BSG Guidelines for the Diagnosis and Treatment of

BSG Guidelines for the Diagnosis and Treatment of Cholangiocarcinoma By Matt Johnson

Background to Guidelines • Mortality rates from intrahepatic cholangiocarcinoma have steeply risen over the course of the last 30 y and continue to rise • 1998+9 = 1000 deaths / year • Men = Women • Previously no clear national consensus for optimal diagnosis and treatment

Risk Factors • Age (65% are >65 y) • PSC (lifetime risk = 5 -15%) • Chronic intraductal gall stones • Bile duct adenoma + biliary papillomatosis • Caroli’s disease (cystic dilatation of ducts) – Lifetime risk = 7% • Choledochal cysts (5% will transform with time) • Smoking • Chemical Exposure ( Thorotrast, aircraft, rubber) • Tropiocal (liver flukes, chronic typhoid carriers)

Anatomical Classification • A) Intrahepatic (20 -25%) • B) Perihilar (50 -60%) – “Klatskin” = involve the duct bifurcation – Many are coded as intrahepatic • C) Distal Extrahepatic (20 -25%) • D) Multifocal (5%)

Bismuth’s Perihilar Classification • Type 1 • • • – Below the confluence Type 2 – Involving the confluence but not the L or R duct Type 3 – Occluding the CHD and involving either the L (IIIa) or the R (IIIb) hepatic duct Type 4 – Multicentric or – Involve the CHD + both the L and the R hepatic ducts

Pathology • WHO Classification of Intrahepatic Carcinomas • Hepatocellular Ca • Combined Hepatocellular Cholangiocarcinoma • Cholangiocarcinoma, intrahepatic • Bile duct cystadenocarcinoma • Undifferentiated carcinoma

Pathology • WHO Classification of Extrahepatic bile duct carcinomas • • • Carcinoma in situ (* = not graded) Adenocarcinoma (95%, graded 1 -4 on glandular tissue) Papillary adenocarcinoma (*) Adenocarcinoma, intestinal type Mucinous adenocarcinoma Clear Cell adenocarcinoma (*) Signet ring adenocarcinoma (grade 3) Adeno-squamous carcinoma Squamous carcinoma (graded on degree of undifferentiation) Small cell carcinoma (“oat cell”) (grade 4) Undifferentiated carcinoma

Molecular Diagnosis • Inactivation of tumour suppressor genes – P 53, APC, Smad-4, bcl-2, p 16 • Mutations in Oncogenes – K-ras, C-myc, C-erb. B-2, C-neu • Chromasomal aneuploidy – 25% of perihilar tumours • These mutations can lead to phenotypic • changes Diagnostic and prognostic usefullness is unclear

Clinical Features • Obstructive Jaundice • RUQ pain, Fever + Rigors – Suggesting cholangitis • Systemic (malaise, weight loss, fatigue) • Deranged LFTs • Usually present late (esp. prox tumours)

Blood tests • • • Obstructive LFTs Transaminases (us. Normal, high with acute obstruction) Deficiency in Vit D, E, A, K (in chronic obstruction) Chronic Systemic Markers (Hb, Alb, LDH) CA 19 -9 (85%) – Can be elevated by obstruction alone – > 100 U/ml = sensitivity of 75% and specificity of 80% • CA 125 (40 -50%) – May signify the presence of peritoneal involvement) • CEA (30%)

Imaging • U/S • CT + • MRI + MRCP + MRA • Cholangiography (MRCP, ERCP, PTC) • EUS • PET • Intra ductal U/S, endoscopic/percutaneous flexible cholangioscopy, radiolabelled ligand imaging

Imaging • U/S – 1 st line for obstruction – Small lesions missed – Colour doppler can reveal compression/ thrombosis of the portal V or hepatic A • CT – Localises lymphadenopathy (NB. + PSC) – Enhanced spiral/ helical CT should be used if involvement of hilum or vascular system suspected • MRI – Optimal for anatomy + extent – MRCP for ductal involvement – MRA for hilar/vascular involvement

