BromocriptineQR CYCLOSET Proposed mechanism of action Morning administration
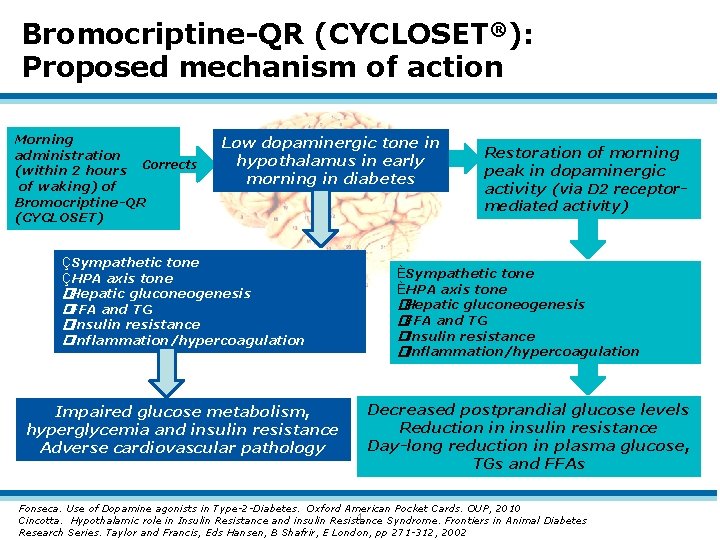
Bromocriptine-QR (CYCLOSET®): Proposed mechanism of action Morning administration (within 2 hours Corrects of waking) of Bromocriptine-QR (CYCLOSET) Low dopaminergic tone in hypothalamus in early morning in diabetes ÇSympathetic tone ÇHPA axis tone �Hepatic gluconeogenesis �FFA and TG �Insulin resistance �Inflammation/hypercoagulation Impaired glucose metabolism, hyperglycemia and insulin resistance Adverse cardiovascular pathology Restoration of morning peak in dopaminergic activity (via D 2 receptormediated activity) ÈSympathetic tone ÈHPA axis tone �Hepatic gluconeogenesis �FFA and TG �Insulin resistance �Inflammation/hypercoagulation Decreased postprandial glucose levels Reduction in insulin resistance Day-long reduction in plasma glucose, TGs and FFAs Fonseca. Use of Dopamine agonists in Type-2 -Diabetes. Oxford American Pocket Cards. OUP, 2010 1 Cincotta. Hypothalamic role in Insulin Resistance and insulin Resistance Syndrome. Frontiers in Animal Diabetes Research Series. Taylor and Francis, Eds Hansen, B Shafrir, E London, pp 271 -312, 2002

Effect of Bromocriptine-QR (CYCLOSET®) monotherapy on glycemic control in type 2 diabetes subjects • Phase 3 double-blind, placebo-controlled, randomized multicenter 24 week study Subjects – 154 obese (BMI=31. 6) type 2 diabetics – A 1 c=7. 5 -11. 0% (mean=8. 9%); FPG=208 mg/dl – Age=18 -70 years (mean=55 y) – No OHAs or insulin – No prescription sympathomimetic drugs – No rotating work schedule or night shift Experimental Design – Subjects admitted to CRC and given standardized breakfast, lunch, and dinner before and after 24 weeks of Bromocriptine-QR (CYCLOSET) (1. 6 -4. 8 mg/day) – Plasma glucose and insulin measured frequently from 7 am-7 pm
