Bronchial Asthma Nathir M Obeidat Bronchial Asthma Definition

Bronchial Asthma Nathir M Obeidat

Bronchial Asthma Definition Bronchial asthma is a clinical syndrome characterised by recurrent episodes of airway obstruction, which resolve spontaneously or as a result of treatment. The reversibility of the airway obstruction in asthma is the feature that distinguishes it from other forms of obstructive lung diseases.

Definition of Bronchial Asthma • A chronic inflammatory disease of the airways in which many cells play a role, in particular, mast cells, eosinophils and T lymphocytes. • In susceptible individuals this inflammation causes recurrent episodes of wheezing, breathlessness, chest tightness and cough, particularly at night and or early morning. • These symptoms are associated with airflow limitation that is at least partially reversible either spontaneously or with treatment.

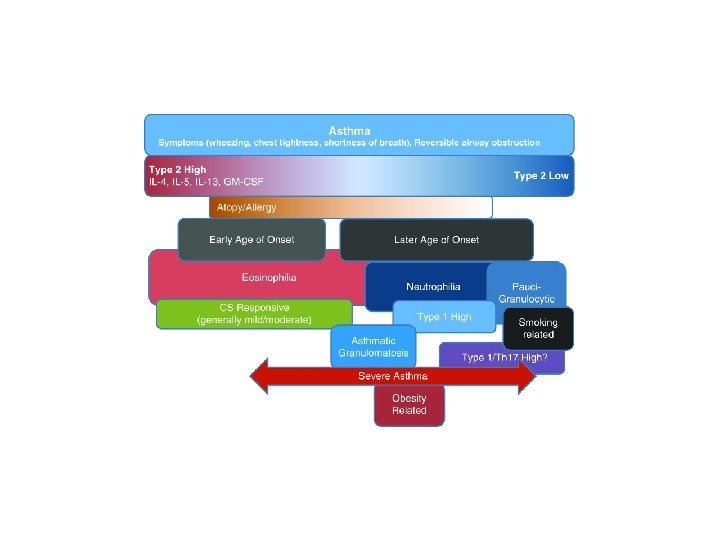
Asthma Phenotypes
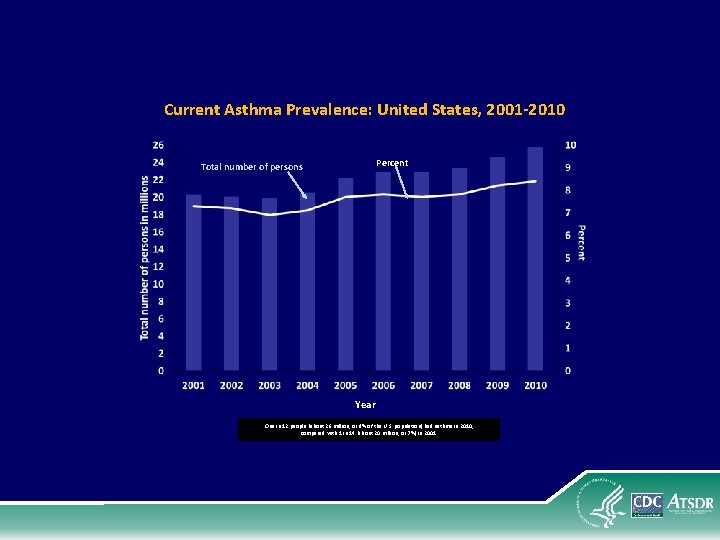
Current Asthma Prevalence: United States, 2001 -2010 Percent Total number of persons Year One in 12 people (about 26 million, or 8% of the U. S. population) had asthma in 2010, compared with 1 in 14 (about 20 million, or 7%) in 2001.

