BRIDG Activities Clinical Data Elements and the role

BRIDG Activities, Clinical Data Elements and the role of MDR Basel, 21. March 2008 Mark Regenass, Isabelle de Zegher, Peter Spiers, Saurin Mehta, Mitra Rocca

Key question § How to develop and maintain an internal data element registry • Supporting data integration • Supporting SDTM submissions

Three strategic decisions § Specify the set of data elements for Novartis Development analogous to SDTM data elements (extend SDTM for Novartis). § Use a ISO 11179 MDR 1) -based system to register the data elements with. § Introduce an Analysis Model for Novartis Development analogous to the BRIDG 2) model mapping the data elements to unambiguously defined concepts. 1) ISO/IEC 11179, Information Technology -- Metadata registries (MDR) 2) Biomedical Research Integrated Domain Group 3 | BRIDG Activity, CDE & MDR | Mark Regenass | 21. 03. 2008
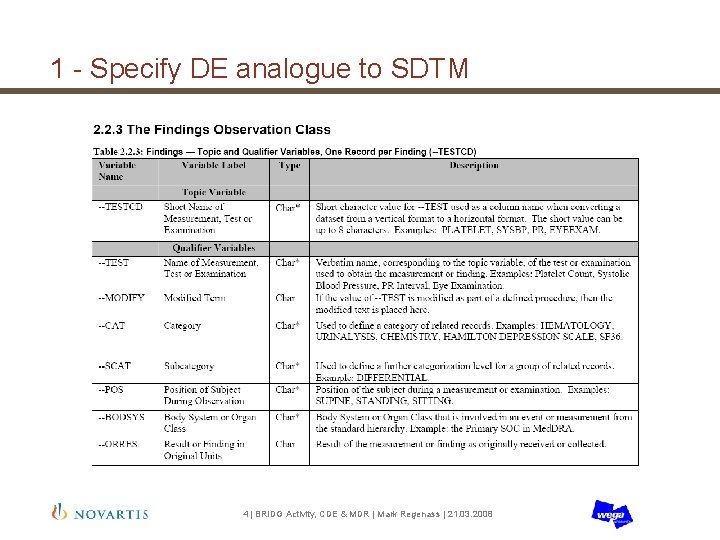
1 - Specify DE analogue to SDTM 4 | BRIDG Activity, CDE & MDR | Mark Regenass | 21. 03. 2008

Extend SDTM for Novartis => SDTM++ Additional attributes per DE Additional DE’s per Domain 5 | BRIDG Activity, CDE & MDR | Mark Regenass | 21. 03. 2008
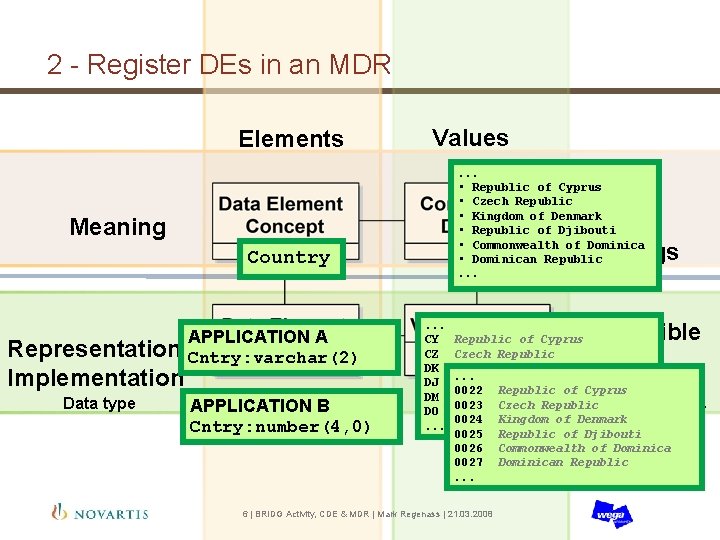