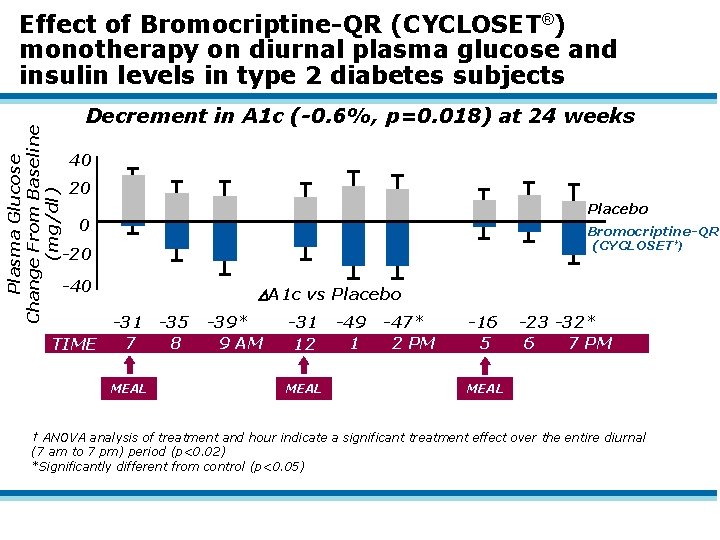
Plasma Glucose Change From Baseline (mg/dl) Effect of Bromocriptine-QR (CYCLOSET®) monotherapy on diurnal plasma glucose and insulin levels in type 2 diabetes subjects Decrement in A 1 c (-0. 6%, p=0. 018) at 24 weeks 40 20 Placebo 0 Bromocriptine-QR (CYCLOSET†) -20 -40 A 1 c vs Placebo -31 -35 8 7 TIME MEAL -39* 9 AM -31 12 MEAL -49 1 -47* 2 PM -16 5 -23 -32* 6 7 PM MEAL † ANOVA analysis of treatment and hour indicate a significant treatment effect over the entire diurnal (7 am to 7 pm) period (p<0. 02) *Significantly different from control (p<0. 05)
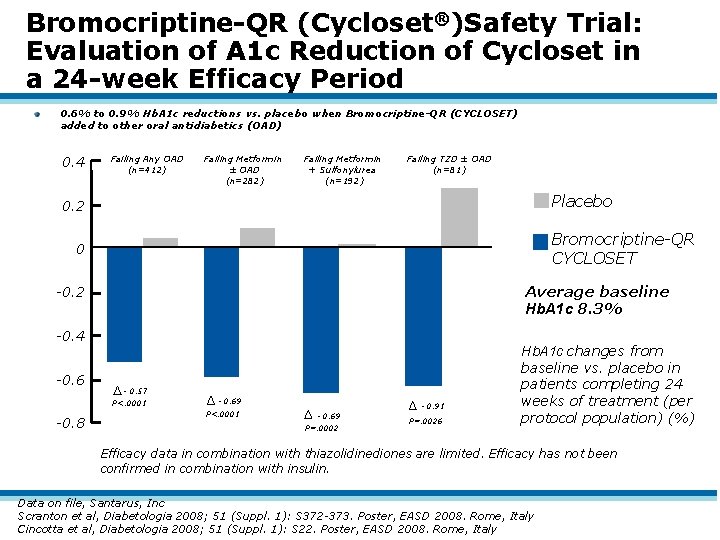
Bromocriptine-QR (Cycloset®)Safety Trial: Evaluation of A 1 c Reduction of Cycloset in a 24 -week Efficacy Period 0. 6% to 0. 9% Hb. A 1 c reductions vs. placebo when Bromocriptine-QR (CYCLOSET) added to other oral antidiabetics (OAD) 0. 4 Failing Any OAD (n=412) Failing Metformin ± OAD (n=282) Failing Metformin + Sulfonylurea (n=192) Failing TZD ± OAD (n=81) Placebo 0. 2 Bromocriptine-QR CYCLOSET 0 Average baseline Hb. A 1 c 8. 3% -0. 2 -0. 4 -0. 6 - 0. 57 P<. 0001 -0. 8 - 0. 69 P<. 0001 - 0. 69 P=. 0002 - 0. 91 P=. 0026 Hb. A 1 c changes from baseline vs. placebo in patients completing 24 weeks of treatment (per protocol population) (%) Efficacy data in combination with thiazolidinediones are limited. Efficacy has not been confirmed in combination with insulin. Data on file, Santarus, Inc Scranton et al, Diabetologia 2008; 51 (Suppl. 1): S 372 -373. Poster, EASD 2008. Rome, Italy Cincotta et al, Diabetologia 2008; 51 (Suppl. 1): S 22. Poster, EASD 2008. Rome, Italy