Risk Factors for Developing Asthma v Genetic characteristics v Occupational exposures v Environmental exposures

Risk Factors for Developing Asthma: Genetic Characteristics Atopy v. The body’s predisposition to develop an antibody called immunoglobulin E (Ig. E) in response to exposure to environmental allergens v. Can be measured in the blood v. Includes allergic rhinitis, asthma, hay fever, and eczema

Pathophysiology of Bronchial Asthma • It is a Complex interaction of cells and mediators, which lead to: 1 - Inflammation 2 - Bronchial hyperresponsiveness 3 - Airflow limitation 4 - Variability. 5 - Reversibility
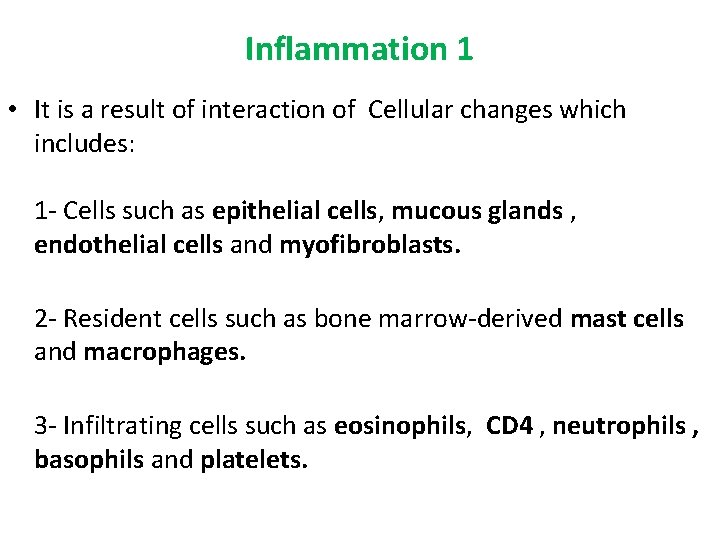
Inflammation 1 • It is a result of interaction of Cellular changes which includes: 1 - Cells such as epithelial cells, mucous glands , endothelial cells and myofibroblasts. 2 - Resident cells such as bone marrow-derived mast cells and macrophages. 3 - Infiltrating cells such as eosinophils, CD 4 , neutrophils , basophils and platelets.

Inflammation 2 - The above cells will lead to generation of mediators that can induce bronchoconstriction. - These mediators includes histamine , platelet activating factor, and some derivatives of arachidonic cascade such as PGD 2 , C 4 , D 4, E 4 (LTC 4, LTD 4 , LE 4). - Infiltration of airways by mast cells, eosinophils, activated T lymphocytes and neutrophils

Inflammation 3 - Mast cells as aresult of Ig. E – Mediated stimulation also release mediators - Cytokines most of which are product of lymphocytes and macrophages - Edema of the airway mucosa is due to inflammation and increased capillary permeability.

Inflammation 4 • Death from severe asthma usually occurs from blockage of airways by mucous plugging. • The presence of mucous plug is associated with hyperplasia and metaplasia of goblet cells

Airway Hyper responsiveness - It is an exaggerated bronchoconstrictive response by the airways to a variety of stimuli such as histamine , methacholine , cold air , and environmental irritants. - It is not known whether bronchial hyperreacyivity ( BHR) is acquired or is present at birth. - it is thought that inflammation is the stimulus for BHR - The degree of BHR usually correlates with the clinical severity of asthma

AIRWAY OBSTRUCTION Causes - Acute bronchoconstriction - Mucous plugging of airways - Bronchial wall edema - Inflammatory cell infiltration - Airway wall remodeling - Smooth muscle hypertrophy

Airflow limitation • Defined as FEV 1/FVC <75%. • During remission this can be normal. • Age Dependent. • Needs spirometry for assement of severity of the disease.

ATOPY • Defined as genetic susceptibility for developing immunoglobilin Ig. E directed to epitopes expressed on common environmental allergens such as dust mites , animal proteins, pollens, and fungi. • Atopic asthma has seasonal variation and avoidance of the offending antigens may result in dramatic improvement. • Atopy manifests as allergic asthma, allergic rhinitis , allergic sinusitis with eosinophilia and high serum Ig. E level

Variability • Defined as a diurnal variation of PEEFR of 20%. • This is diagnostic for asthma. • The degree of variability is proportional to the severity of asthma. • A high degree of variability signals unstable asthma that demands increased medications

Cost of asthma Direct Medical Care Indirect Costs Hospital stay Social security Intensive care Loss of work output Emergency department Loss of school days Primary care Medications GINA Guidelines 2010 Impact on individual / family / society

