BSB 3503 Biomanufacturing CHAPTER 3 Biomanufacturing Processes Author

BSB 3503 - Biomanufacturing CHAPTER 3 Biomanufacturing Processes Author: Nurul Azyyati Sabri Co-Author / Editor: Rama Yusvana Faculty Industrial Sciences & Technology yusvana@ump. edu. my

Product Development & Regulatory Milestones • Include Preclinical, Clinical, Biologics License Application (BLA), and Non-disclosure Agreement (NDA) Submission • International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH) was formed in 1990 • Help biopharmaceutical entities to register and develop safe, quality, and effective drugs • Include therapeutic proteins such as monoclonal antibodies, cytokines, etc
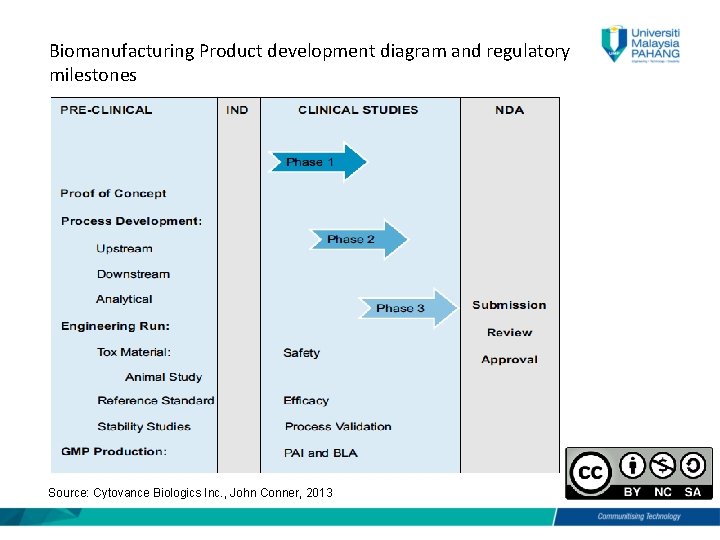
Biomanufacturing Product development diagram and regulatory milestones Source: Cytovance Biologics Inc. , John Conner, 2013

• Biologic products are derived from living systems that may or may not be modified • Mainly by Recombinant DNA technology to produce therapeutic products • Such as monoclonal antibody, a vaccine, cytokines, enzymes etc

• Some are naturally occurred such as whole blood and blood components, organ and tissue transplantations • Artificially via Recombinant DNA technology such as “insulin”, monoclonal antibodies, signaling-type proteins, and receptor-type proteins • Biologics can be defined as a large complex molecule produced by or extracted from living system such as bacteria or mammalin cells
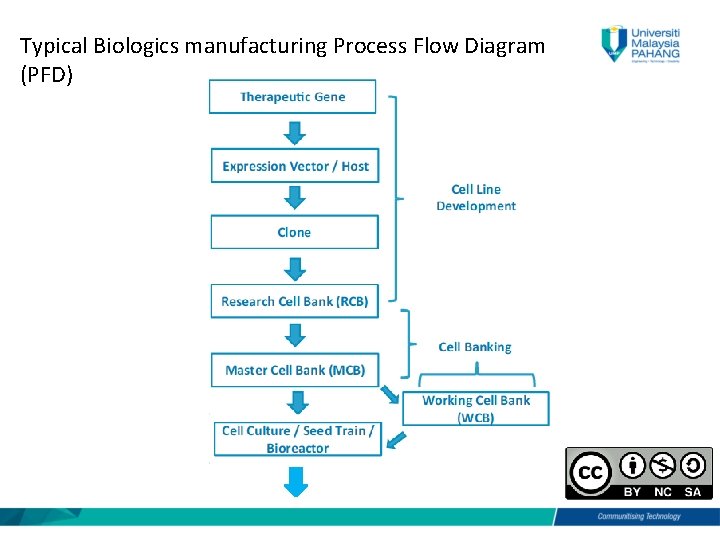
Typical Biologics manufacturing Process Flow Diagram (PFD)
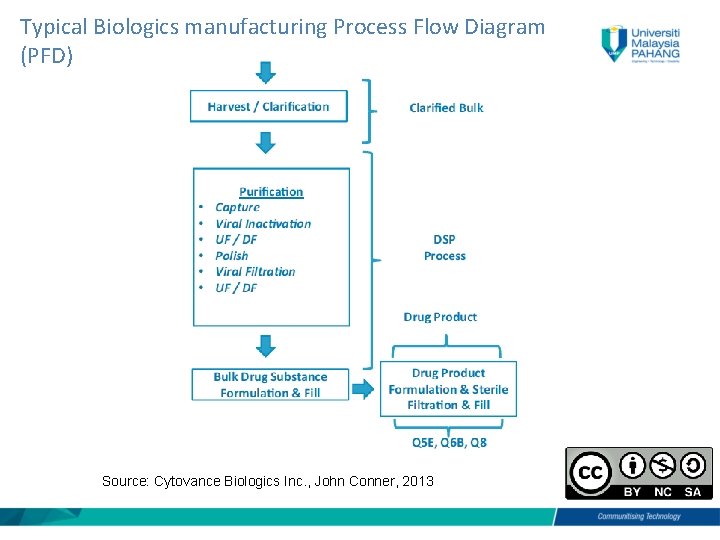
Typical Biologics manufacturing Process Flow Diagram (PFD) Source: Cytovance Biologics Inc. , John Conner, 2013
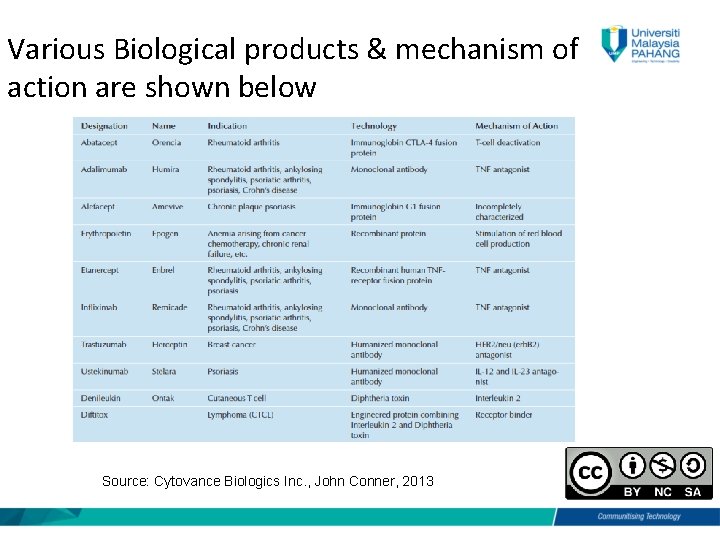
Various Biological products & mechanism of action are shown below Source: Cytovance Biologics Inc. , John Conner, 2013