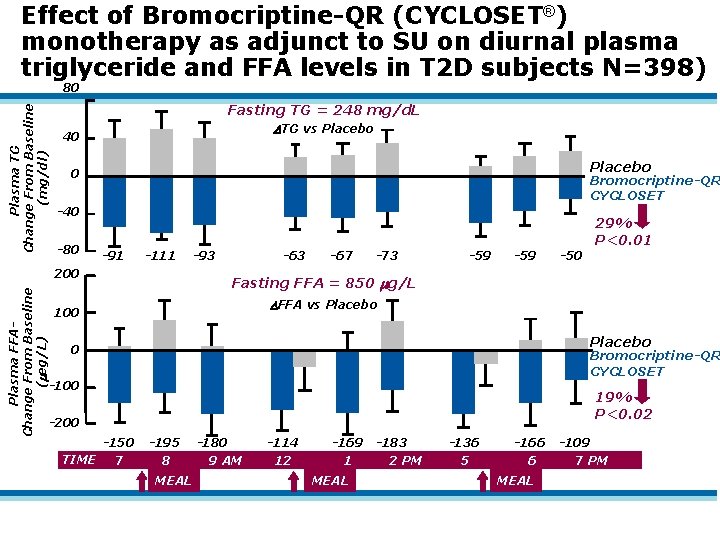
Effect of Bromocriptine-QR (CYCLOSET®) monotherapy as adjunct to SU on diurnal plasma triglyceride and FFA levels in T 2 D subjects N=398) Plasma TG Change From Baseline (mg/dl) 80 Fasting TG = 248 mg/d. L TG vs Placebo 40 Placebo 0 Bromocriptine-QR CYCLOSET -40 -80 29% P<0. 01 -91 -111 Plasma FFAChange From Baseline ( eg/L) 200 -93 -67 -73 -59 -50 Fasting FFA = 850 g/L FFA vs Placebo 100 Placebo 0 Bromocriptine-QR CYCLOSET -100 19% P<0. 02 -200 -150 TIME 7 -195 8 MEAL -180 9 AM -114 12 -169 1 MEAL -183 2 PM -136 5 -166 6 MEAL -109 7 PM
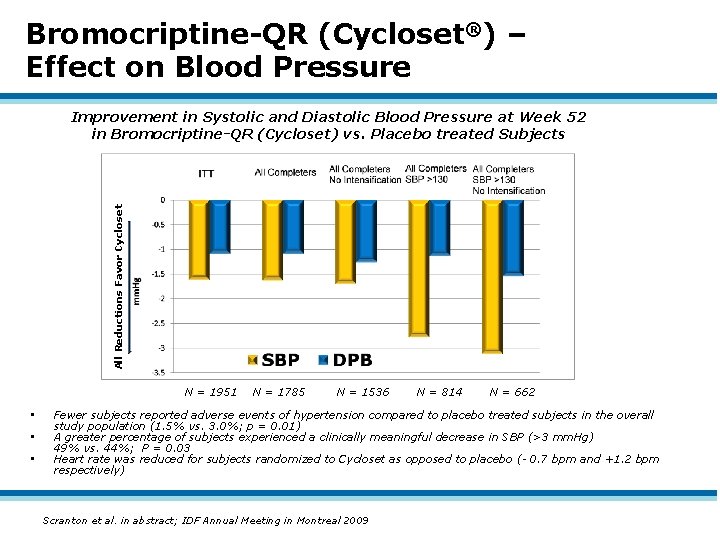
Bromocriptine-QR (Cycloset®) – Effect on Blood Pressure All Reductions Favor Cycloset Improvement in Systolic and Diastolic Blood Pressure at Week 52 in Bromocriptine-QR (Cycloset) vs. Placebo treated Subjects N = 1951 • • • N = 1785 N = 1536 N = 814 N = 662 Fewer subjects reported adverse events of hypertension compared to placebo treated subjects in the overall study population (1. 5% vs. 3. 0%; p = 0. 01) A greater percentage of subjects experienced a clinically meaningful decrease in SBP (>3 mm. Hg) 49% vs. 44%; P = 0. 03 Heart rate was reduced for subjects randomized to Cycloset as opposed to placebo (- 0. 7 bpm and +1. 2 bpm respectively) Scranton et al. in abstract; IDF Annual Meeting in Montreal 2009
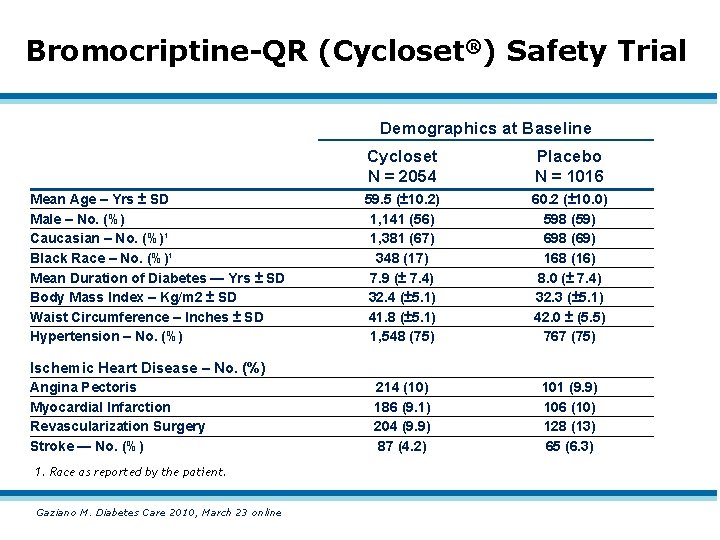
