Breathing Easier Through Spectroscopy Studying Free Radical Reactions

Breathing Easier Through Spectroscopy: Studying Free Radical Reactions In Air Pollution Chemistry by Frequency Comb Spectroscopy Mitchio Okumura California Institute of Technology
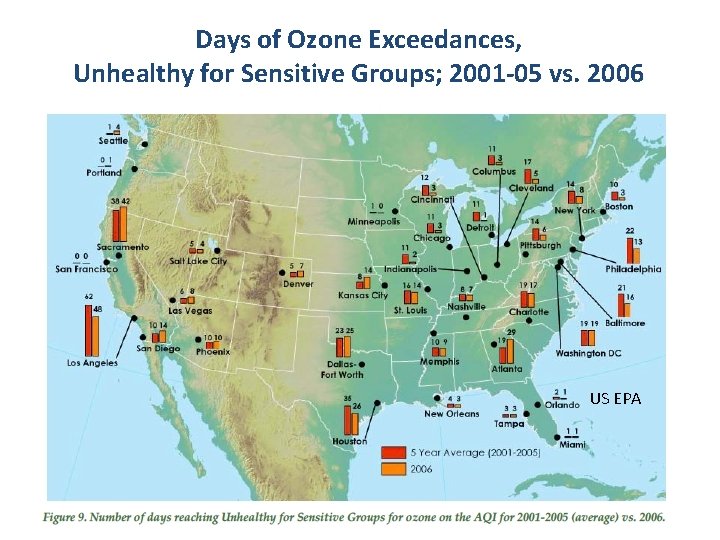
Days of Ozone Exceedances, Unhealthy for Sensitive Groups; 2001 -05 vs. 2006 US EPA
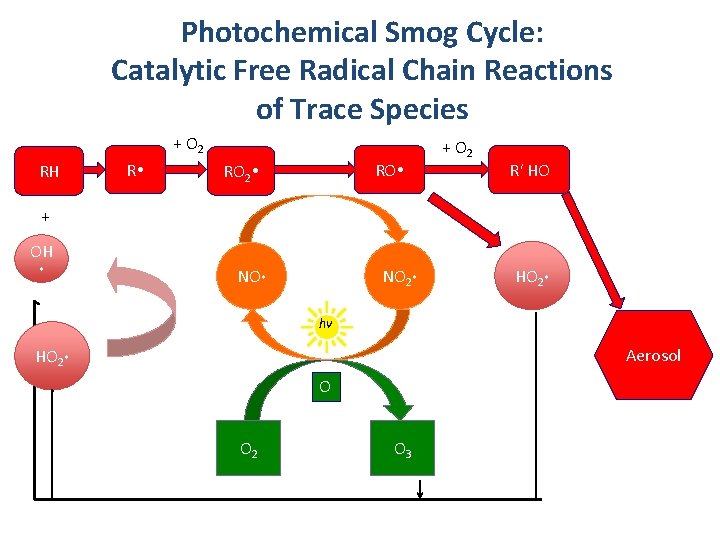
Photochemical Smog Cycle: Catalytic Free Radical Chain Reactions of Trace Species + O 2 RH R RO 2 + O 2 R' HO + OH NO 2 HO 2 hv Aerosol HO 2 O 3

Time-resolved Spectroscopy Of Gas Phase Reactions Norrish and Porter's Flash Photolysis Experiment time NO (v) vs time

Time-Resolved Free Radical Kinetics Caltech Group 1. Pulsed and cw Cavity Ringdown Spectroscopy 2. VUV Photoionization Mass Spectrometry (Sandia, ALS) 3. Frequency Comb Laser Spectroscopy (Ye group, JILA)

PROOF OF PRINCIPLES EXPERIMENT: Detection of the DOCO Radical by Time Resolved Frequency Comb Spectroscopy (TRFCS) J. Phys. Chem. Letters, 5, 2241 -1146 (2014). Adam Fleisher, Bryce Byork, Kevin Cossel, Bryan Changala, Benjamin Spaun, Oliver Heckl , and Jun Ye JILA, NIST and University of Colorado Thinh Bui, Mitchio Okumura California Institute of Technology
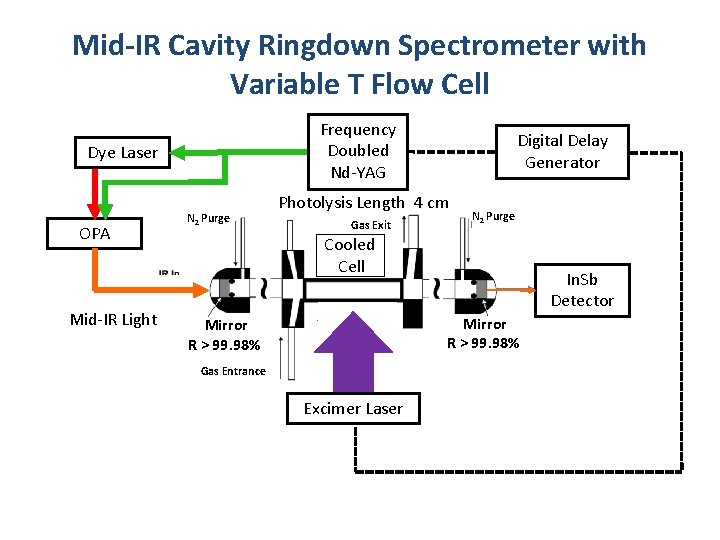
Mid-IR Cavity Ringdown Spectrometer with Variable T Flow Cell Frequency Doubled Nd-YAG Dye Laser OPA Mid-IR Light N 2 Purge Digital Delay Generator Photolysis Length 4 cm Gas Exit N 2 Purge Cooled Cell In. Sb Detector Mirror R > 99. 98% Gas Entrance Excimer Laser
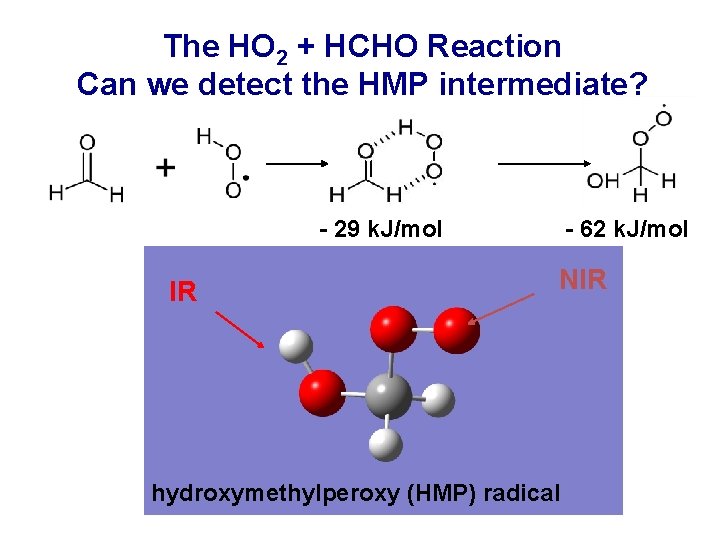
The HO 2 + HCHO Reaction Can we detect the HMP intermediate? - 29 k. J/mol IR - 62 k. J/mol NIR hydroxymethylperoxy (HMP) radical
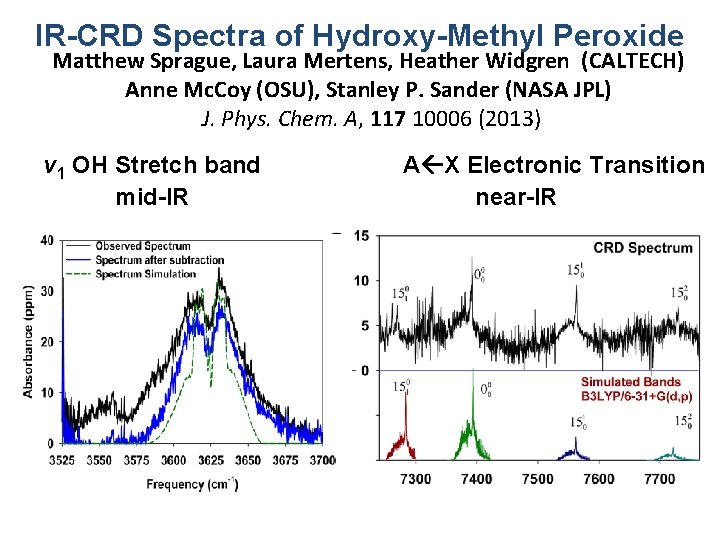
IR-CRD Spectra of Hydroxy-Methyl Peroxide Matthew Sprague, Laura Mertens, Heather Widgren (CALTECH) Anne Mc. Coy (OSU), Stanley P. Sander (NASA JPL) J. Phys. Chem. A, 117 10006 (2013) v 1 OH Stretch band mid-IR A X Electronic Transition near-IR

