 0. 65 wt% 1052 ppm 1 mm Homogeneous volatile contents and distributions 3 compositional groups consistent with their XH 2 Oin conditions of synthesis 9
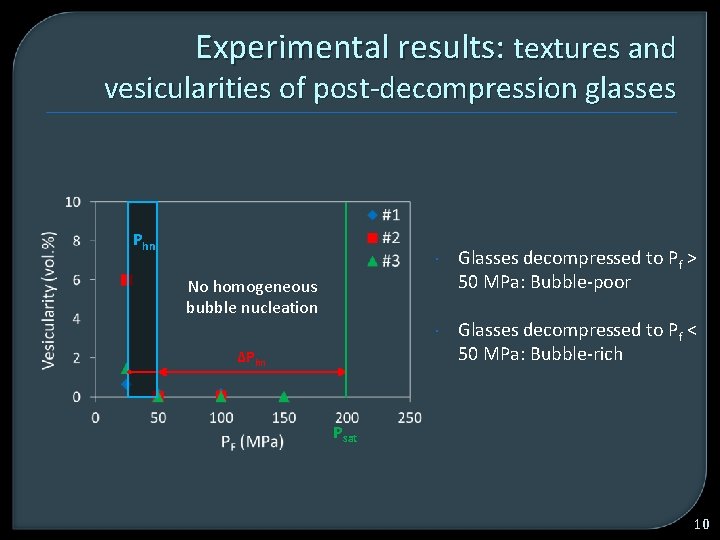
Experimental results: textures and vesicularities of post-decompression glasses Phn Glasses decompressed to Pf > 50 MPa: Bubble-poor Glasses decompressed to Pf < 50 MPa: Bubble-rich No homogeneous bubble nucleation ΔPhn Psat 10
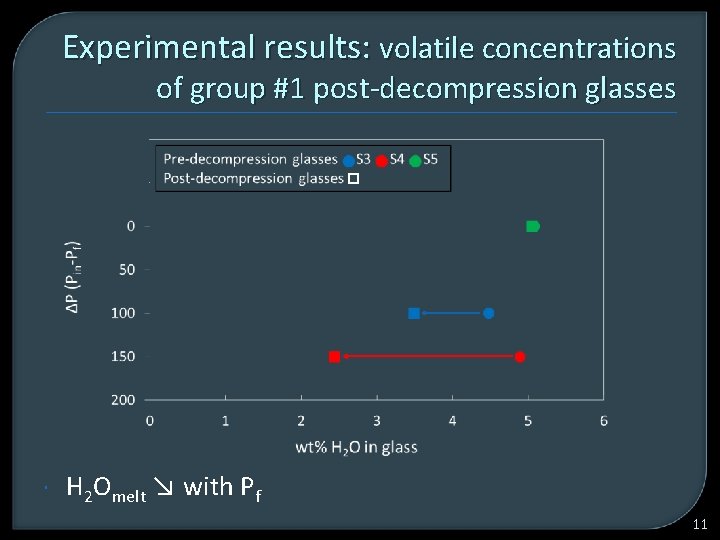
Experimental results: volatile concentrations of group #1 post-decompression glasses H 2 Omelt ↘ with Pf 11
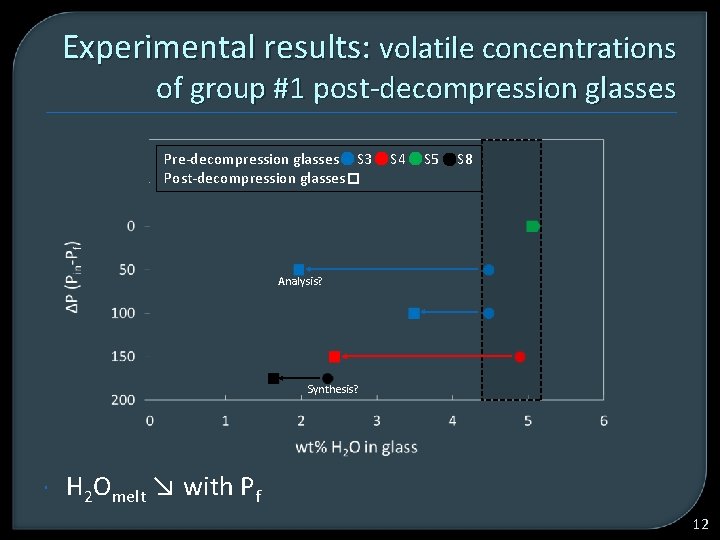
Experimental results: volatile concentrations of group #1 post-decompression glasses Pre-decompression glasses S 3 Post-decompression glasses S 4 S 5 S 8 Analysis? Synthesis? H 2 Omelt ↘ with Pf 12
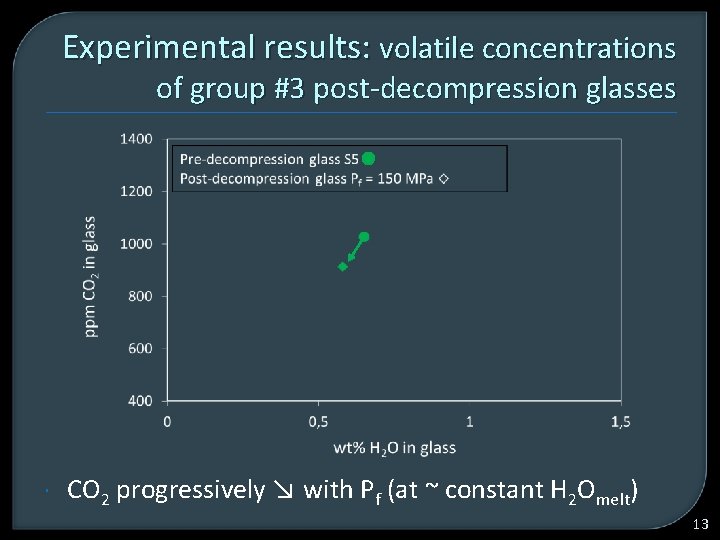