2 - Register DEs in an MDR Elements Values. . . • Republic of Cyprus • Czech Republic • Kingdom of Denmark • Republic of Djibouti • Commonwealth of Dominica • Dominican Republic. . . Set of Value Meanings Meaning Country Representation Implementation Data type APPLICATION A Cntry: varchar(2) APPLICATION B Cntry: number(4, 0) . . . CY CZ DK DJ DM DO. . . Permissible Values Republic of Cyprus Czech Republic Kingdom of Denmark. . . Republic of Djibouti 0022 Republic of Cyprus Commonwealth of Dominica Codes/Ranges/… 0023 Czech Republic Dominican Republic 0024 Kingdom of Denmark 0025 Republic of Djibouti 0026 Commonwealth of Dominica 0027 Dominican Republic. . . 6 | BRIDG Activity, CDE & MDR | Mark Regenass | 21. 03. 2008
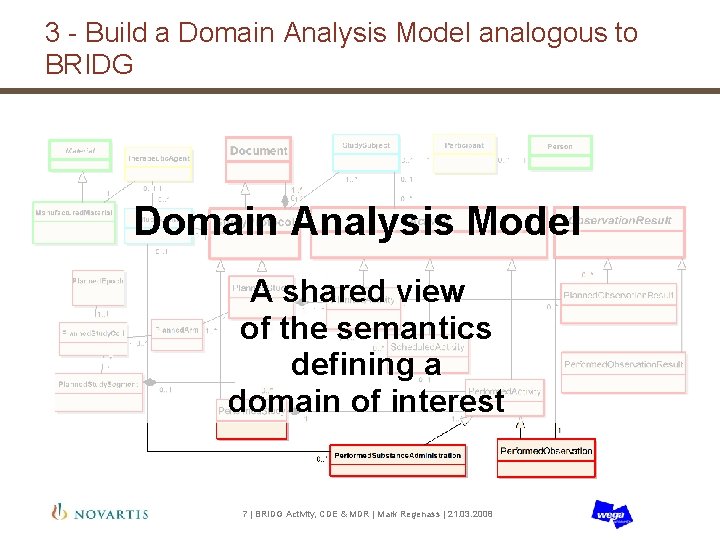
3 - Build a Domain Analysis Model analogous to BRIDG Domain Analysis Model A shared view of the semantics defining a domain of interest 7 | BRIDG Activity, CDE & MDR | Mark Regenass | 21. 03. 2008
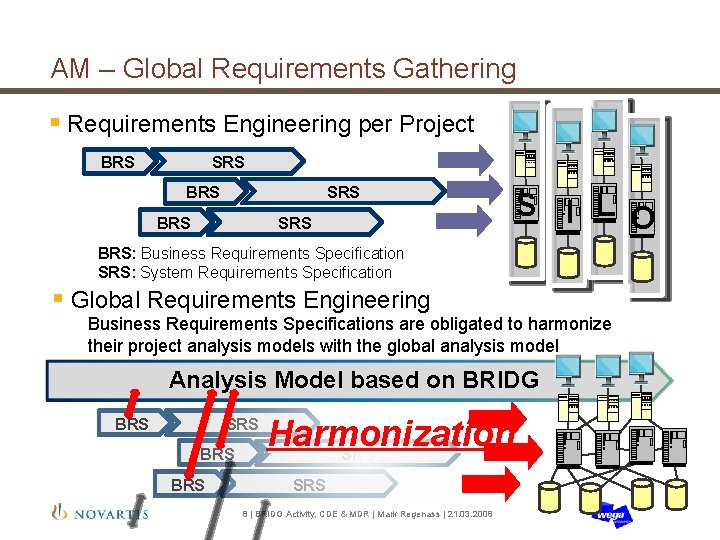
AM – Global Requirements Gathering § Requirements Engineering per Project BRS SRS BRS S I L O BRS: Business Requirements Specification SRS: System Requirements Specification § Global Requirements Engineering Business Requirements Specifications are obligated to harmonize their project analysis models with the global analysis model Analysis Model based on BRIDG BRS SRS BRS Harmonization SRS 8 | BRIDG Activity, CDE & MDR | Mark Regenass | 21. 03. 2008