Bromocriptine-QR (Cycloset®) Safety Trial Demographics at Baseline Mean Age – Yrs ± SD Male – No. (%) Caucasian – No. (%)¹ Black Race – No. (%)¹ Mean Duration of Diabetes — Yrs ± SD Body Mass Index – Kg/m 2 ± SD Waist Circumference – Inches ± SD Hypertension – No. (%) Cycloset N = 2054 Placebo N = 1016 59. 5 (± 10. 2) 1, 141 (56) 1, 381 (67) 348 (17) 7. 9 (± 7. 4) 32. 4 (± 5. 1) 41. 8 (± 5. 1) 1, 548 (75) 60. 2 (± 10. 0) 598 (59) 698 (69) 168 (16) 8. 0 (± 7. 4) 32. 3 (± 5. 1) 42. 0 ± (5. 5) 767 (75) 214 (10) 186 (9. 1) 204 (9. 9) 87 (4. 2) 101 (9. 9) 106 (10) 128 (13) 65 (6. 3) Ischemic Heart Disease – No. (%) Angina Pectoris Myocardial Infarction Revascularization Surgery Stroke — No. (%) 1. Race as reported by the patient. Gaziano M. Diabetes Care 2010, March 23 online
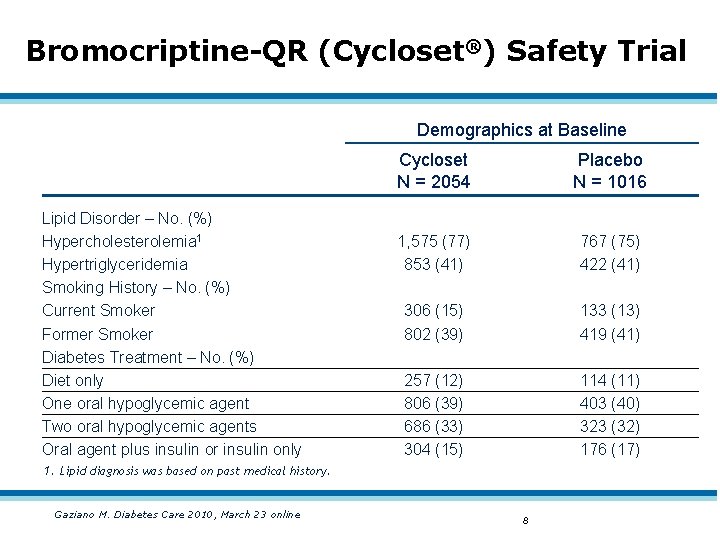
Bromocriptine-QR (Cycloset®) Safety Trial Demographics at Baseline Lipid Disorder – No. (%) Hypercholesterolemia 1 Hypertriglyceridemia Smoking History – No. (%) Current Smoker Former Smoker Diabetes Treatment – No. (%) Diet only One oral hypoglycemic agent Two oral hypoglycemic agents Oral agent plus insulin or insulin only Cycloset N = 2054 Placebo N = 1016 1, 575 (77) 853 (41) 767 (75) 422 (41) 306 (15) 802 (39) 133 (13) 419 (41) 257 (12) 806 (39) 686 (33) 304 (15) 114 (11) 403 (40) 323 (32) 176 (17) 1. Lipid diagnosis was based on past medical history. Gaziano M. Diabetes Care 2010, March 23 online 8
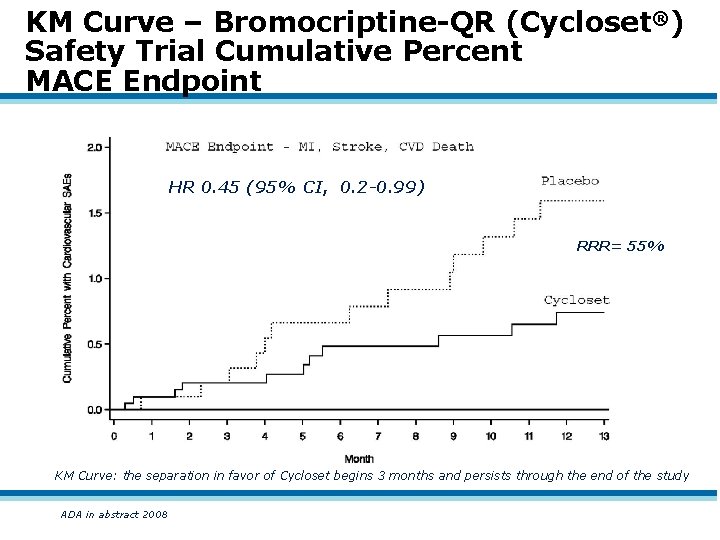
KM Curve – Bromocriptine-QR (Cycloset®) Safety Trial Cumulative Percent MACE Endpoint HR 0. 45 (95% CI, 0. 2 -0. 99) RRR= 55% KM Curve: the separation in favor of Cycloset begins 3 months and persists through the end of the study ADA in abstract 2008
- Slides: 9