Pros and Cons of "Low Resolution" Pulsed CRDS for Free Radical Kinetics • broadly tunable • good sensitivity IR-UV nmin ~ 1011 -1013 /cc) – can require high concentrations of precursors, radicals; fast rates • time resolution 1 -100 us – 'SKAR' deconvolution required for fast processes (t ≲ τringdown) • Linewidth issues Γlaser > 40 x Γline – Intensity and lineshape are problems • Low rep rate 10's Hz to 1 k. Hz – one time-point per excimer shot in time-dependent kinetics

Mid-Infrared Frequency Comb System Collaboration with Jun Ye Experiments at JILA, University of Colorado, Boulder F. Adler et al. Opt. Lett. 34, 1330 (2009).

Motivation: The OH + CO ➝ H + CO 2 Reaction IN THE ATMOSPHERE: • Rate Constant k (298, 1 atm) ≈ 2 × 10− 13 cm 3 s− 1 • CO concentrations Free Troposphere Polluted urban air [CO] ≈ 50 -200 ppb [CO] ≈ 100 -2000 ppb • OH + CO is the major tropospheric sink for OH globally
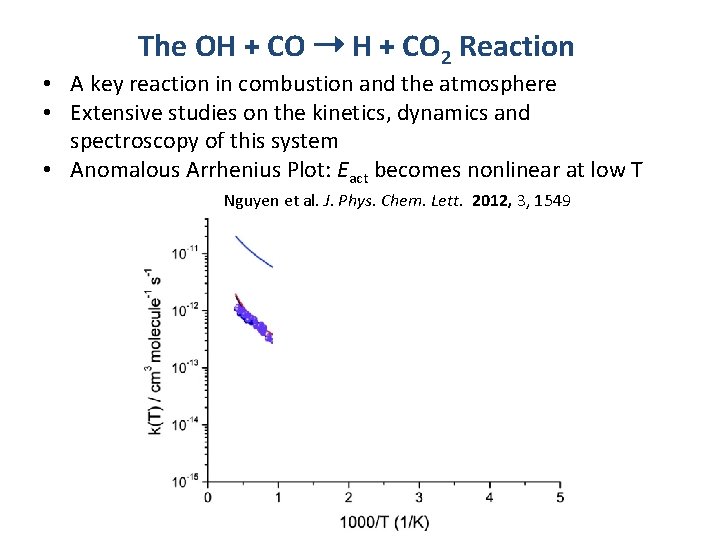
The OH + CO ➝ H + CO 2 Reaction • A key reaction in combustion and the atmosphere • Extensive studies on the kinetics, dynamics and spectroscopy of this system • Anomalous Arrhenius Plot: Eact becomes nonlinear at low T Nguyen et al. J. Phys. Chem. Lett. 2012, 3, 1549 High Pressure Low Pressure
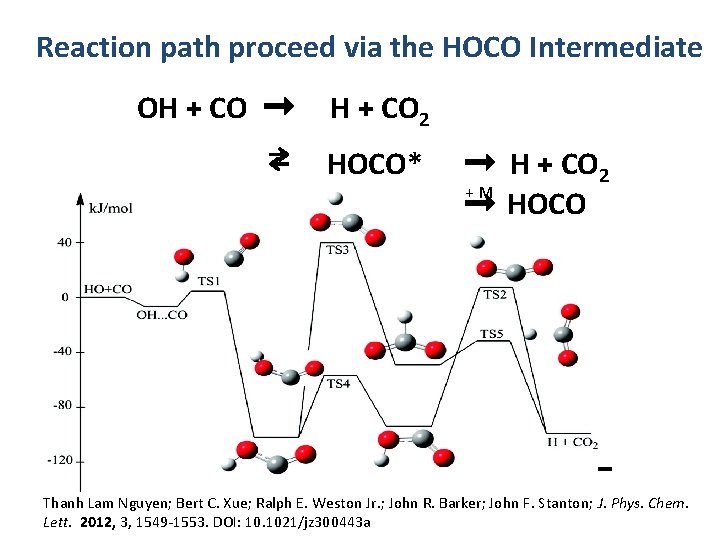
Reaction path proceed via the HOCO Intermediate OH + CO ➞ H + CO 2 ⇄ HOCO* OH + CO ➞ H + CO 2 + M ➞ HOCO c-HOCO t-HOCO -50 kcal/mol H + CO 2 Thanh Lam Nguyen; Bert C. Xue; Ralph E. Weston Jr. ; John R. Barker; John F. Stanton; J. Phys. Chem. Lett. 2012, 3, 1549 -1553. DOI: 10. 1021/jz 300443 a