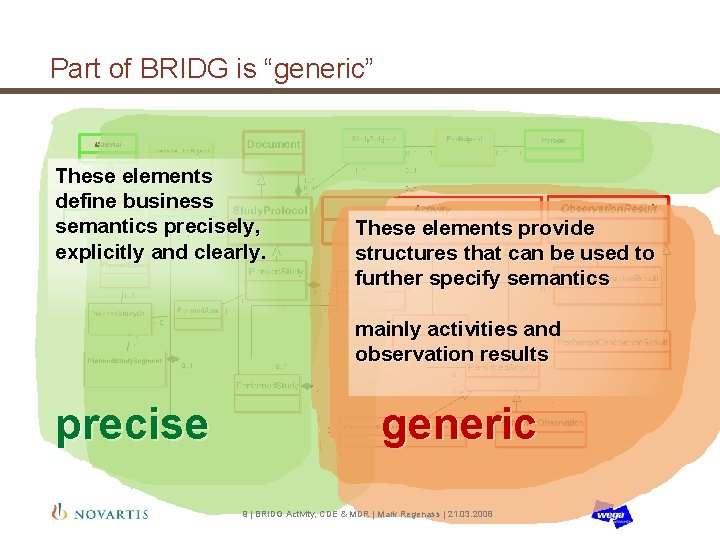
Part of BRIDG is “generic” These elements define business semantics precisely, explicitly and clearly. These elements provide structures that can be used to further specify semantics mainly activities and observation results precise generic 9 | BRIDG Activity, CDE & MDR | Mark Regenass | 21. 03. 2008
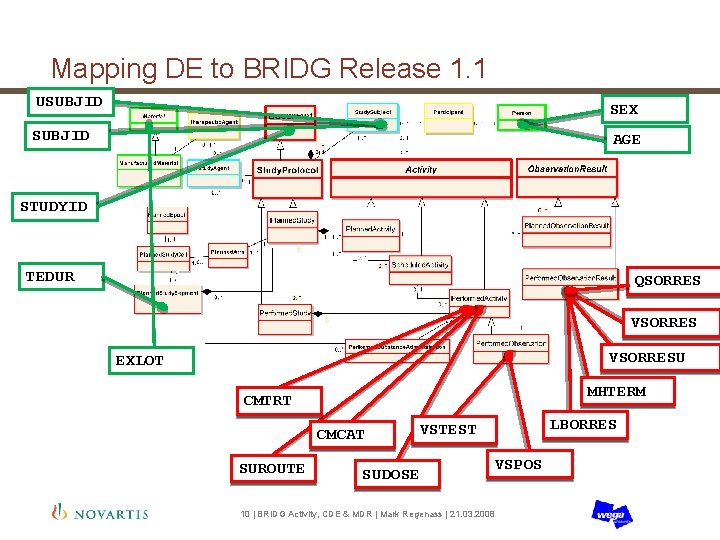
Mapping DE to BRIDG Release 1. 1 USUBJID SEX SUBJID AGE STUDYID TEDUR QSORRES VSORRESU EXLOT MHTERM CMTRT CMCAT SUROUTE LBORRES VSTEST SUDOSE VSPOS 10 | BRIDG Activity, CDE & MDR | Mark Regenass | 21. 03. 2008
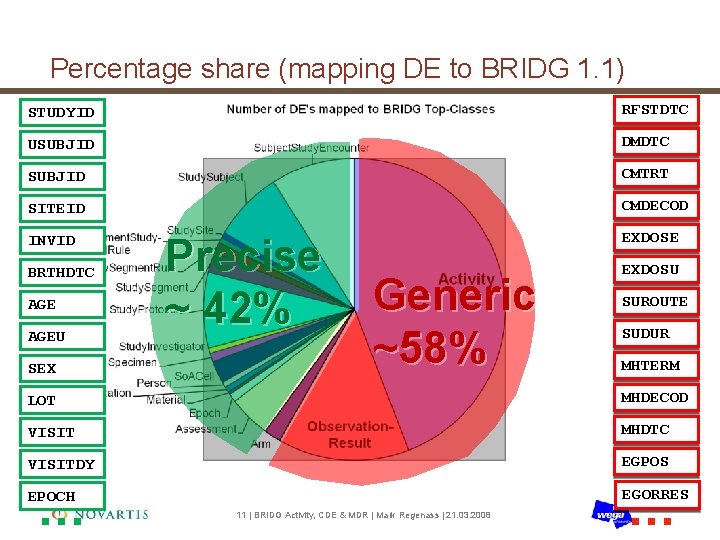
Percentage share (mapping DE to BRIDG 1. 1) STUDYID RFSTDTC USUBJID DMDTC SUBJID CMTRT SITEID CMDECOD INVID EXDOSE BRTHDTC AGEU SEX Precise ~ 42% Generic ~58% EXDOSU SUROUTE SUDUR MHTERM LOT MHDECOD VISIT MHDTC VISITDY EGPOS … … EGORRES EPOCH 11 | BRIDG Activity, CDE & MDR | Mark Regenass | 21. 03. 2008
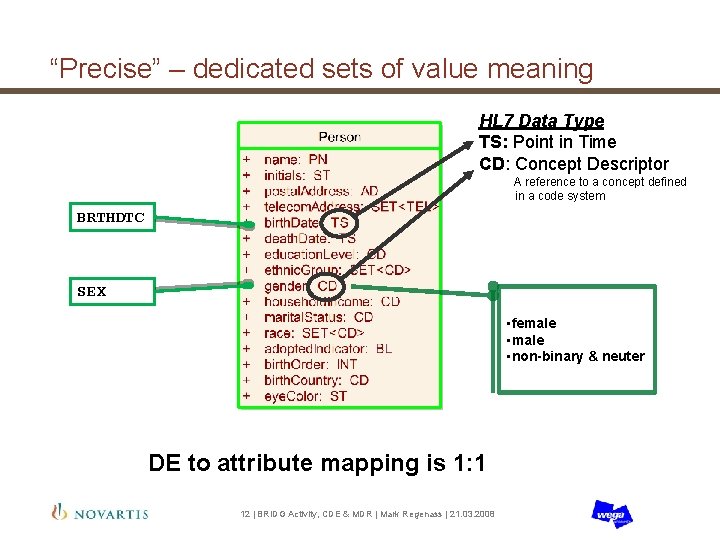
“Precise” – dedicated sets of value meaning HL 7 Data Type TS: Point in Time CD: Concept Descriptor A reference to a concept defined in a code system BRTHDTC SEX • female • non-binary & neuter DE to attribute mapping is 1: 1 12 | BRIDG Activity, CDE & MDR | Mark Regenass | 21. 03. 2008
