BTY 100 Lec3 3 Biobricks Created By Mamta

BTY 100 -Lec#3. 3 Biobricks Created By: Mamta Sharma

Outline Introduction to Biomolecules Proteins Carbohydrates Lipids
Biobricks. Biomolecules Proteins Carbohydrates Lipids Nucleic Acids

PROTEINS • Proteins are large biological molecules consisting of one or more chains of amino acids. • Proteins are biochemical compounds consisting of one or more polypeptides typically folded into a globular or fibrous form, facilitating a biological function. • It takes part in maintaining the structural Integrity of the cell, transport and storage of small molecules , catalysis, regulation, immune system.
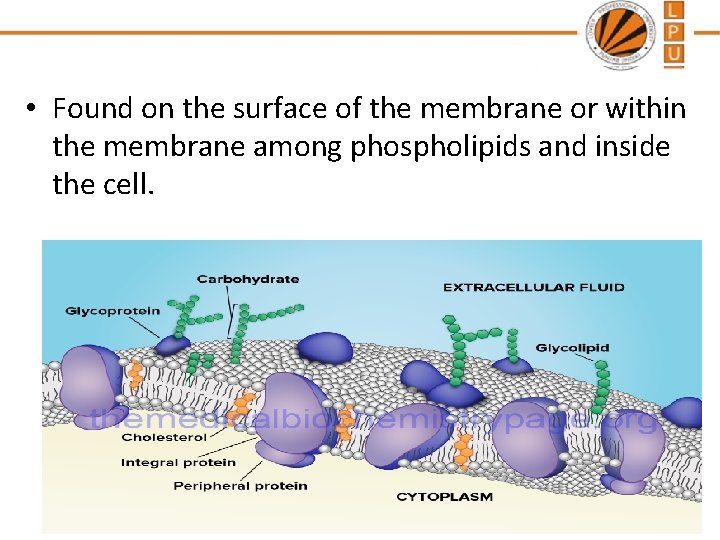
• Found on the surface of the membrane or within the membrane among phospholipids and inside the cell.
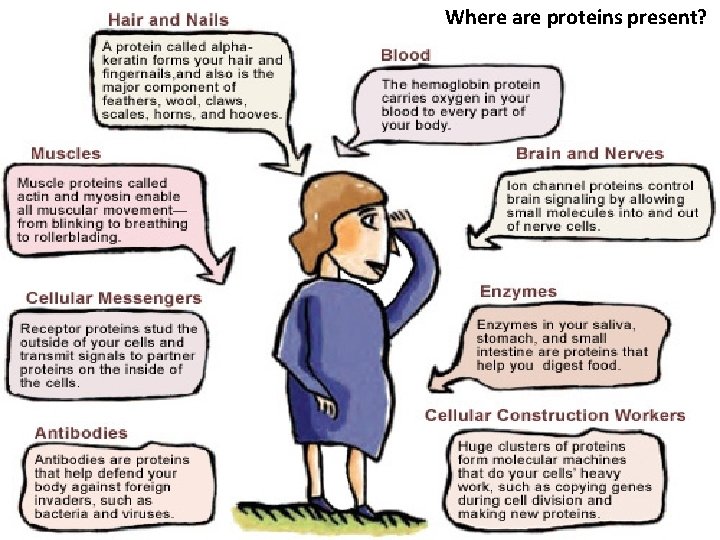
Where are proteins present?