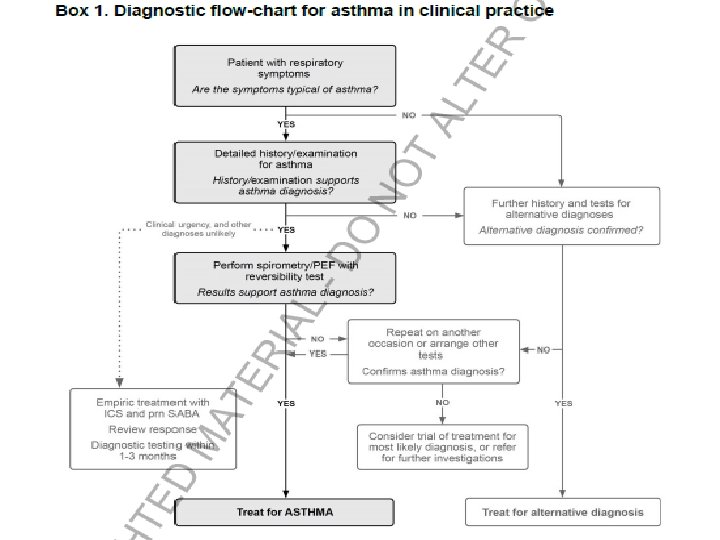
Diagnosis • History • Physical Examination • PFT - Vitalogram - Spirometry - FVL - Lung Volumes - DLCO - PFM - ABG

Diagnosis • CXR • Ig. E Level and Eosinophills

FVL • Location of the changes : - Above the line (Expiratory Arm) Or - Below the line (Inspiratory arm) • Shape of the Loop
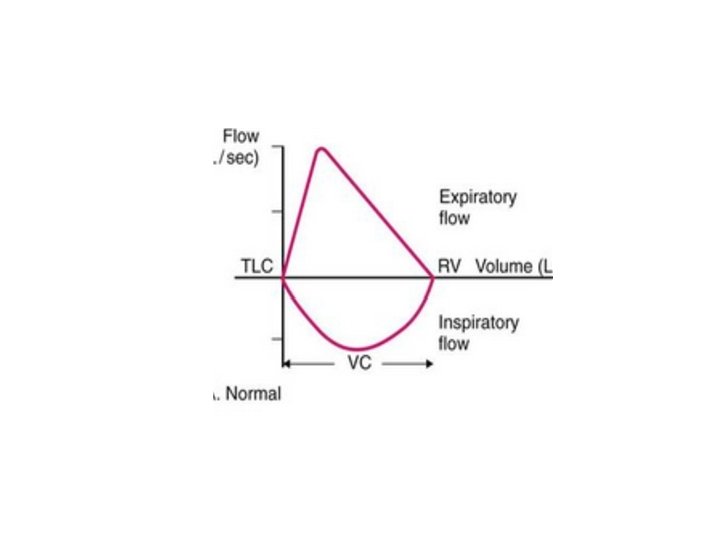
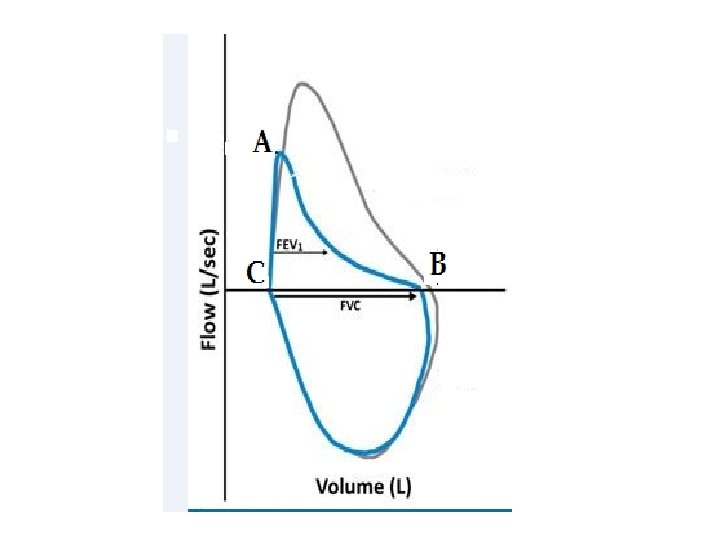
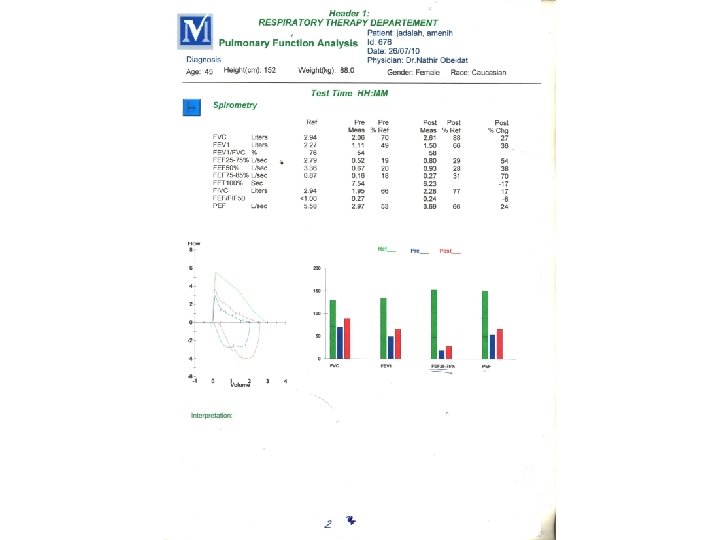
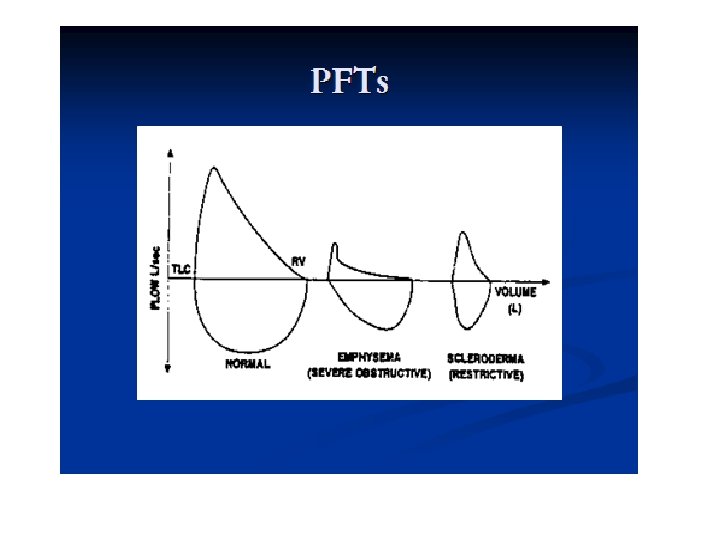