Downstream Biomanufacturing During the downstream process, impurities are removed from the final end product that will be made available to consumers.
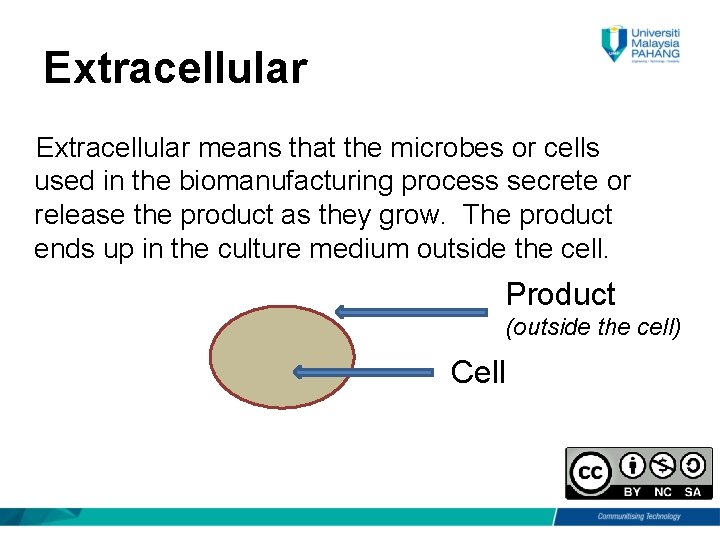
Extracellular means that the microbes or cells used in the biomanufacturing process secrete or release the product as they grow. The product ends up in the culture medium outside the cell. Product (outside the cell) Cell

Cell Separation The stage of downstream processing where the product is separated from the cells using a filter or centrifuge.
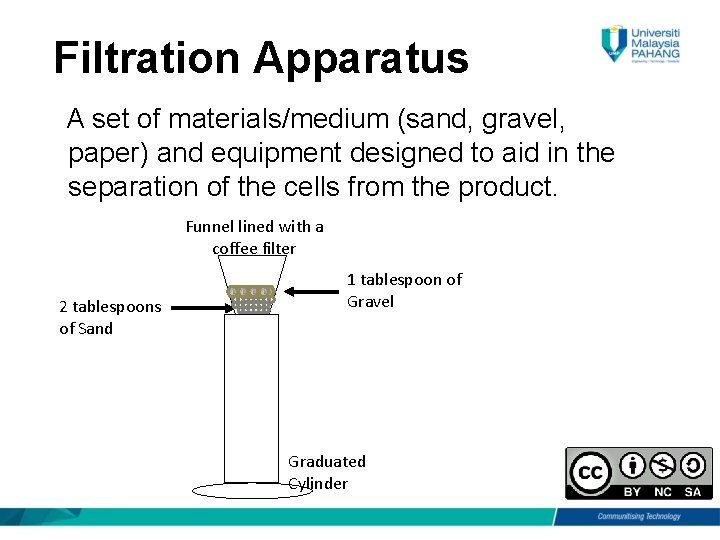
Filtration Apparatus A set of materials/medium (sand, gravel, paper) and equipment designed to aid in the separation of the cells from the product. Funnel lined with a coffee filter 2 tablespoons of Sand 1 tablespoon of Gravel Graduated Cylinder

Quality Control A system of monitoring the quality of the process by taking samples and testing their concentration and/or activity.

Yield Percentage A mathematical way to calculate how much product was recovered after the filtration process.
Standard Curve A standard curve is a method of plotting data in order to determine the level of product recovery over time.

Concentration Percentage A number based on comparing the color of the filtrate to a known sample or “standard curve. ”
- Slides: 16