BRIDG Update HL 7 Working Group Meeting San

BRIDG Update HL 7 Working Group Meeting San Antonio, Texas 16 January 2012 Monday Q 4, 3: 30 -5: 00

Agenda • • • BRIDG Overview Recent Accomplishments Current Efforts Ballot Status RCRIM Harmonization Schedule Next Steps 2

BRIDG Overview • BRIDG Purpose: A collaborative effort to produce a shared view of the dynamic and static semantics that collectively define a shared domain-of-interest. • Domain-of-interest/scope: Protocol-driven research and its associated regulatory artifacts, i. e. the data, organization, resources, rules, and processes involved in the formal assessment of the utility, impact, or other pharmacological, physiological, or psychological effects of a drug, procedure, process, subject characteristic, biologic, cosmetic, food or device on a human, animal, or other subject or substance plus all associated regulatory artifacts required for or derived from this effort, including data specifically associated with post-marketing adverse event reporting. • Stakeholders: • Process: Board of Directors prioritizes projects and committee consults with projects and harmonizes project models into main model with help of project analysts/SMEs 3
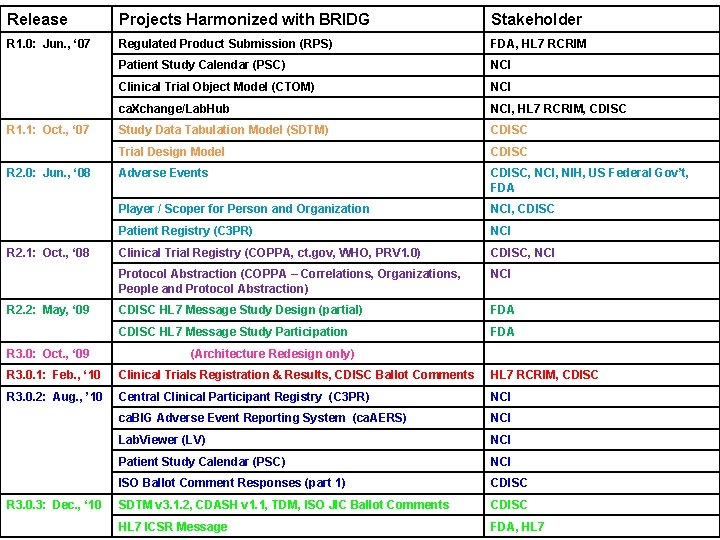
Release Projects Harmonized with BRIDG Stakeholder R 1. 0: Jun. , ‘ 07 Regulated Product Submission (RPS) FDA, HL 7 RCRIM Patient Study Calendar (PSC) NCI Clinical Trial Object Model (CTOM) NCI ca. Xchange/Lab. Hub NCI, HL 7 RCRIM, CDISC Study Data Tabulation Model (SDTM) CDISC Trial Design Model CDISC Adverse Events CDISC, NCI, NIH, US Federal Gov’t, FDA Player / Scoper for Person and Organization NCI, CDISC Patient Registry (C 3 PR) NCI Clinical Trial Registry (COPPA, ct. gov, WHO, PRV 1. 0) CDISC, NCI Protocol Abstraction (COPPA – Correlations, Organizations, People and Protocol Abstraction) NCI CDISC HL 7 Message Study Design (partial) FDA CDISC HL 7 Message Study Participation FDA R 1. 1: Oct. , ‘ 07 R 2. 0: Jun. , ‘ 08 R 2. 1: Oct. , ‘ 08 R 2. 2: May, ‘ 09 R 3. 0: Oct. , ‘ 09 (Architecture Redesign only) R 3. 0. 1: Feb. , ‘ 10 Clinical Trials Registration & Results, CDISC Ballot Comments HL 7 RCRIM, CDISC R 3. 0. 2: Aug. , ’ 10 Central Clinical Participant Registry (C 3 PR) NCI ca. BIG Adverse Event Reporting System (ca. AERS) NCI Lab. Viewer (LV) NCI Patient Study Calendar (PSC) NCI ISO Ballot Comment Responses (part 1) CDISC SDTM v 3. 1. 2, CDASH v 1. 1, TDM, ISO JIC Ballot Comments CDISC HL 7 ICSR Message FDA, HL 7 R 3. 0. 3: Dec. , ‘ 10 4

Recent Accomplishments • Harmonization of: – MD Anderson/National Marrow Donor Program (NMDP) Hematopoietic Cell Transplantation (HCT) project data elements (5 sets of 7) – NCI Case Report Form (CRF) Standard Elements (Rounds 1 & 2) – National Center for Research Resources’ Clinical and Translational Science Award (CTSA) Human Studies Database (HSDB) project • Began harmonization of CDISC Statistics DAM • Updated BRIDG Harmonization Package 5