Frequency Comb Lasers • Leading to in advances AMO Physics (Hansch, Hall, Physics Nobel Prize 2005) • Metrology and spectroscopy • Combines advances in laser stabilization, ultrafast lasers, and coherent and nonlinear optics CAN FREQUENCY COMB LASERS BE APPLIED TO FREE RADICAL KINETICS AND DYNAMICS?
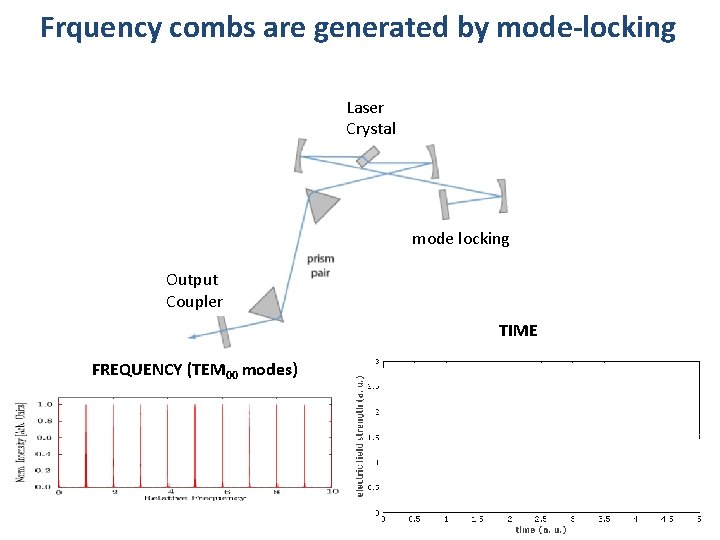
Frquency combs are generated by mode-locking Laser Crystal mode locking mirror Output Coupler TIME FREQUENCY (TEM 00 modes)
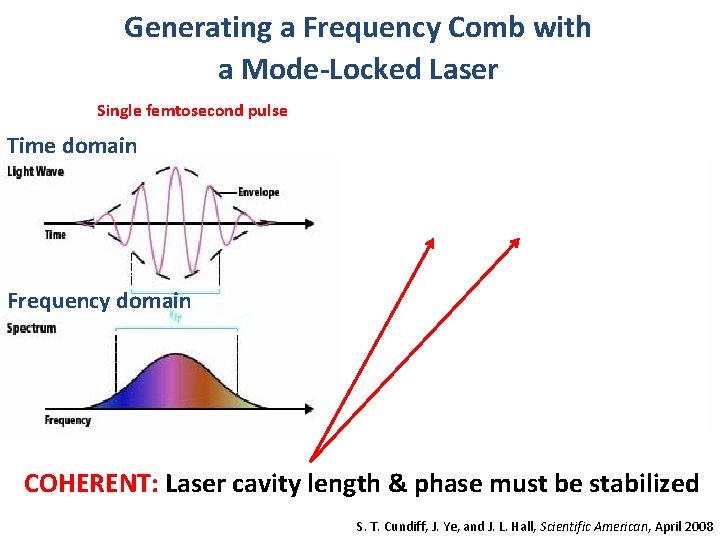
Generating a Frequency Comb with a Mode-Locked Laser Single femtosecond pulse Train of pulses stabilized mode-locked laser Time domain Δϕ 2Δϕ E(t) 1/frep Frequency domain frep FREQUENCY COMB COHERENT: Laser cavity length & phase must be stabilized S. T. Cundiff, J. Ye, and J. L. Hall, Scientific American, April 2008
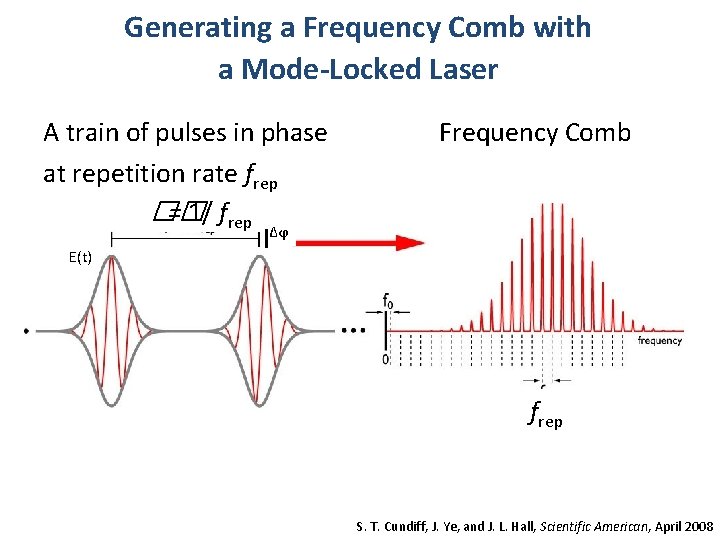
Generating a Frequency Comb with a Mode-Locked Laser A train of pulses in phase at repetition rate frep �� = 1/ frep Frequency Comb Δϕ E(t) frep FREQUENCY COMB S. T. Cundiff, J. Ye, and J. L. Hall, Scientific American, April 2008
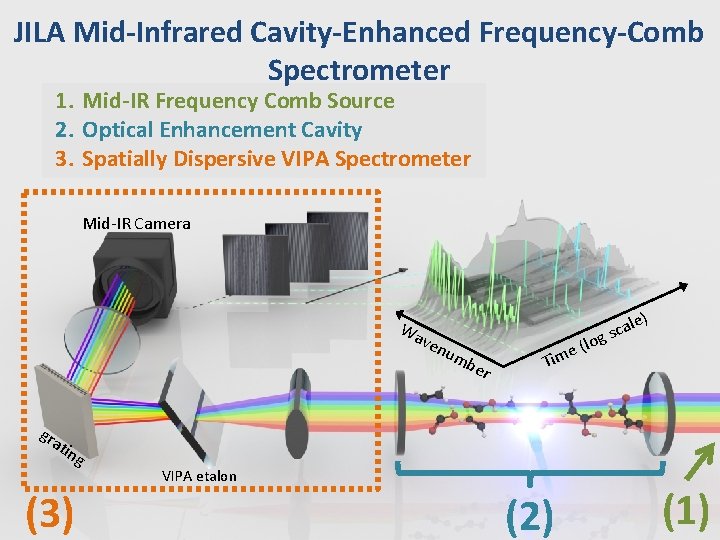
JILA Mid-Infrared Cavity-Enhanced Frequency-Comb Spectrometer 1. Mid-IR Frequency Comb Source 2. Optical Enhancement Cavity 3. Spatially Dispersive VIPA Spectrometer Mid-IR Camera Wa ven ) um ber o (l e m i T le a c s g gra t ing (3) VIPA etalon (2) (1)
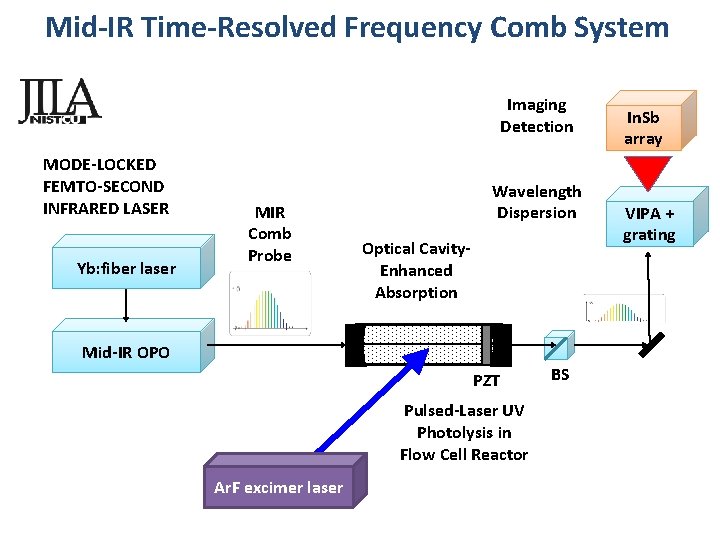
Mid-IR Time-Resolved Frequency Comb System Imaging Detection MODE-LOCKED FEMTO-SECOND INFRARED LASER Yb: fiber laser MIR Comb Probe Wavelength Dispersion Optical Cavity. Enhanced Absorption Mid-IR OPO PZT Pulsed-Laser UV Photolysis in Flow Cell Reactor Ar. F excimer laser BS In. Sb array VIPA + grating