Proteins classified by function • CATALYTIC: enzymes • STORAGE: ovalbumen (in eggs), casein (in milk), zein (in maize) • TRANSPORT: haemoglobin • COMMUNICATION: hormones (eg insulin) and neurotransmitters • CONTRACTILE: actin, myosin, dynein (in microtubules) • PROTECTIVE: Immunoglobulin, fibrinogen, blood clotting factors • TOXINS: snake venom • STRUCTURAL: cell membrane proteins, keratin (hair), collagen Make up about 15% of the cell
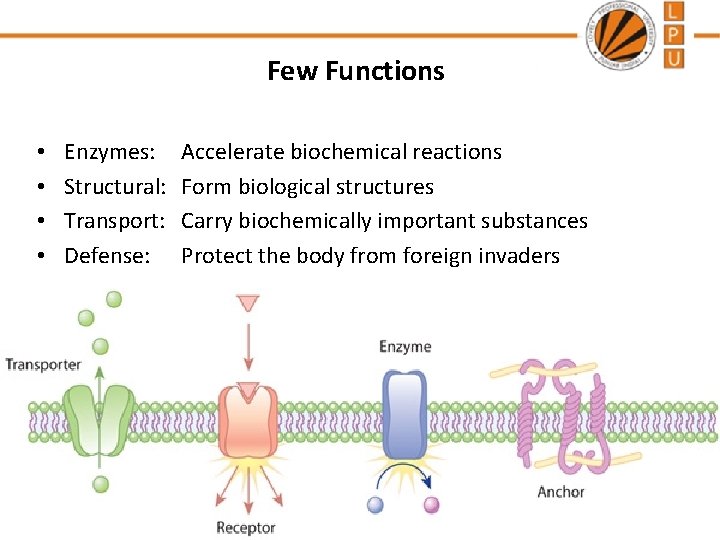
Few Functions • • Enzymes: Structural: Transport: Defense: Accelerate biochemical reactions Form biological structures Carry biochemically important substances Protect the body from foreign invaders
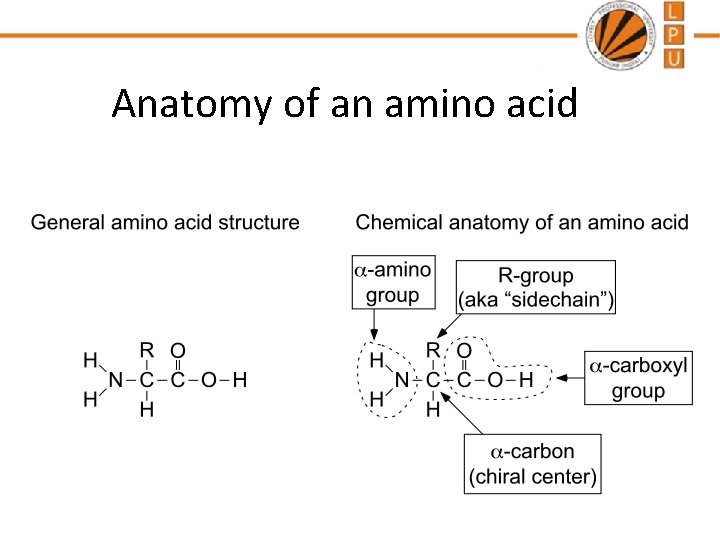
Anatomy of an amino acid
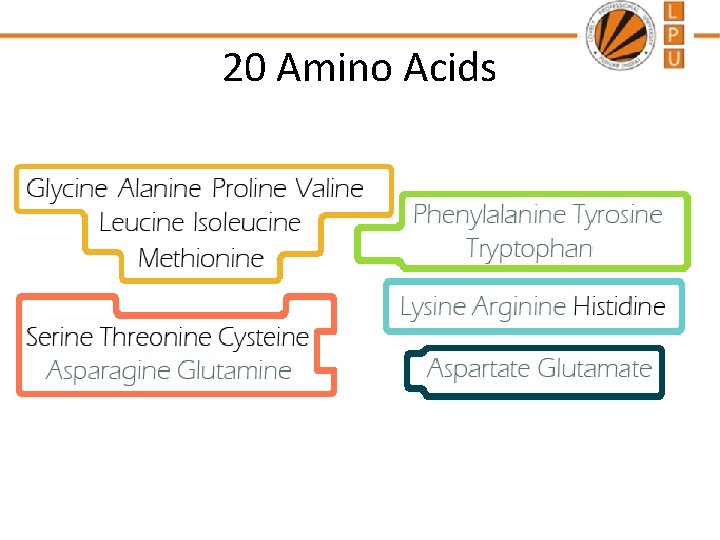
20 Amino Acids

• On the basis of nutritional value they have been categorized as essential and nonessential amino acids • Essential: which cannot be synthesized by the body or cannot be synthesized at an adequate rate and are obtained through diet. • Adults require 9 (Phenylalanine Valine Tryptophan Threonine Isoleucine Methionine Histidine Leucine Lysine) while infants and children require 10 ( arginine) • The remaining amino acids are Non essential, i. e those that can be synthesized by the body.