Current Efforts • Harmonization of: – MD Anderson/National Marrow Donor Program (NMDP) Hematopoietic Cell Transplantation (HCT) project (Set 6 of 7) • Adding bone marrow/stem cell transplant concepts to BRIDG – Genzyme’s Registry. NXT! • Adding rare disease registry concepts to BRIDG • CDISC Statistics Domain Analysis Model • HL 7 Clinical Trials Registration & Results (CTR&R) R 2 • Re-harmonization of HL 7 Study Design Message • Internal efforts – Migrating from GForge to Jira for project tracking – Updating BRIDG website – Creating a script to automatically add mapping tags from mapping spreadsheet to EAP file – Managing enhancement requests provided by community via GForge 6

Next Release of BRIDG • BRIDG R 3. 1 • Slated for February 2012 • Will include: • MD Anderson/National Marrow Donor Program (NMDP) Hematopoietic Cell Transplantation (HCT) project (first 6 sets) • NCI Case Report Form (CRF) Standard Elements (Rounds 1 & 2) • National Center for Research Resources’ Clinical and Translational Science Award (CTSA) Human Studies Database (HSDB) project • CDISC Statistics model • Clinical Trials Registration and Results (CTR&R) R 2 • Re-harmonization of HL 7 Study Design Message 7

RCRIM Ballot Reconciliation • Reconciliations were provided to and accepted by comment submitters. • Ballot Reconciliation Package was approved on the December 20 th 2011 RCRIM telecon. 8

ISO Ballot Status • The ISO Central Secretariat sent comments to the BRIDG team regarding the BRIDG JIC ballot. The BRIDG team responded with proposals to address each comment. CDISC is now working with the new ISO TC 215 Technical Program Manager to determine the next steps for the ballot. • The BRIDG re-ballot deadline is Jan 2012. CDISC is currently working with the ISO TC 215 program manager and has submitted a request for an extension. We should hear back on the results of the request later this month. • Next round of ISO Balloting – need 6 months lead time – 1 month for ISO HQ to review/prepare ballot and 5 months for the Draft International Standard (DIS) ballot. • No set balloting schedule in ISO, but we try to align with a TC 215 Spring or Fall meeting so the ballot can be discussed in person. • The HL 7 ballot needs to end at the same time as the ISO ballot. • Proposed ballot dates: – ISO ballot opens March 2012; closes by end of September 2012 – HL 7 ballot opens August 2012 for September ballot 9
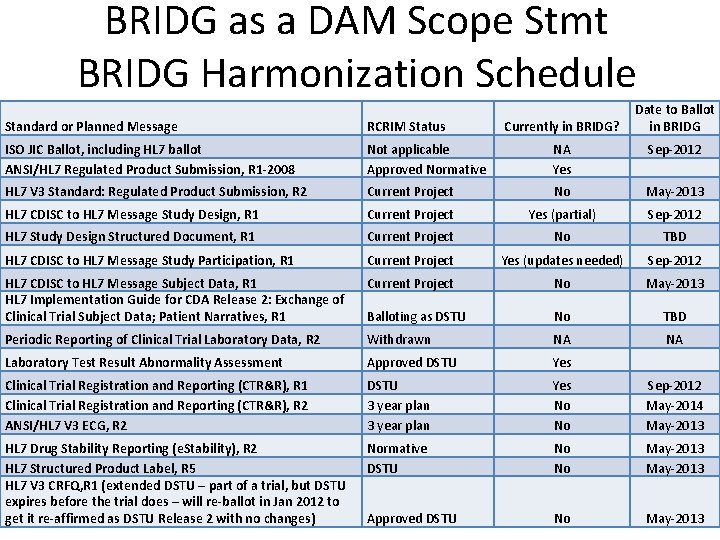
BRIDG as a DAM Scope Stmt BRIDG Harmonization Schedule Currently in BRIDG? Date to Ballot in BRIDG Standard or Planned Message RCRIM Status ISO JIC Ballot, including HL 7 ballot ANSI/HL 7 Regulated Product Submission, R 1 -2008 Not applicable Approved Normative NA Yes Sep-2012 HL 7 V 3 Standard: Regulated Product Submission, R 2 Current Project No May-2013 HL 7 CDISC to HL 7 Message Study Design, R 1 Current Project Yes (partial) Sep-2012 HL 7 Study Design Structured Document, R 1 Current Project No TBD HL 7 CDISC to HL 7 Message Study Participation, R 1 Current Project Yes (updates needed) Sep-2012 HL 7 CDISC to HL 7 Message Subject Data, R 1 HL 7 Implementation Guide for CDA Release 2: Exchange of Clinical Trial Subject Data; Patient Narratives, R 1 Current Project No May-2013 Balloting as DSTU No TBD Periodic Reporting of Clinical Trial Laboratory Data, R 2 Withdrawn NA NA Laboratory Test Result Abnormality Assessment Approved DSTU Yes Clinical Trial Registration and Reporting (CTR&R), R 1 Clinical Trial Registration and Reporting (CTR&R), R 2 ANSI/HL 7 V 3 ECG, R 2 DSTU 3 year plan Yes No No Sep-2012 May-2014 May-2013 HL 7 Drug Stability Reporting (e. Stability), R 2 HL 7 Structured Product Label, R 5 HL 7 V 3 CRFQ, R 1 (extended DSTU – part of a trial, but DSTU expires before the trial does – will re-ballot in Jan 2012 to get it re-affirmed as DSTU Release 2 with no changes) Normative DSTU No No May-2013 Approved DSTU No 10 May-2013