1. Mid-Infrared Frequency Comb Laser Comb-Laser-Pumped Optical Parametric Oscillator Mid-Infrared Radiation tunable between 2. 8 -4. 8 μm 10 W, 1 μm Yb Fiber Comb > 500 m. W idler power 2. 8 – 4. 8 μm Synchronously Pumped PPLN OPO F. Adler et al. Opt. Lett. 34, 1330 (2009).
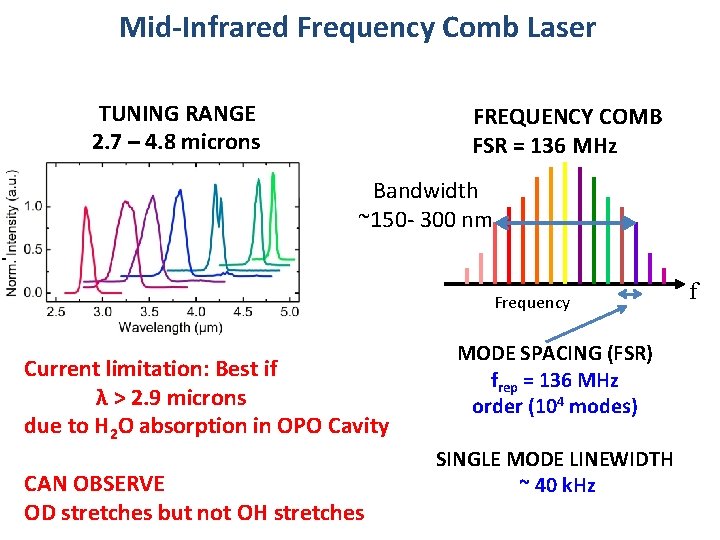
Mid-Infrared Frequency Comb Laser TUNING RANGE 2. 7 – 4. 8 microns ϕo+nϕo ϕo FREQUENCY COMB FSR = 136 MHz Bandwidth ~150 - 300 nm 7. 35 ns Current limitation: Best if λ > 2. 9 microns due to H 2 O absorption in OPO Cavity CAN OBSERVE OD stretches but not OH stretches Frequency MODE SPACING (FSR) frep = 136 MHz order (104 modes) SINGLE MODE LINEWIDTH ~ 40 k. Hz f
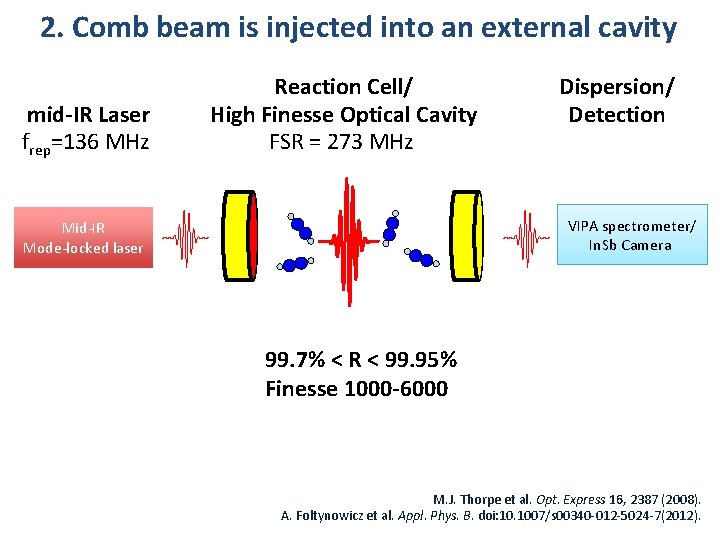
2. Comb beam is injected into an external cavity mid-IR Laser frep=136 MHz Reaction Cell/ High Finesse Optical Cavity FSR = 273 MHz Dispersion/ Detection VIPA spectrometer/ In. Sb Camera Mid-IR Mode-locked laser 99. 7% < R < 99. 95% Finesse 1000 -6000 M. J. Thorpe et al. Opt. Express 16, 2387 (2008). A. Foltynowicz et al. Appl. Phys. B. doi: 10. 1007/s 00340 -012 -5024 -7(2012).
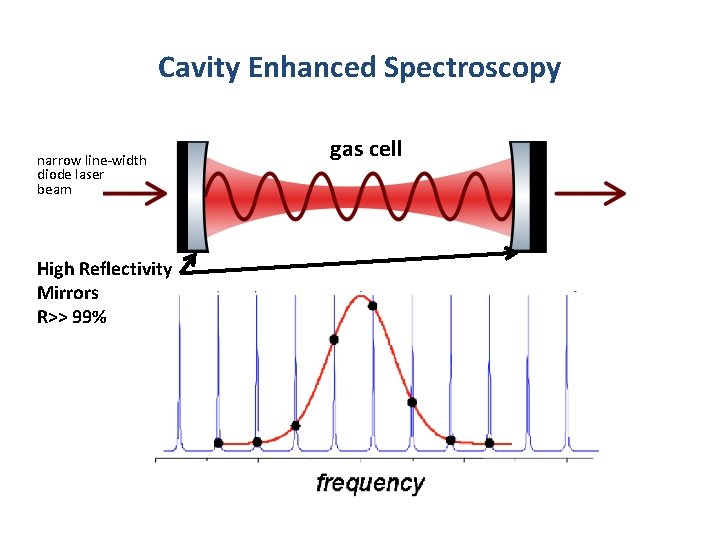
Cavity Enhanced Spectroscopy narrow line-width diode laser beam High Reflectivity Mirrors R>> 99% gas cell
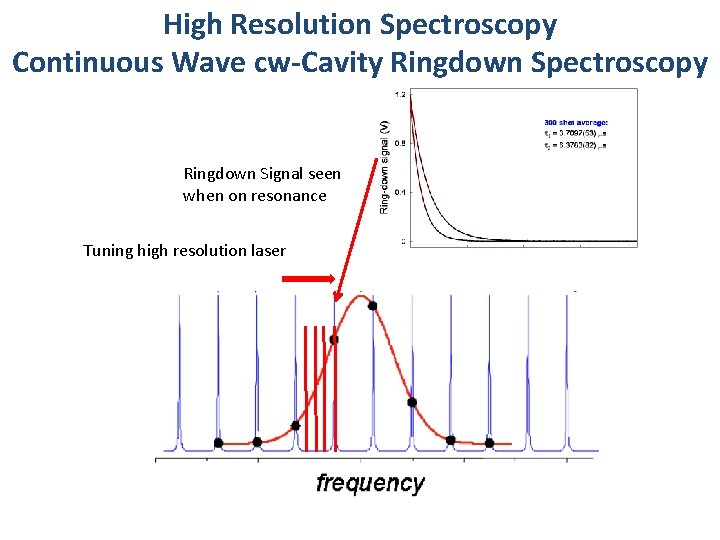
High Resolution Spectroscopy Continuous Wave cw-Cavity Ringdown Spectroscopy Ringdown Signal seen when on resonance Tuning high resolution laser
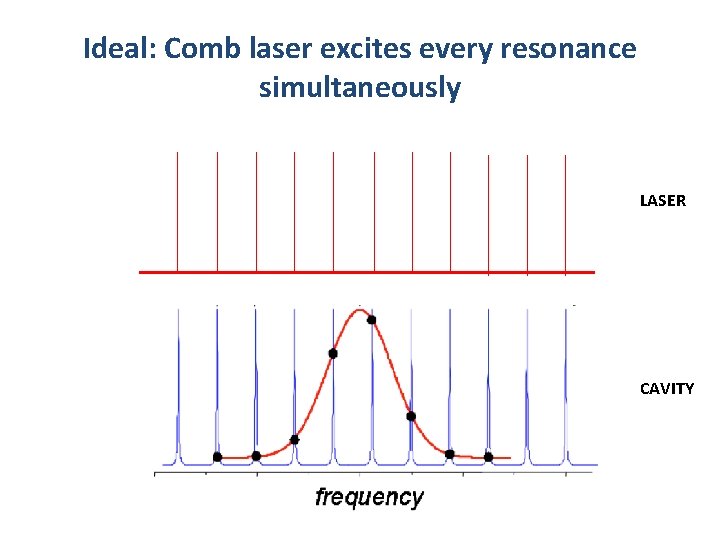
Ideal: Comb laser excites every resonance simultaneously LASER CAVITY
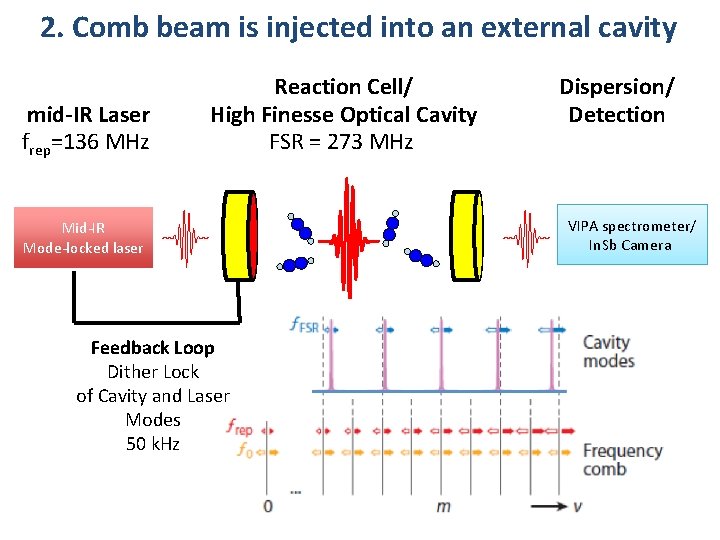
2. Comb beam is injected into an external cavity mid-IR Laser frep=136 MHz Reaction Cell/ High Finesse Optical Cavity FSR = 273 MHz VIPA spectrometer/ In. Sb Camera Mid-IR Mode-locked laser Feedback Loop Dither Lock of Cavity and Laser Modes 50 k. Hz Dispersion/ Detection 99. 7% < R < 99. 95% Finesse 1000 -6000