Amino Acids Are Joined By Peptide Bonds In Peptides - a-carboxyl of one amino acid is joined to aamino of a second amino acid (with removal of water) - only a-carboxyl and a-amino groups are used, not R-group carboxyl or amino groups
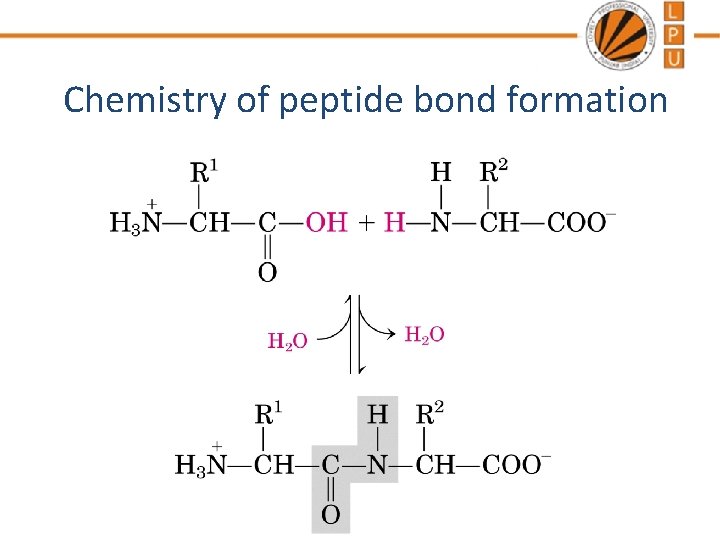
Chemistry of peptide bond formation
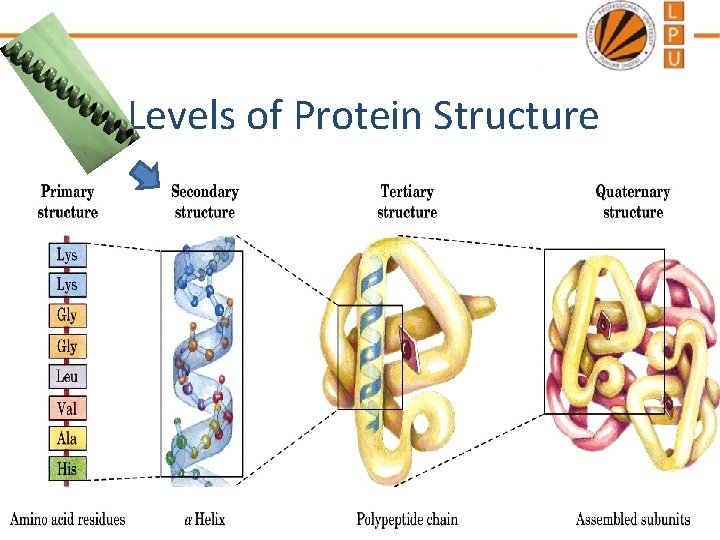
Levels of Protein Structure

Protein Denaturation • Denaturation is a process in which proteins lose the quaternary, tertiary structure and secondary structure. • At the molecular level, natural proteins are shaped like coils or springs. • When natural proteins are exposed to heat, salt, or acid, they denature—that is, their coils unwind.

Carbohydrates

Carbohydrates • Also called saccharides i. e. “sugars” such as glucose are synthesized in plants by photosynthesis. • Major source of energy in diet • Made up of C, H and O. • General formula- Cx(H 2 O)y, where x and y are variable numbers. • Major source of energy in consumers diet. • These are oxidized in living cells to produce CO 2, H 2 O and energy.

Classes • • Monosaccharides, Disaccharides Oligosaccharides Polysaccharides.