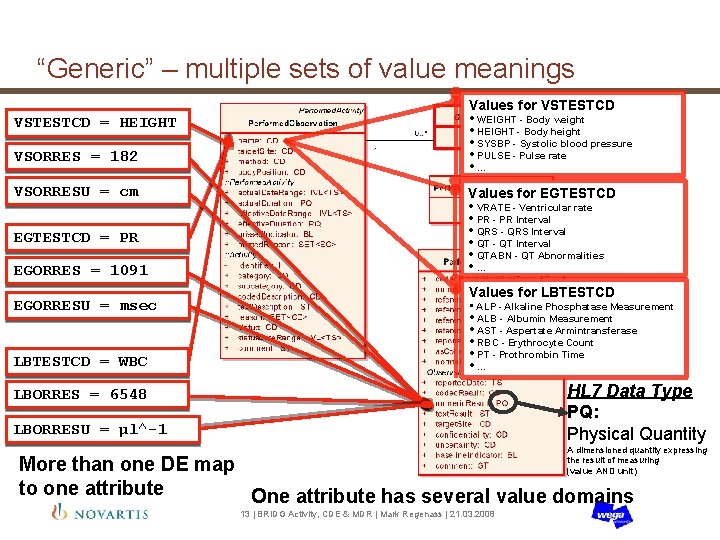
“Generic” – multiple sets of value meanings Values for VSTESTCD = HEIGHT VSORRES = 182 VSORRESU = cm • WEIGHT - Body weight • HEIGHT - Body height • SYSBP - Systolic blood pressure • PULSE - Pulse rate • … Values for EGTESTCD EGORRES = 1091 • VRATE - Ventricular rate • PR - PR Interval • QRS - QRS Interval • QT - QT Interval • QTABN - QT Abnormalities • … EGORRESU = msec Values for LBTESTCD • ALP - Alkaline Phosphatase Measurement EGTESTCD = PR LBTESTCD = WBC • ALB - Albumin Measurement • AST - Aspertate Armintransferase • RBC - Erythrocyte Count • PT - Prothrombin Time • … HL 7 Data Type PQ: Physical Quantity LBORRES = 6548 LBORRESU = µl^-1 A dimensioned quantity expressing the result of measuring (value AND unit) More than one DE map to one attribute One attribute has several value domains 13 | BRIDG Activity, CDE & MDR | Mark Regenass | 21. 03. 2008
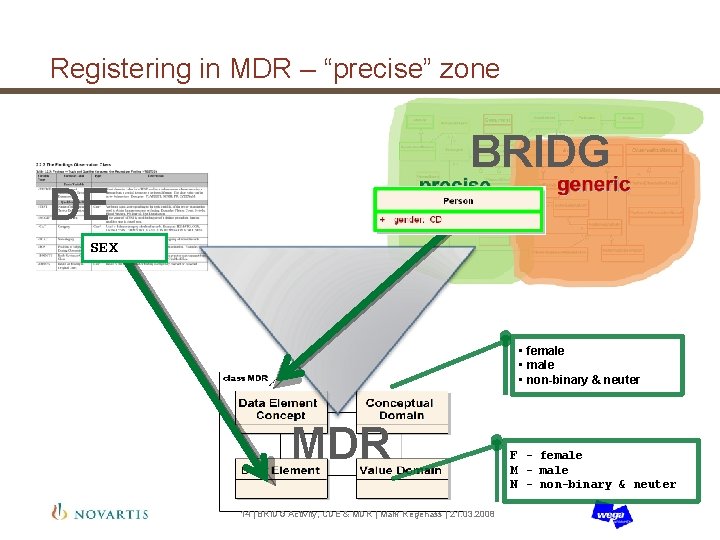
Registering in MDR – “precise” zone BRIDG DE SEX • female • non-binary & neuter MDR 14 | BRIDG Activity, CDE & MDR | Mark Regenass | 21. 03. 2008 F - female M - male N - non-binary & neuter