3. Dispersive Imaging Detector: Virtually Imaged Phase Array (VIPA) L. Nugent-Glandorf et al. , Opt. Lett. 37, 3285 (2012). VERTICAL HIGH RESOLUTION (ETALON – VIRTUALLY IMAGED PHASE ARRAY) HORIZONTAL LOW RESOLUTION IR CAMERA
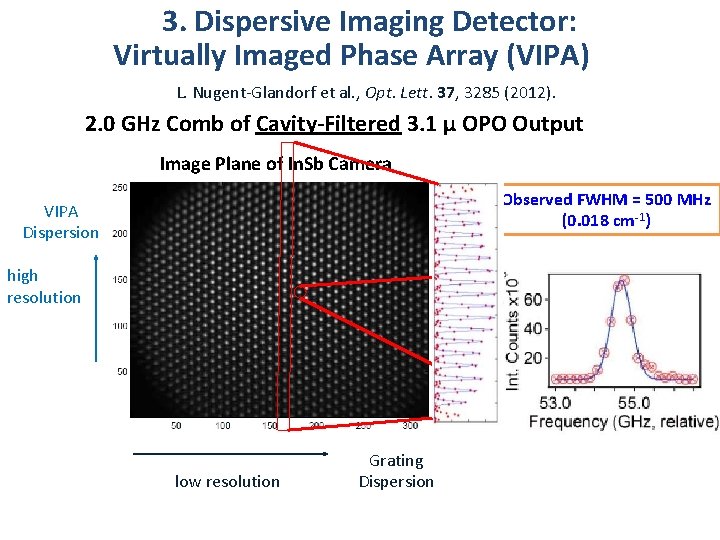
3. Dispersive Imaging Detector: Virtually Imaged Phase Array (VIPA) L. Nugent-Glandorf et al. , Opt. Lett. 37, 3285 (2012). 2. 0 GHz Comb of Cavity-Filtered 3. 1 µ OPO Output Image Plane of In. Sb Camera Observed FWHM = 500 MHz (0. 018 cm-1) VIPA Dispersion high resolution low resolution Grating Dispersion
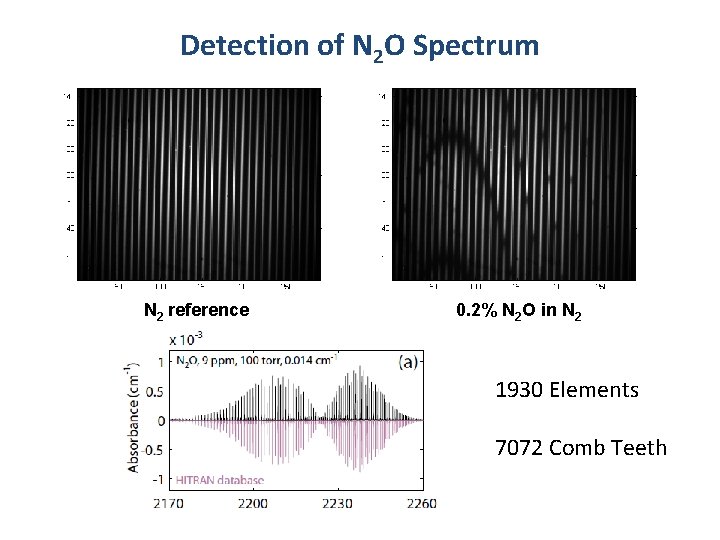
Detection of N 2 O Spectrum N 2 reference 0. 2% N 2 O in N 2 1930 Elements 7072 Comb Teeth

4. Timing: Limited by IR In. Sb Camera 193 nm pulse t=0 τ ~~ In. Sb CAMERA Integration ~~ 4 ms 25 μs integration window pre-photolysis reference images Laser: In. Sb CAMERA signal images 100 fs pulse every 7. 35 ns INTEGRATION TIME READOUT TIME VARIABLE EXCIMER/CAMERA DELAY > 10 μs (25, 50 μs) 4 ms τ K. C. Cossel et al. , Appl. Phys. B 100, 917 (2010)
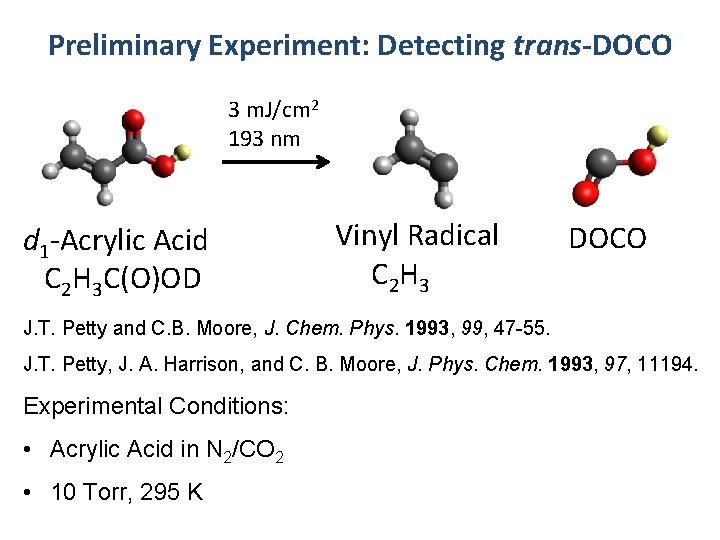
Preliminary Experiment: Detecting trans-DOCO 3 m. J/cm 2 193 nm d 1 -Acrylic Acid C 2 H 3 C(O)OD Vinyl Radical C 2 H 3 DOCO J. T. Petty and C. B. Moore, J. Chem. Phys. 1993, 99, 47 -55. J. T. Petty, J. A. Harrison, and C. B. Moore, J. Phys. Chem. 1993, 97, 11194. Experimental Conditions: • Acrylic Acid in N 2/CO 2 • 10 Torr, 295 K