Monosaccharides • Monosaccharides are the single sugar units. • Their general formula is (CH 2 O)n. • They are classified according to the number of carbon atoms as trioses (3 C), tetroses (4 C), pentoses (5 C), hexoses (6 C) and heptoses (7 C). • Of these pentoses and hexoses are most common. • Monosaccharides act as intermediates in metabolism, and are also the primary energy source of the body.
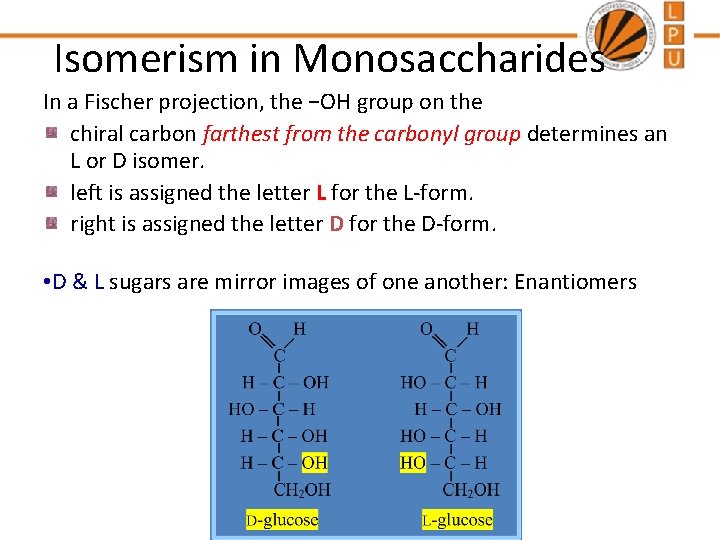
Isomerism in Monosaccharides In a Fischer projection, the −OH group on the chiral carbon farthest from the carbonyl group determines an L or D isomer. left is assigned the letter L for the L-form. right is assigned the letter D for the D-form. • D & L sugars are mirror images of one another: Enantiomers
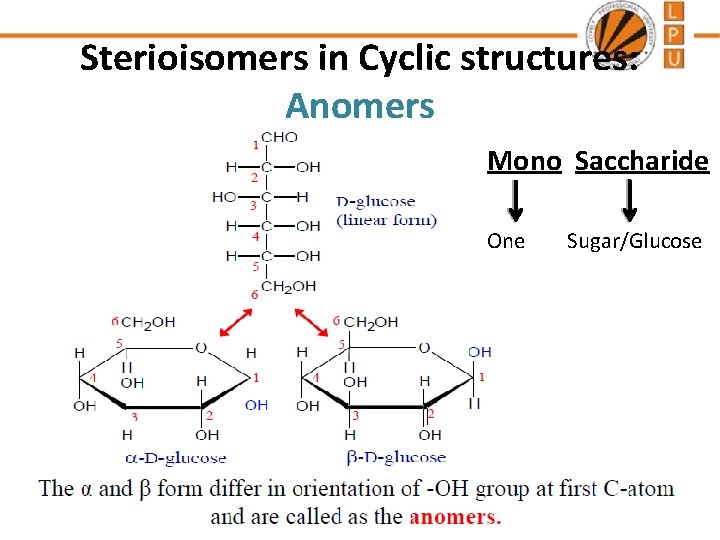
Sterioisomers in Cyclic structures: Anomers Mono Saccharide One Sugar/Glucose
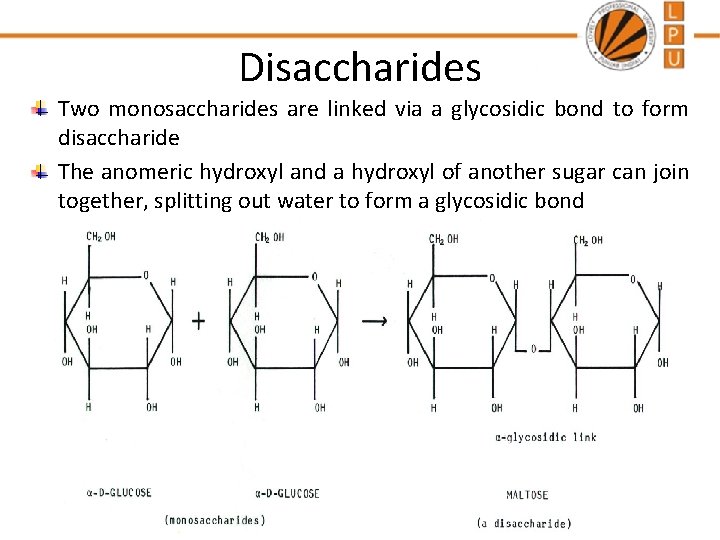
Disaccharides Two monosaccharides are linked via a glycosidic bond to form disaccharide The anomeric hydroxyl and a hydroxyl of another sugar can join together, splitting out water to form a glycosidic bond

Examples • Lactose is made up of glucose and galactose • Maltose is made up of two units of glucose • Sucrose is made up of glucose and fructose
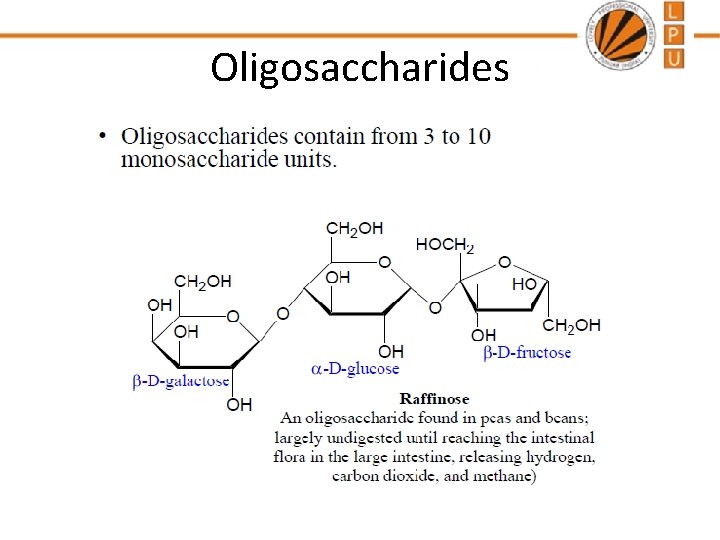
Oligosaccharides

Polysaccharides • Polysaccharides are the macromolecules formed by joining of many molecules of monosaccharides. • Example: Starch is a polymer of α-glucose, and is a major fuel store in plants. It has two components amylose (straightchain) and amylopectin(branched).