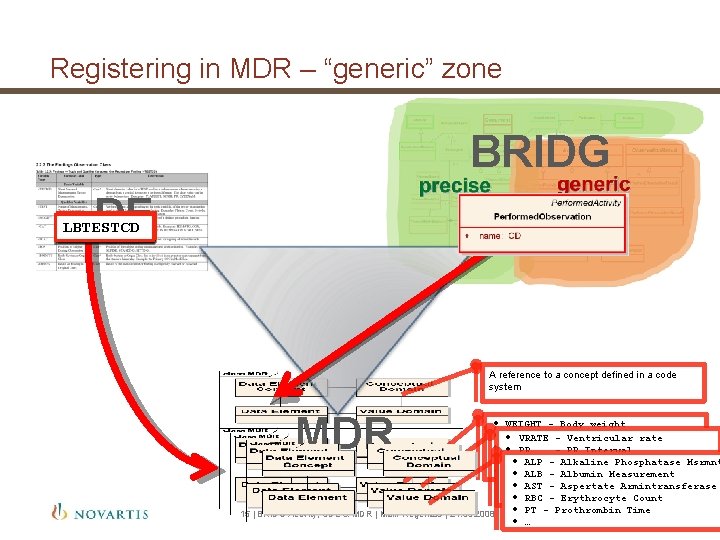
Registering in MDR – “generic” zone BRIDG DE EGTESTCD VSTESTCD LBTESTCD A reference to a concept defined in a code system MDR • WEIGHT - Body weight • • HEIGHT height rate VRATE--Body Ventricular • • SYSBP Systolic blood pressure PR - PR Interval • • PULSE Pulse rate • QRS ALP -- Alkaline Phosphatase Msrmnt QRS Interval • • … • QTALB -- Albumin Measurement QT Interval AST -- Aspertate Armintransferase • • QTABN QT Abnormalities • • … RBC - Erythrocyte Count • PT - Prothrombin Time 15 | BRIDG Activity, CDE & MDR | Mark Regenass | 21. 03. 2008 • …

Conclusion § SDTM needs to be extended to fully cover the need of data element definition from data collection onwards. § Use of BRIDG as a source for unambiguous data element concepts requires 2 different types of mapping: • “precise”: one-to-one mapping between a DE and a BRIDG concept; business semantics precisely, explicitly and clearly defined. • “generic”: many DE fit within a single BRIDG concept, this BRIDG concept is providing structure to further specify semantics (mainly to define specific activities and observation results). § An ISO 11179 based MDR can cover all requirements for data element registration only when applied with some flexibility.
- Slides: 16