BROMINATION OF TOLUENE Chemistry 318 Fall 2018 Schedule

BROMINATION OF TOLUENE Chemistry 318 Fall 2018

Schedule of day ■ PPE and Pre-lab check – at the door ■ Quiz ■ Recitation ■ Safety – Put bags away – Goggles – Gloves – Lab Coat ■ Lab part 1 ■ IR Review ■ Lab part 2

Due Dates ■ Today – Notebook copies of Bromination of Toluene before leaving the lab ■ Next Week – Spectroscopy Problem Set (part I & part II #1) – At beginning of lab – Bromination of Toluene Report

In Lab Today ■ Review of Electrophilic Aromatic Substitution ■ Bromination of toluene experiment ■ Review of IR Spectroscopy ■ Demonstration of FT-IR; salt plates; %Transmittance vs. Absorption mode

Notebook Keeping ■ Notebooks should be prepared according to the instructions in the Manual pp. 1 – 20. ■ For this experiment, a Data Table that includes chemical safety notes, MW, density, mass and calculated moles (as measured during lab) is appropriate. –Information such as bpt. , mpt. , density, MW, etc. should always be entered in your notebook as part of your pre-lab and obtained from approved sources. See the Lab Manual “Citation” section. No Wikipedia – No Chembook (the Wikipedia of the chemistry world) – no web-based information.

Notebook Keeping (cont. ) ■ Record all observations – everything you see, smell, feel (temperature changes), etc. Be clear and specific in your descriptions. ■ All calculations (in your notebook and on your report) must be clearly shown and labeled – show your work and pay attention to units and significant digits.

Experiment Notes n Bromine (Br 2) is a hazardous and extremely corrosive substance − Lab coat, gloves, and goggles at all times! Change gloves as necessary. − Keep vials in hood and prepare salt plates there n. Your instructor will handle the toluene and bromine. n. Preparation of staple: sand it vigorously to remove plastic coating. Bend to fit in bottom of vial. n. Moisten the p. H paper before inserting it in the air space above the liquid in the vial.

Experiment Notes n During the (forty-five minute) wait time we will review spectroscopy. n Do not change any setting on the IR. n Na. Cl plates are very fragile and expensive. Handle with great care and only by the edges.
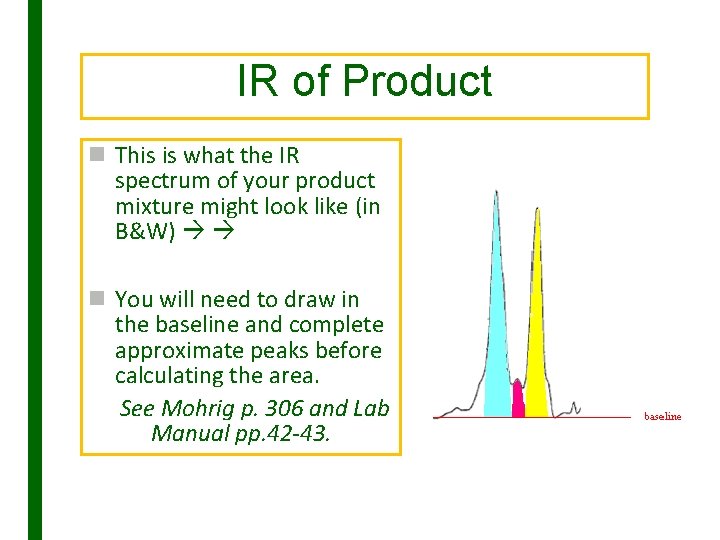
IR of Product n This is what the IR spectrum of your product mixture might look like (in B&W) n You will need to draw in the baseline and complete approximate peaks before calculating the area. See Mohrig p. 306 and Lab Manual pp. 42 -43.

Peak Area by Triangulation Peak Area = h * w½ * 1. 064 Where h = Peak Height w½ = width of peak at ½ the peak height Note: The curve is assumed Gaussian

Lab Report & Forms ■ See the Report Form for Bromination of Toluene in the Lab Manual (page 163). ■ Follow the instructions for writing reports found at the end of this experiment in the Lab Manual and on Black. Board under “Notebook & Report Formats” and “Lab Reports-in General”. ■ Assemble and staple your report in this order: – – Report Form Calculations IR spectrum Written report