Lipids

Lipids • Lipids are a large and diverse group of naturally occurring organic compounds that are related by their solubility in nonpolar organic solvents and general insolubility in water. • They are formed by condensation reaction between fatty acids and alcohols. • Lipids are esters of moderate to long chain fatty acids • The main biological functions of lipids include energy storage, as structural components of cell membranes, and as important signalling molecules. • Lipids are non-polar (hydrophobic) compounds, soluble in organic solvents such as chloroform, methanol.
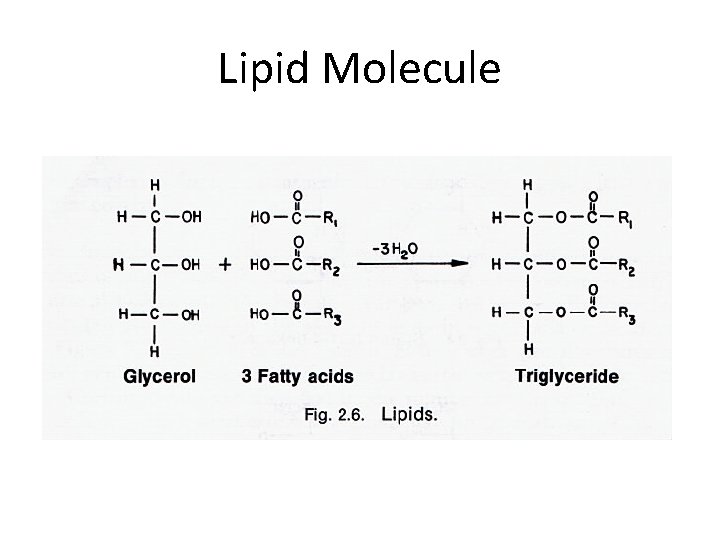
Lipid Molecule

Fatty Acids • The lipid building blocks. • Fatty acids consist of a hydrocarbon chain with a carboxylic acid at one end. • The fatty acid chains are usually between 10 and 20 Carbon atoms long. • The fatty "tail" is non-polar (Hydrophobic) while the Carboxyl "head" is a little polar (Hydrophillic). • Eg: A 16 -C fatty acid: CH 3(CH 2)14 -COO • The carbon chain of fatty acids may be saturated or unsaturated.
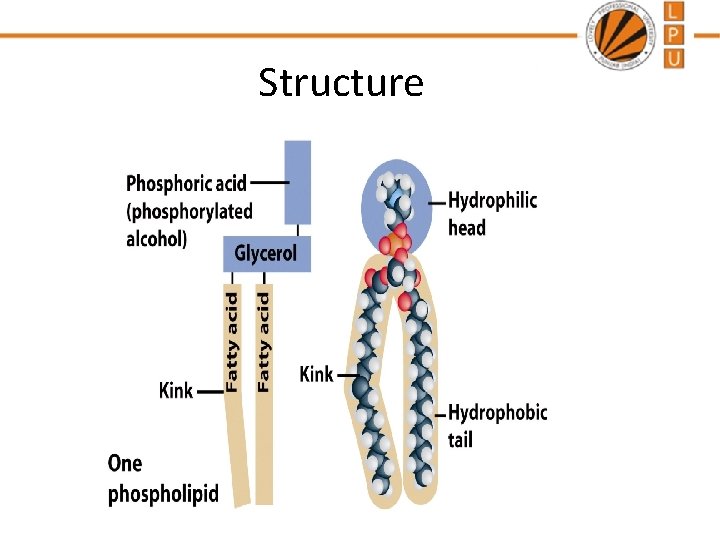
Structure

• Fatty acids are of two types: • Saturated : only single bonds between Carbon atoms • Unsaturated : contain one or more double bonds. • The double bond gives unsaturated fatty acids a bend in the chain. This bend affects the chemical characteristics of unsaturated fatty acids.

Saturated Fatty. Acids Contain only single C–C bonds Closely packed Strong attractions between chains High melting points Solids at room temperature Eg: Butter, Ghee

Unsaturated Fatty Acids Contain one or more double C=C bonds Nonlinear chains do not allow molecules to pack closely Few interactions between chains Low melting points Liquids at room temperature Eg: Vegetable oils

Which Fatty acids should you consume? ? ? • • Avoid saturated fats Avoid Trans Fats Avoid Cholesterol Add Omega fatty acids
Next Class: Genetic Basis of Life
- Slides: 35