Chemistry in high-finesse cavity/flow cell N 2 pump N 2 P gauge M 2 N 2 purge Optical Length 54. 88 cm Photolysis Region 3 cm Typical chemistry conditions: 10 Torr N 2 buffer gas 10 m. J 193 nm excimer pulse [AA] ∼ 3 x 1014 cm-3 1 -2% dissociation M 1 N 2 purge Laser range: 2400 -2700 cm-1 signal-averaging: rep rate 0. 2 Hz typ. 960 averages VIPA bandwidth: 65 cm-1 7072 comb teeth 1930 Resolvable elements Camera integration time: 25 µs or 50 µs
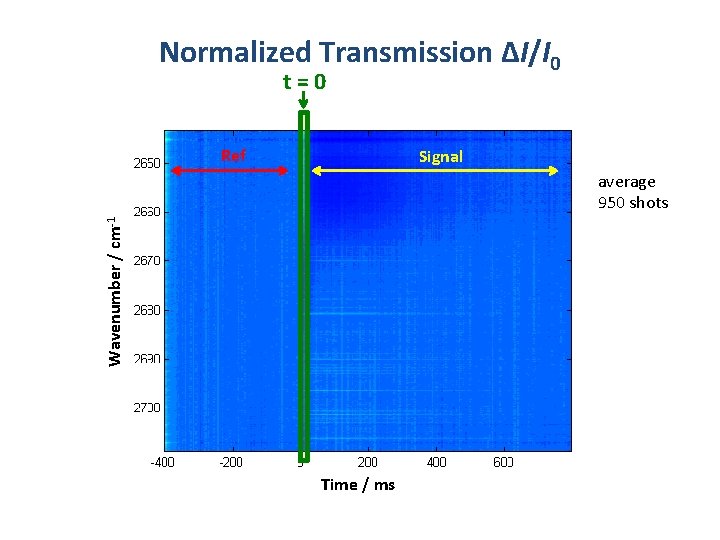
Normalized Transmission ΔI/I 0 t=0 Ref Signal Wavenumber / cm-1 average 950 shots Time / ms
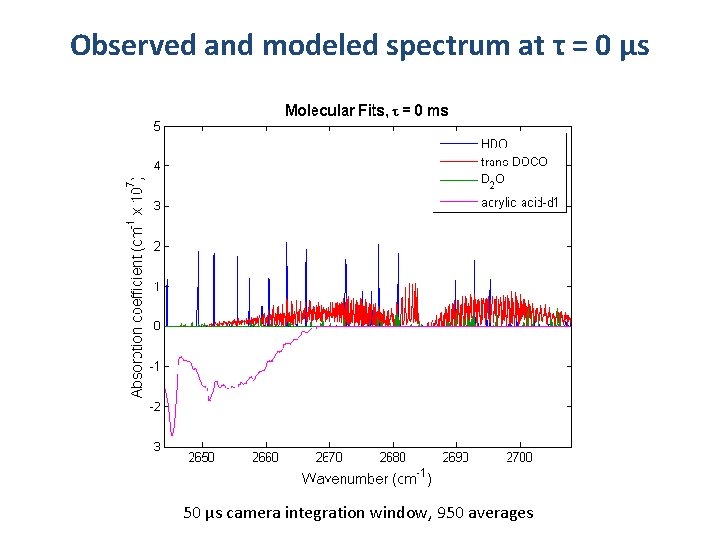
Observed and modeled spectrum at τ = 0 μs 50 μs camera integration window, 950 averages
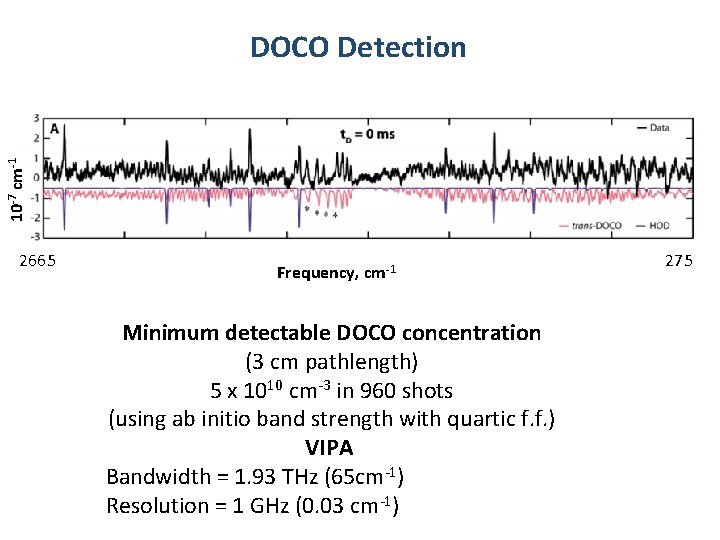
10 -7 cm-1 DOCO Detection 2665 Frequency, cm-1 Minimum detectable DOCO concentration (3 cm pathlength) 5 x 1010 cm-3 in 960 shots Fe (using ab initio band strength with quartic f. f. ) VIPA Bandwidth = 1. 93 THz (65 cm-1) Resolution = 1 GHz (0. 03 cm-1) 275
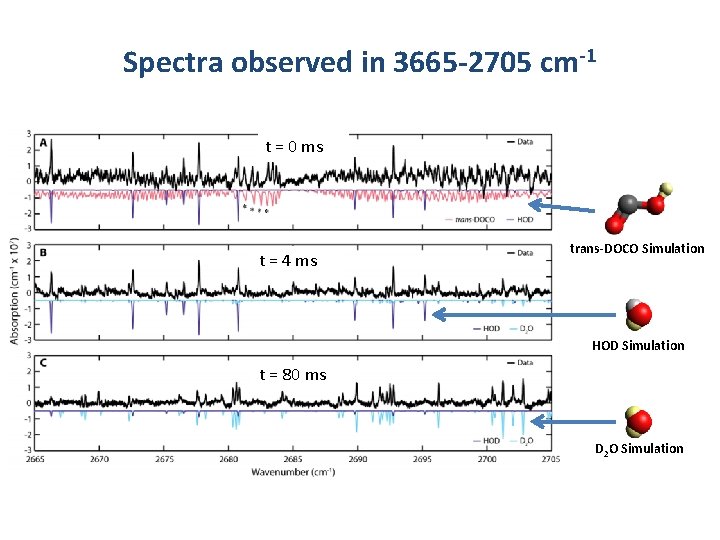
Spectra observed in 3665 -2705 cm-1 t = 0 ms t = 4 ms trans-DOCO Simulation HOD Simulation t = 80 ms D 2 O Simulation
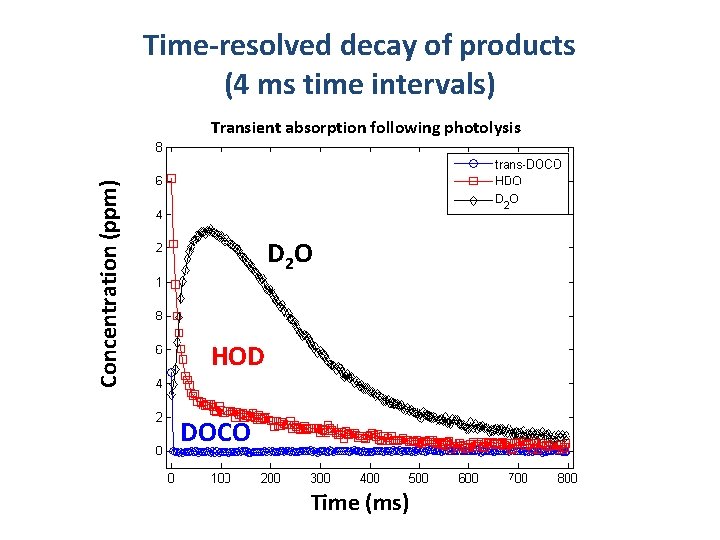
Time-resolved decay of products (4 ms time intervals) Concentration (ppm) Transient absorption following photolysis D 2 O HOD DOCO Time (ms)
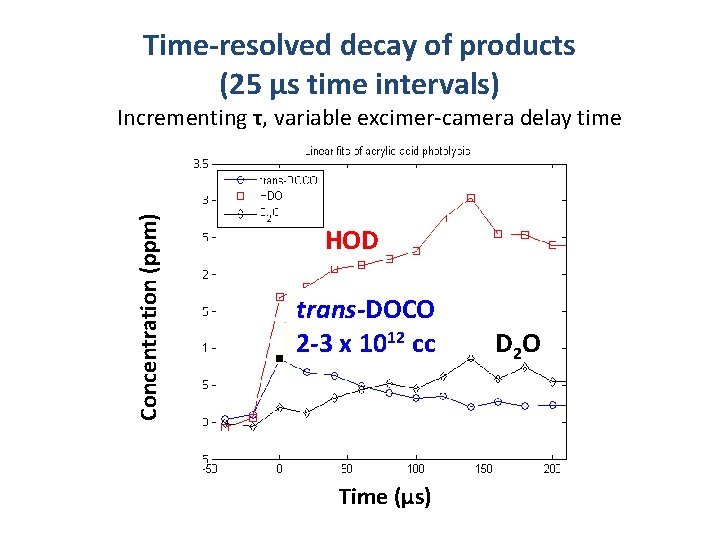
Time-resolved decay of products (25 μs time intervals) HOD trans-DOCO 2 -3 x 1012 cc Average Concentration (ppm) Incrementing τ, variable excimer-camera delay time Time (µs) D 2 O
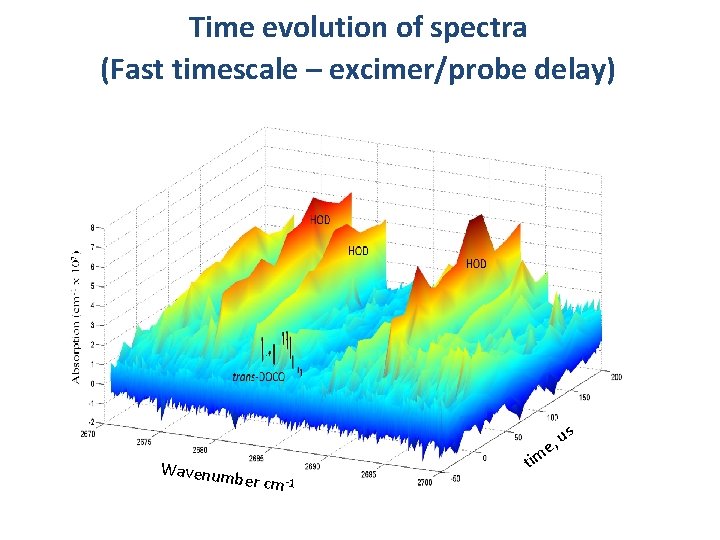
Time evolution of spectra (Fast timescale – excimer/probe delay) Wavenum us , e ber cm -1 tim
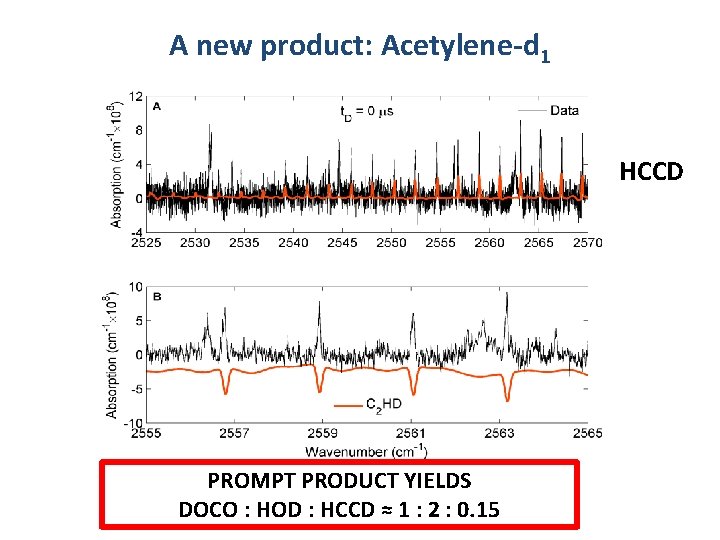
A new product: Acetylene-d 1 HCCD PROMPT PRODUCT YIELDS DOCO : HOD : HCCD ≈ 1 : 2 : 0. 15