Electrophilic Aromatic Substitution See the Solomons lecture text for an indepth discussion of Electrophilic Aromatic Substitution.
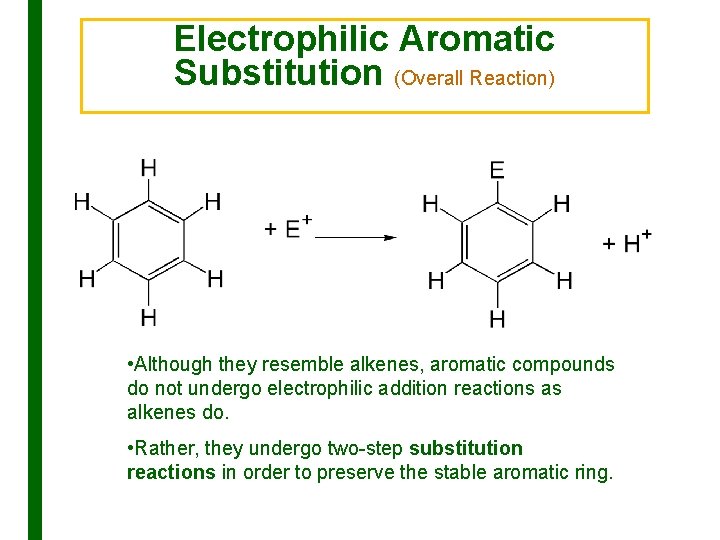
Electrophilic Aromatic Substitution (Overall Reaction) • Although they resemble alkenes, aromatic compounds do not undergo electrophilic addition reactions as alkenes do. • Rather, they undergo two-step substitution reactions in order to preserve the stable aromatic ring.
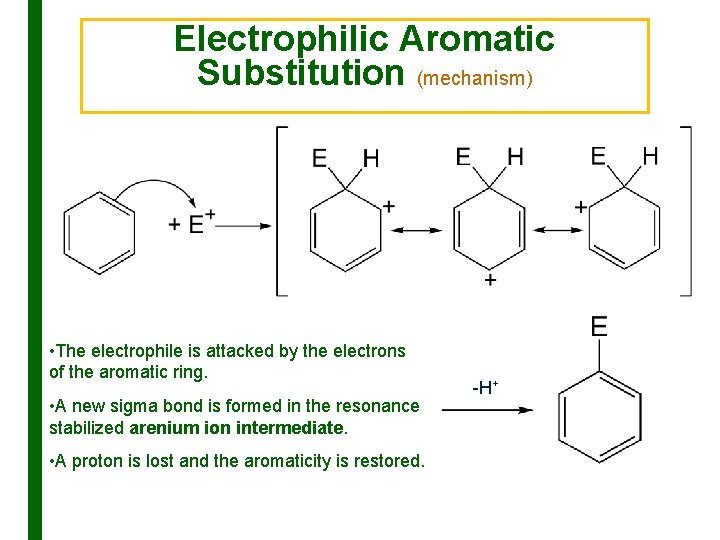
Electrophilic Aromatic Substitution (mechanism) • The electrophile is attacked by the electrons of the aromatic ring. • A new sigma bond is formed in the resonance stabilized arenium ion intermediate. • A proton is lost and the aromaticity is restored. -H+

Electrophilic Aromatic Substitution (mechanism) ■ Electrophiles (electron deficient species; Lewis acids), are attacked by the aromatic sextet of pi electrons in the aromatic ring. ■ An arenium ion intermediate is formed in which the ring has lost its aromaticity. ■ The arenium ion breaks the sigma bond to H+, and the electron pair becomes a pair of pi electrons in the aromatic ring. ■ There are several substitution reagents used in organic chemistry synthesis: − − − Halogenation – X 2 (X = Cl, Br) Nitration – HONO 2/H 2 SO 4 Sulfonation – SO 3/H 2 SO 4 Friedel-Crafts Alkylation – RCl/Al. Cl 3 Friedel-Crafts Acylation – R(C=O)Cl), Al. Cl 3

Electrophilic Aromatic Substitution Bromination ■ In bromination of arenes, the electrophilic bromine is generated by reacting Br-Br with a Lewis acid metal catalyst. ■ A common catalyst is Fe. Br 3. However, this substance can be produced by the very fast reaction between Fe 0 and Br 2 (as in this experiment). ■ The electrophile is likely to be the very electrophilic Brd+ in the catalytic complex [Br─Br. Fe. Br 3], although frequently Br+ itself is shown in textbooks.

Electrophilic Aromatic Substitution Reactivity & Orientation ■ The reactivity of BENZENE itself (no substituent) is used as a reference compound. ■ When there is a substituent group (G) on the benzene ring, the substituent determines both the position and the rate of substitution of the INCOMING electrophile. ■ Substituents affect which position (orientation ) on the ring is attacked by the electrophile in the reaction (o-, m-, or p-). ■ Substituents also affect how fast the electrophile attacks the ring (reactivity).
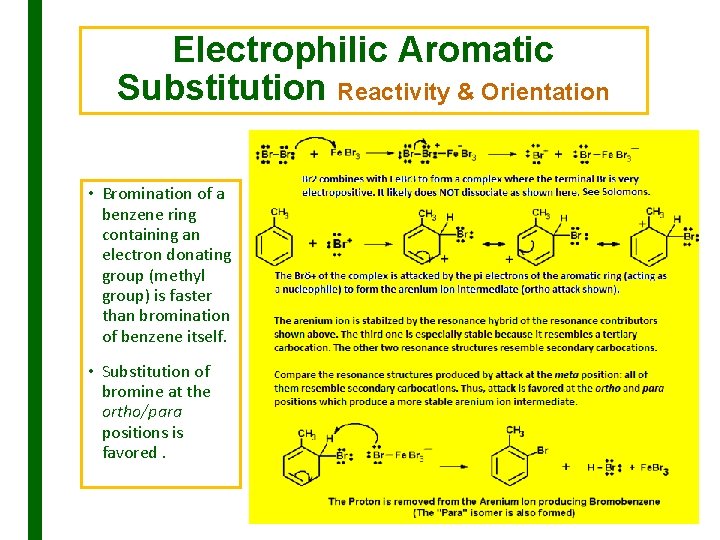
Electrophilic Aromatic Substitution Reactivity & Orientation • Bromination of a benzene ring containing an electron donating group (methyl group) is faster than bromination of benzene itself. • Substitution of bromine at the ortho/para positions is favored.
- Slides: 19