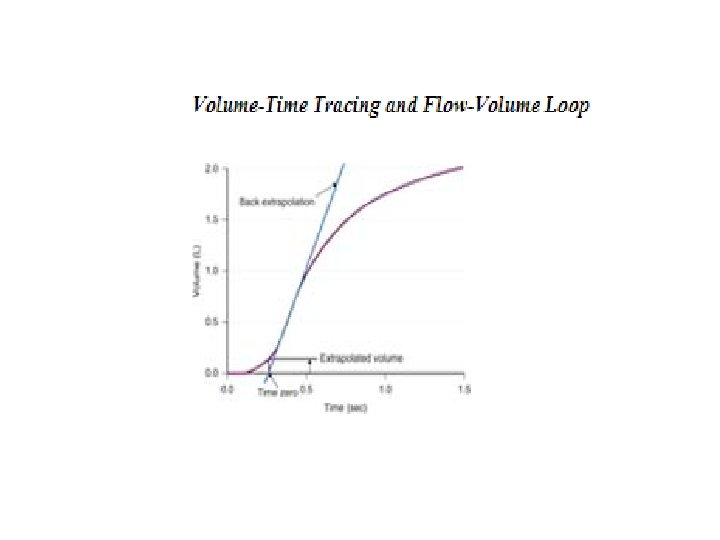
SPIROMETRY • • Volume-time tracing. Flow-volume tracing.

Asthma: Treatment options & guidelines

Goals of Treatment of Asthma • In spite of Immunotherapy, No real hope for cure of Bronchial Asthma. But • Treatment can achieve Asthma control and decrease exacerbations.