Previous Observations on UV Photolysis of Acrylic Acid C 3 C 1 D C 3 -O and C 2 -C 3 bond fission C 2 Why do we observe prompt HOD? HCCD? ? Bulk experiments (UV photolysis) HOCO (+ products) C-C bond fission CO 2 (+ products) Decarboxylation CO (+ products) Decarbonylation OH (+ products) C-O bond fission Molecular Beam Dissociation (Butler & co-workers) HOCO + C 2 H 3 OH + C 3 H 3 O

What are the sources of isotope exchange products? C 3 C 1 C 2 DOCO HOD, HCCD ? ? • Secondary chemistry? No. Bimolecular reactions cannot occur in first 50 us at these concentrations • Wall reactions? No. (probe volume 1 cm from walls) • Water impurities? AA-H 2 O clusters? No. All HOD converts to D 2 O, system is passivated. • Impure precursors (exchange on walls) possibly!! • AA dimers or clusters? PAA = 20 m. Torr is too low, but Acrylic acid is VERY STICKY Possibly primary AA photochemistry? ?
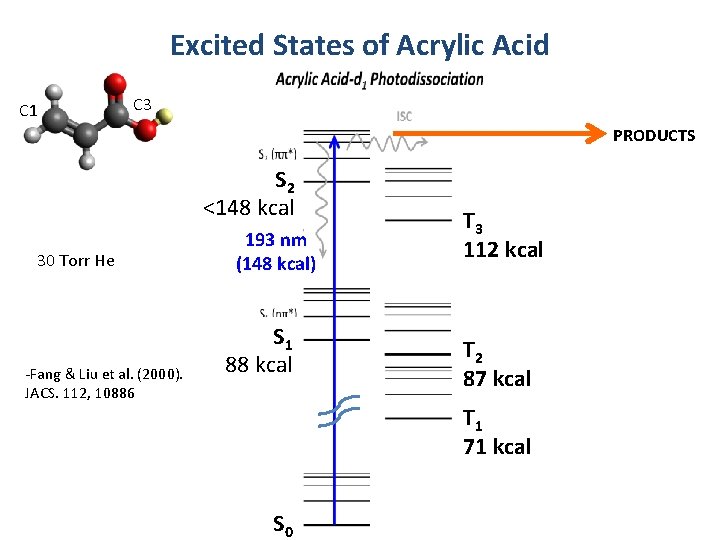
Excited States of Acrylic Acid C 1 C 3 PRODUCTS S 2 <148 kcal 30 Torr He -Fang & Liu et al. (2000). JACS. 112, 10886 193 nm (148 kcal) S 1 88 kcal T 3 112 kcal T 2 87 kcal T 1 71 kcal S 0