Experimental results: volatile concentrations of group #3 post-decompression glasses CO 2 progressively ↘ with Pf (at ~ constant H 2 Omelt) 13
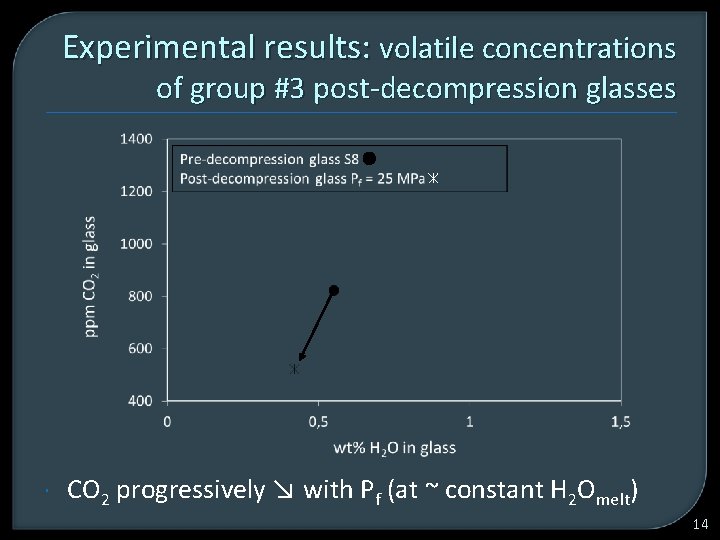
Experimental results: volatile concentrations of group #3 post-decompression glasses CO 2 progressively ↘ with Pf (at ~ constant H 2 Omelt) 14
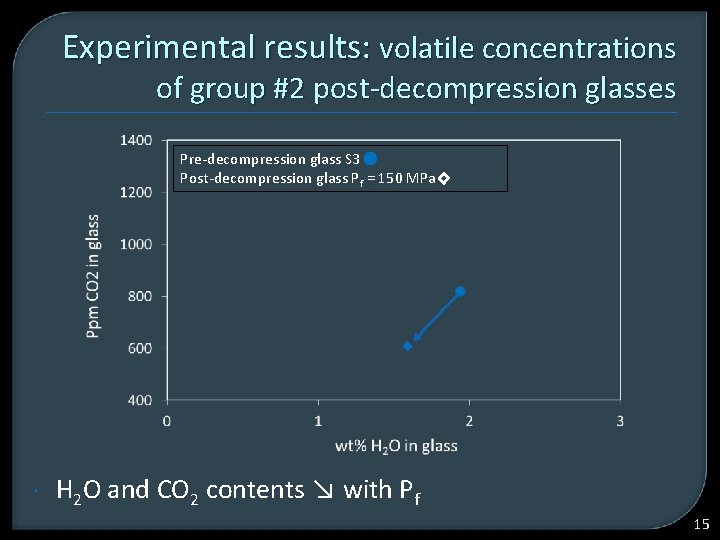
Experimental results: volatile concentrations of group #2 post-decompression glasses Pre-decompression glass S 3 Post-decompression glass Pf = 150 MPa H 2 O and CO 2 contents ↘ with Pf 15
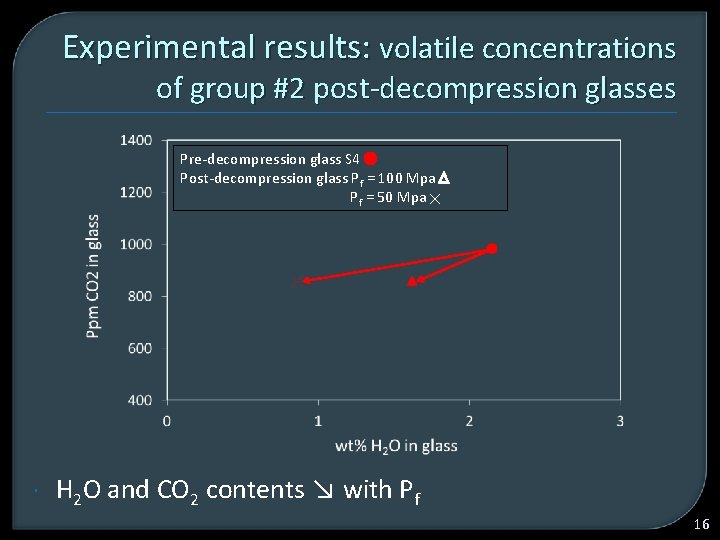
Experimental results: volatile concentrations of group #2 post-decompression glasses Pre-decompression glass S 4 Post-decompression glass Pf = 100 Mpa Pf = 50 Mpa H 2 O and CO 2 contents ↘ with Pf 16
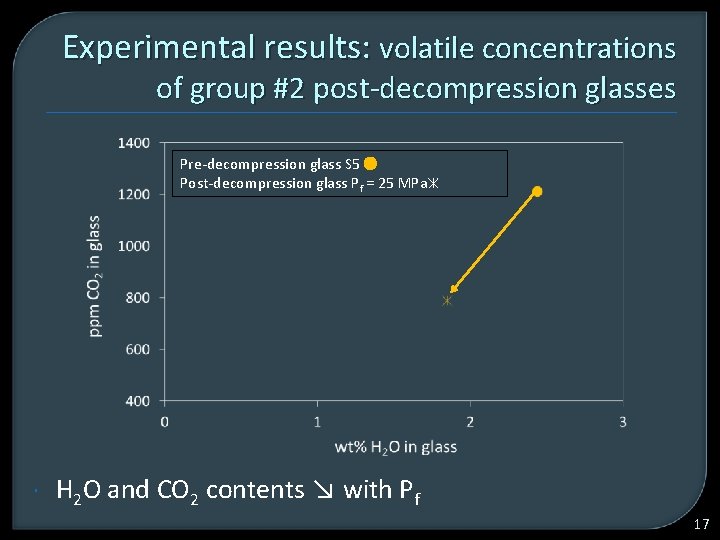