Treatment objectives • No asthma attacks • No emergency visits • Minimal need for quick relief (as needed) ß 2 -agonist • Maintain normal physical activity including exerc. No chronic symptoms ise • Maintain lung function as close to normal as possible • Minimal (or no) adverse effects from medicine GINA Guidelines 2010

Domains of Asthma Control Impairment of Function : 1 - Subjective Impairment: - Frequency and intensity of symptoms. - Limitations of daily activities. 2 - Objective Impairment: - Variations and Severity of Impairment of lung function. Future risk: - Likelihood of exacerbations. - Progressive loss of lung function. - Adverse effects from medications.
Control of airway inflammation BMJ. 2006 Apr 1; 332(7544): 767– 771

Is the Pathophysiological impact of Asthma limited to the Large Airways? Asthma Control Assessment Uncontrolled Incorrect Diagnosis Incorrect Inhaler Technique • Large vs Small Airway • Role of Small Airway Disease • Phenotypes Inadequate Treatment Adapted from Haughney J, Price D, Kaplan A, et al. . Respir Med. 2008; 102(12): 1681 -93. Poor Adherence fumarate

Pharmacological therapy Relievers • • Inhaled fast-acting -agonists : 2 Albuterol Fenoterol Inhaled anticholinergics • Ipratropium bromide Controllers Inhaled corticosteroids Inhaled long-acting 2 -agonists Inhaled cromones Oral anti-leukotrienes Oral theophyllines Oral corticosteroids

The choice of treatment should be guided by: Level of asthma control Current treatment Pharmacological properties and availability of the various forms of asthma treatment Economic considerations Cultural preferences and differing health care systems need to be considered
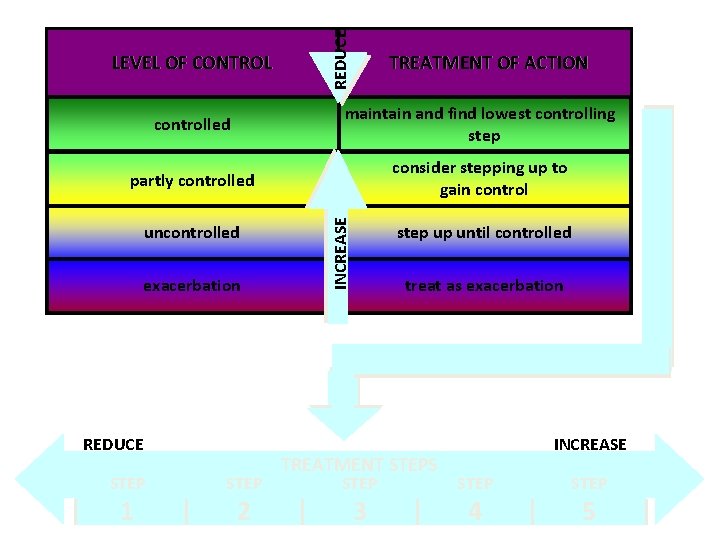
REDUCE LEVEL OF CONTROL TREATMENT OF ACTION maintain and find lowest controlling step partly controlled consider stepping up to gain control uncontrolled exacerbation REDUCE STEP 1 STEP 2 INCREASE controlled step up until controlled treat as exacerbation TREATMENT STEPS STEP 3 INCREASE STEP 4 STEP 5

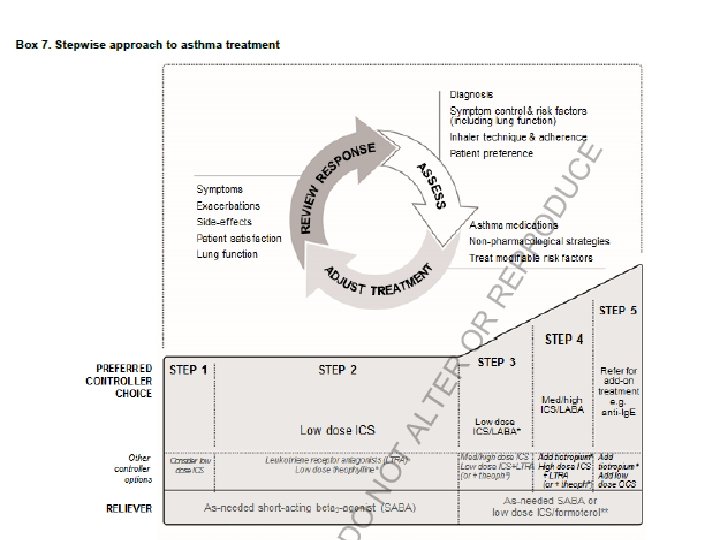
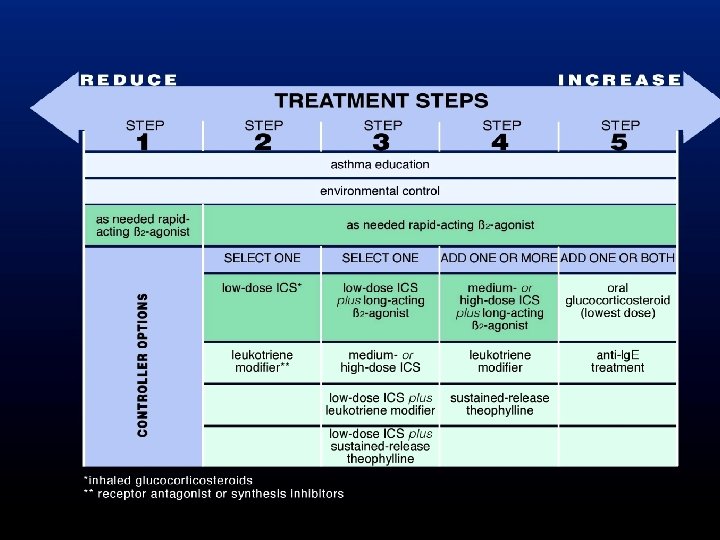