Imaging • Cholangiography – Essential for early diagnosis and assessing resectability – MRCP is non-invasive – ERCP (preferred) or PTC allows cytology and stenting – Cytology (+ in 30%) – Angiography will predict resectability NB = Biopsies should be avoided if resectability is possible
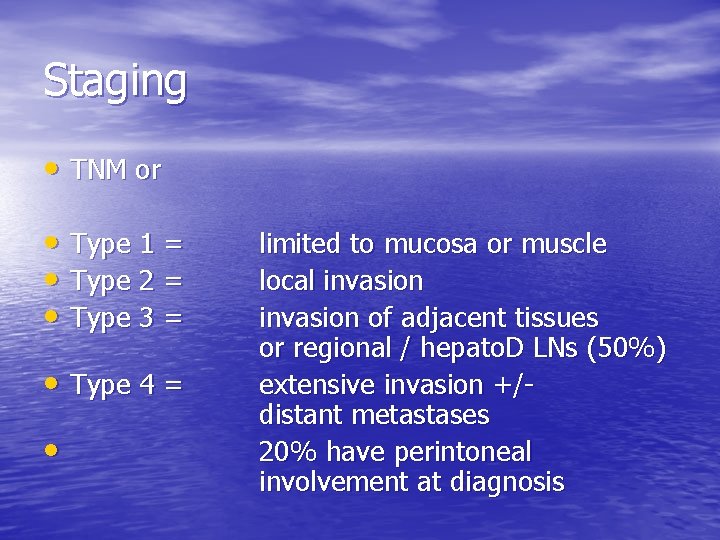
Staging • TNM or • Type 1 = • Type 2 = • Type 3 = • Type 4 = • limited to mucosa or muscle local invasion of adjacent tissues or regional / hepato. D LNs (50%) extensive invasion +/distant metastases 20% have perintoneal involvement at diagnosis

Imaging Recommendation • U/S (for initial screening) • CXR • MRI / MRCP – or contrast enhanced spiral /helical CT • Invasive Cholangiography – For tissue diagnosis +/- therapeutic decompression • Laparoscopy (if considered resectable)

Excluding Metastatic Disease • Metastatic adenocarcinoma can mimic cholangiocarcinoma • Pancreas = MRI, CT, EUS • Stomach = AXR, OGD • Breast = Ex, Mammography (if mass) • Lung = CXR • Colon = colonoscopy, spiral CT

Treatment – Curative Surgery • 5 y Survival for intrahepatic Ca = 9 -18% • 5 y Survival for proximal Ca = 9 -18% • 5 y Survival for distal Ca = 20 -30% • Survival depends on – stage with tumour free margins – absence of LN involvement • Median Survival – With hilar involvement = 12 -24/12 – Without hilar involvement = 18 -30/12

Surgery - OLTx • Contra-indicated due to rapid recurrence and death within 3 y • Survival may improve in some with chemoirradiation

Palliative Procedures • Stenting – Reduces sepsis – Improves survival – Surgical bypass has not proved superior • Irradiation • Intraoperative Coeliac plexus block

Resection Reporting • 1) Tumour – Type – Extent – Blood / lymphatic involvement – Perineural invasion (worse prognosis) • 2) Margins • 3) Regional LNs (peripancreatic = distant) • 4) Additional pathological findings (PSC) • 5) Metastases

Decompression • Pre-op biliary drainage / stenting is not advised if resection being considered • May be necessary in severely malnourished or in acute suppurative cholangitis • Preop placement of biliary catheters may be a helpful technical aid when dissecting a proximal Ca

Stenting • Complex Cholangio. Ca – MRCP will help planning management – ? Bilateral > unilateral • Plastic Vs Metal – Metal stents in those due to survive >6/12 – Metal = shorter hospital stay – Stenosis of metal stents can be treated with • Cotton-Leung plastic stent through lumen • Mesh metal stent • Semicovered stents (? reduce Ca ingrowth)

Oncology • 50% at presentation are LN+ • Treat patients early • Aim for symptom control via a multidisciplinary group as this translates into survival advantage • Aim to stabilize disease progress

Chemotherapy • • Response correlates to performance status at onset QOL is improved No evidence to support post-surgical adjuvant therapy No strong evidence for a survival benefit, but phase II trials suggest – Single agent 5 FU partial response = 10 -20% – Single agent gemcitabine partial response = 20 -30% – Combination partial response = 20 -40% – Gemcitabine + cisplatin partial response = 30 -50% • Downstaging can convert to operability

Radiotherapy • External Beam XRT (+Chemo. Tx) – – – Palliation of painful mets + bleeding No evidence for adjuvant post op XRT No improvement of QOL or Survival in advanced tumours – ? Chemoradiation • Local radiation – intra-operative or intra-luminal brachytherapy – No good data to support their use – could be promising

- Slides: 26