Acrylic Acid Photolysis at 193 nm: Ab Initio Dynamics on the Excited Singlet S 2 State Dorit Shemesh and R. Benny Gerber Hebrew University, UC Irvine and University of Helsinki

Methodology: Dynamics on S 2 surface (only) Semi-empirical OM 2 and OM 2/MRCI potentials Ground State (300 K) (initial wavepacket): optimized with OM 2 Molecular dynamics on OM 2/MRCI surface (10 ps) 100 structures as initial configurations Assume Vertical Transition Excited State Molecular dynamics on OM 2/MRCI surface (100 ps)
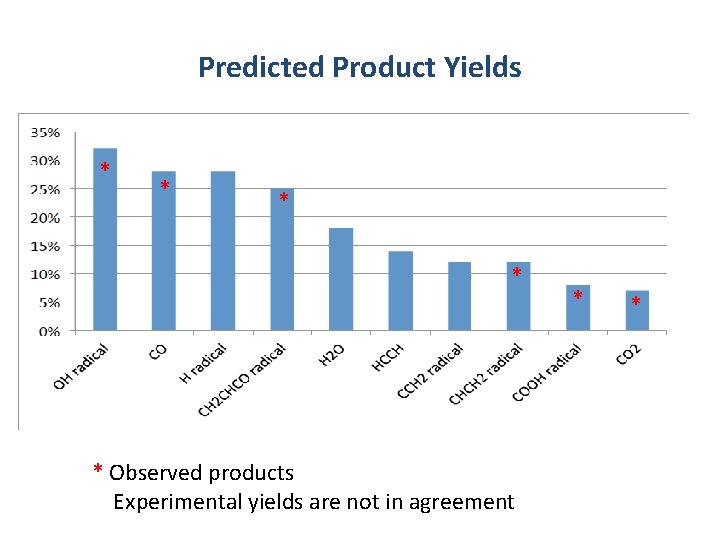
Predicted Product Yields * * * Observed products Experimental yields are not in agreement * *
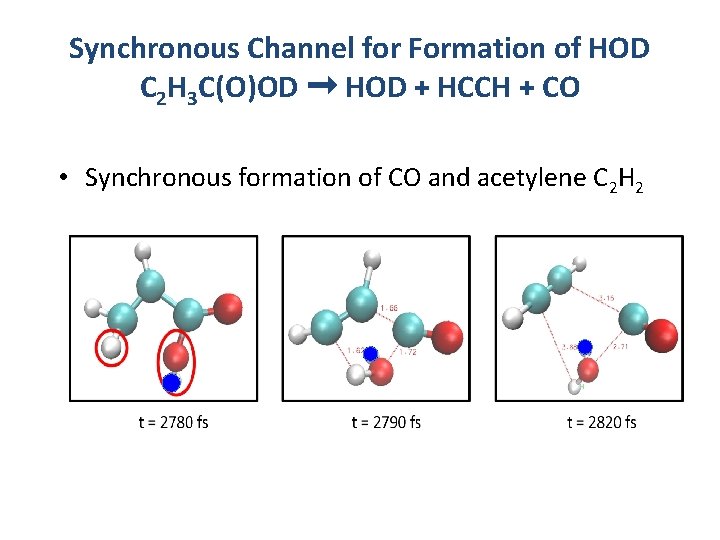
Synchronous Channel for Formation of HOD C 2 H 3 C(O)OD ➞ HOD + HCCH + CO • Synchronous formation of CO and acetylene C 2 H 2
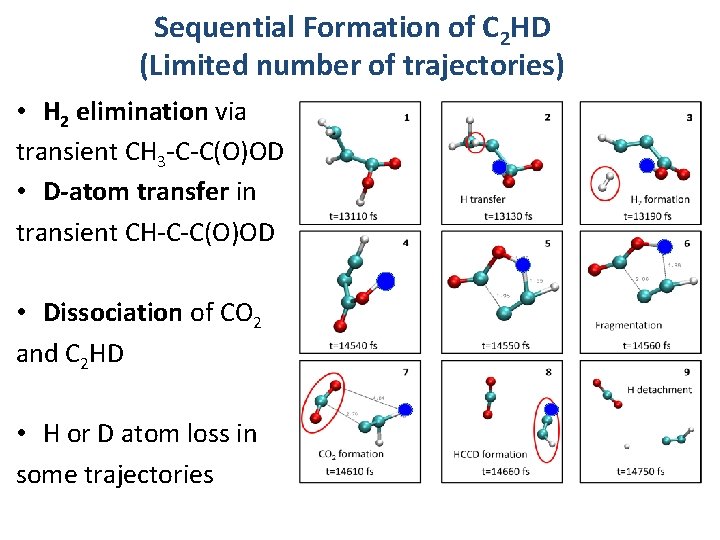
Sequential Formation of C 2 HD (Limited number of trajectories) • H 2 elimination via transient CH 3 -C-C(O)OD • D-atom transfer in transient CH-C-C(O)OD • Dissociation of CO 2 and C 2 HD • H or D atom loss in some trajectories

Caveats Semi-empirical S 2 surface, corrected Classical trajectories Dynamics on single excited state surface No surface crossings to other states: Internal conversion (IC) to S 1, S 0 Intersystem crossing (ISC) to T 2, T 1 • Experiment needs to be repeated • •

Summary: Mid-IR Time-Resolved Frequency Comb Spectroscopy • Broadband absorption spectra in the mid-IR Tuning Range 2. 8 -4. 8 µ Single-shot bandwidth 65 cm-1 (VIPA-limited) • High sensitivity approaching single mode cw-CRDS DOCO sensitivity 5 x 1010/cm-3 in 960 shots (limited by flow cell) 3 cm path length • VIPA-limited spectral resolution ≥ 0. 018 cm-1 • Good time-resolution 10 - 20 µs Camera integration, Cavity response time Full time • Cavity-enhanced sensitivity

Caveats for MIR TRFCS • Best sensitivity for narrow lines (rotationally resolved) • Mid-IR experiments limited by detection system – In. Sb Camera has limited sensitivity, range, pixels (and $$$$) – Better performance in NIR, Visible (Faster, cheaper, better) • Cavity Enhanced, not Cavity Ringdown – need to measure ΔI / I 0 • Cannot yet resolve single comb lines

Improvements • Improved Mid-IR mirrors (throughput 20%) • Flow cell (limited to < 1 Hz photolysis rep rate) Other Approaches • Frequency Comb Velocity Modulation (Ye, Cornell) • Dual Frequency Comb Spectroscopy

Acknowledgments JUN YE GROUPJILA|NIST and CU AJ Fleisher, Bryce J. Bjork, Bryan Changala, Ben Spaun, Oliver Heck, Kevin C. Cossel Alexandra Foltynowicz, Piotr Masliowski OKUMURA GROUP Caltech Thinh Bui UC IRVINE/HEBREW UNIVERSITY Dorit Shemesh, R. Benny Gerber

- Slides: 55