Beta 2 -Adrenoceptor agonists are the most powerful known bronchodilators and play a pivotal role in every step of this algorithm. Hanania N. Current Opinion in Pulmonary Medicine 2010,

Review Response and Adjusting the Dose • 2 -4 weeks after starting controller therapy. • 1 -3 months regular visit. • 4 -6 weeks regular visit in Pregnancy. • within 1 week after exacerbation. The frequency of visits depends on: 1. Initial levelof control 2. previous response to treatment. 3. His ability and willingness to be engaged in self – management plan.

Stepping Up Treatment • Sustained step up every 2 -3 months If symptoms persists or exacerbation in spite of 2 -3 months controller therapy. • Before stepping up consider: 1. Incorrect Inhaler Technique 2. Adherence 3. Modified risk ( Smoking) 4. Symptoms of comorbid condition(AR, GERD)

Day to Day Adjustment • By The Patient’s himself. • Smart Symbicort.

Stepping Down Approach • When Asthma is controlled Well. • Asthma controlled achieved for 3 months. • You aim to find the lowest dose of treatment that maintain: 1. symptoms free 2. no exacerbation 3. minimize side effects.

Step down Technique • Appropriate Timing ( no infection , no travell no Pregnancy). • Document the baseline , symptoms and PFT. • Reduce ICS dose by 20 -50% EVRY 2 -3 months

Treating Modifiable Risk Factoers • Decrease Exacerbation Risk by optimizing medications. • Self monitoring of symptoms and by using PEF. • Avoid Tobacco smoke Exposure. • Confirmed Food Allergy: Avoidance and availability of Epinephrine. • For Severe Asthma : Refer to especial centre.

Asthma Exacerbation • Exacerbations of asthma are episodes of progressive increase in shortness of breath, cough, wheezing, or chest tightness • Exacerbations are characterized by decreases in expiratory airflow that can be quantified and monitored by measurement of lung function (FEV 1 or PEF) • Severe exacerbations are potentially life-threatening and treatment requires close supervision

Asthma Exacerbations Management ü Primary therapies for exacerbations: • Repetitive administration of rapid-acting inhaled β 2 agonist • Early introduction of systemic glucocorticosteroids. • Oxygen supplementation Closely monitor response to treatment with serial measures of lung function GINA Guidelines 2010

Non Pharmacological Strategies and interventions • Smoking Cessation Advice. • Regular Physical Activities. • Avoid NSAIDs and Aspirin. • Occupational Asthma.

Treatment in special Population’s contexts • Pregnancy • Elderly • Allergic Rhinitis and Sinusitis • Obesity • GERD • Anxiety • Aspirin induced Respiratory distress.

Identifying patients at high risk of Asthma related death • Asthma needed Intubation and Ventilations. • Asthma Hospitalization or ICU care for Asthma in the last 12 months. • Currently not on ICS or poor inhaler adhirence. • currently using or recently stopping OCS. • Over use of SABA more than canister/month • Lack of written Asthma action plan. • confirmed food aleergy.
- Slides: 54