Experimental results: volatile concentrations of group #2 post-decompression glasses Pre-decompression glass S 5 Post-decompression glass Pf = 25 MPa H 2 O and CO 2 contents ↘ with Pf 17
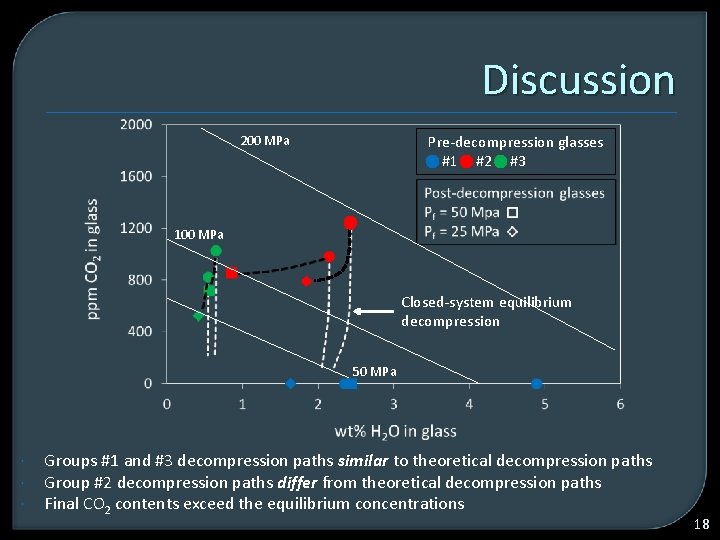
Discussion Pre-decompression glasses #1 #2 #3 200 MPa 100 MPa Closed-system equilibrium decompression 50 MPa Groups #1 and #3 decompression paths similar to theoretical decompression paths Group #2 decompression paths differ from theoretical decompression paths Final CO 2 contents exceed the equilibrium concentrations 18

Conclusions Homogeneous bubble nucleation occurs between 50 and 25 MPa → Large supersaturations are required: ∆Phn = 150 - 175 MPa Both equilibrium and disequilibrium decompression paths can be obtained Final glass compositions can be CO 2 supersaturated 19

Thank you for your attention 20
- Slides: 20