Next Steps • Continue harmonization of: – – Hematopoietic Cell Transplantation (HCT) concepts CDISC Statistics Domain Analysis Model Genzyme’s Registry. NXT! NCI CRF Standard (remaining 2 of 4 rounds) • Manage and resolve enhancement requests from the community • Updates to harmonization of FDA’s HL 7 Study Participation message • HL 7 Ballot September 2012 in concert with ISO Ballot • Update BRIDG as a DAM Harmonization Process Document • Coordinate with RCRIM to withdraw Study Design and Study Participation DAMs from ballot 11

Items on the Horizon • Harmonization of: • CDISC Operational Data Model – Study Design Model (ODMSDM) • Observational Medical Outcomes Partnership (OMOP) • NCI Clinical Trials Reporting Program (CTRP) • CDISC Standard for Exchange of Non-Clinical Data (SEND) • CDISC SDTM for Devices • Exploring the generation of an OWL view of BRIDG • Exploring the identification of vocabularies for BRIDG codeable attributes • Exploring the inclusion of dynamic semantics in BRIDG 12
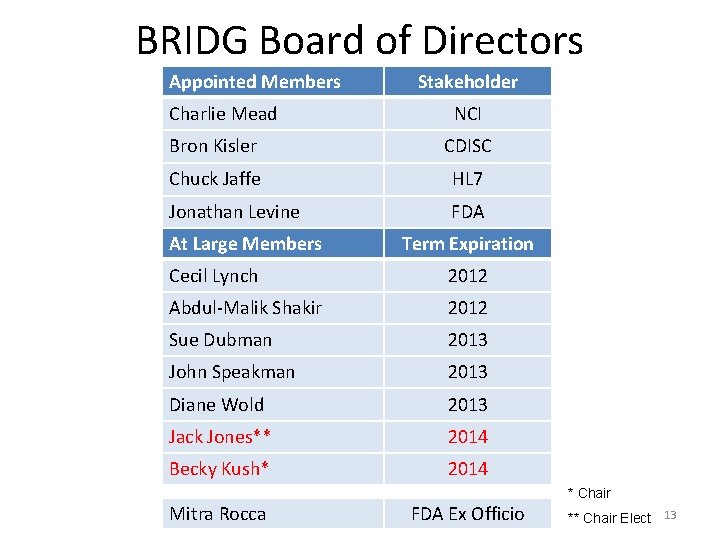
BRIDG Board of Directors Appointed Members Charlie Mead Stakeholder NCI Bron Kisler CDISC Chuck Jaffe HL 7 Jonathan Levine FDA At Large Members Term Expiration Cecil Lynch 2012 Abdul-Malik Shakir 2012 Sue Dubman 2013 John Speakman 2013 Diane Wold 2013 Jack Jones** 2014 Becky Kush* 2014 * Chair Mitra Rocca FDA Ex Officio ** Chair Elect 13

Contact Information • BRIDG Project Website – www. bridgmodel. org • SCC Contact Listserv – bridg. THC-L@list. nih. gov • BRIDG Users Listserv – BRIDG_USERS@list. nih. gov • RCRIM: BRIDG as a DAM Listserv – bridg@lists. hl 7. org 14

Q&A 15

Back up slides 16

Semantic Coordination Committee Name Affiliation Charlie Mead NCI Becky Angeles NCI / Scen. Pro, Inc. Smita Hastak NCI / Scen. Pro, Inc. Steve Sandberg NCI / Scen. Pro, Inc. Wendy Ver Hoef NCI / Scen. Pro, Inc. Julie Evans CDISC Lloyd Mc. Kenzie NCI / Gordon Point Informatics 17 17
- Slides: 17