Breaking News on Results of Recently Completed Trials

Breaking News on Results of Recently Completed Trials in Progressive MS Fred D. Lublin, M. D. Corinne Goldsmith Dickinson Center for Multiple Sclerosis Icahn School of Medicine at Mount Sinai New York, NY, USA
Disclosures Research Support: Acorda, Biogen Idec, Genzyme, National Institute of Health, National Multiple Sclerosis Society, Novartis, Sanofi, Teva, Transparency Life Sciences, Celgene Consultant: Actelion, Acorda, Bayer, Biogen Idec, EMD Serono, Forward-Pharma, Genentech, Genzyme, Johnson & Johnson, Med. Immune, Novartis, Receptos, Revalesio, Roche, Sanofi-aventis, Questcor, Teva, to-BBB technologies, Xeno. Port, Abbvie, Toyama, Akros Other: Genzyme Shareholder: Cognition Pharmaceuticals Discussion of Off-Label, Investigational, or Experimental Drug Use: Disclosed

Breaking News: We are making headway Door is open to solving the problem of progressive MS But…. . The challenges

Where we are Successful studies ORATORIO Biotin: 1 for 2 Simvastatin Decent pipeline Siponimod Mastinib Ibudilast Idebenone GZ 402668 Progressive MS Alliance

Challenges in Study Design What to target Inflammation Degeneration Who to target Age, sex, duration of disease, activity (prior and during) Best end-point confirmed vs. sustained Event rates Duration of study

Targeting Unmet Needs: Progressive Disease What have we learned from prior studies? PROMISE OLYMPUS INFORMS ORATORIO ASCEND TRANSFORM How should we measure outcome? Composite scoring EDSS Other, ? cognition Phase II – proof of concept studies – volume?

2013 MS Phenotype Descriptions: Progressive Disease 2013 MS disease modifiers phenotypes Active* and with progression† Active but without progression Progressive disease (PP, SP) Not active but with progression Not active and without progression (stable disease) *Activity = clinical relapses and/or MRI (Gd-enhancing MRI lesions; new/enlarging T 2 lesions) †Progression measured by clinical evaluation at least annually Gd, gadolinium; MRI, magnetic resonance imaging
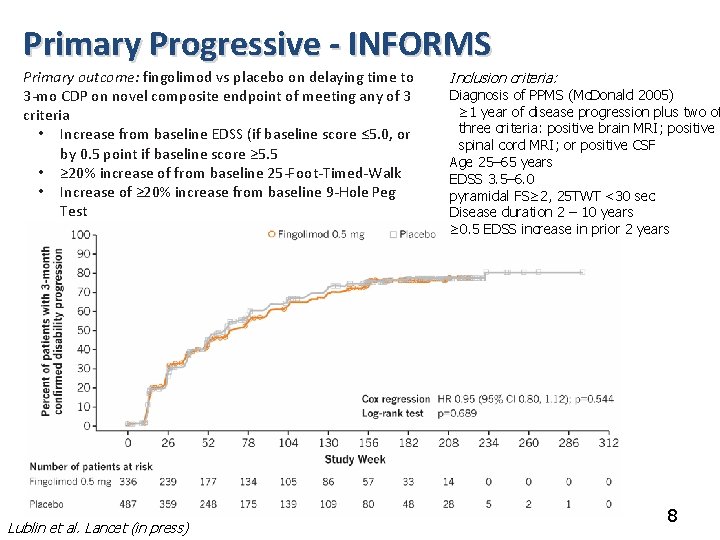
Primary Progressive - INFORMS Primary outcome: fingolimod vs placebo on delaying time to 3 -mo CDP on novel composite endpoint of meeting any of 3 criteria • Increase from baseline EDSS (if baseline score ≤ 5. 0, or by 0. 5 point if baseline score ≥ 5. 5 • ≥ 20% increase of from baseline 25 -Foot-Timed-Walk • Increase of ≥ 20% increase from baseline 9 -Hole Peg Test Lublin et al. Lancet (in press) Inclusion criteria: Diagnosis of PPMS (Mc. Donald 2005) ≥ 1 year of disease progression plus two of three criteria: positive brain MRI; positive spinal cord MRI; or positive CSF Age 25– 65 years EDSS 3. 5– 6. 0 pyramidal FS≥ 2, 25 TWT <30 sec Disease duration 2 – 10 years ≥ 0. 5 EDSS increase in prior 2 years 8
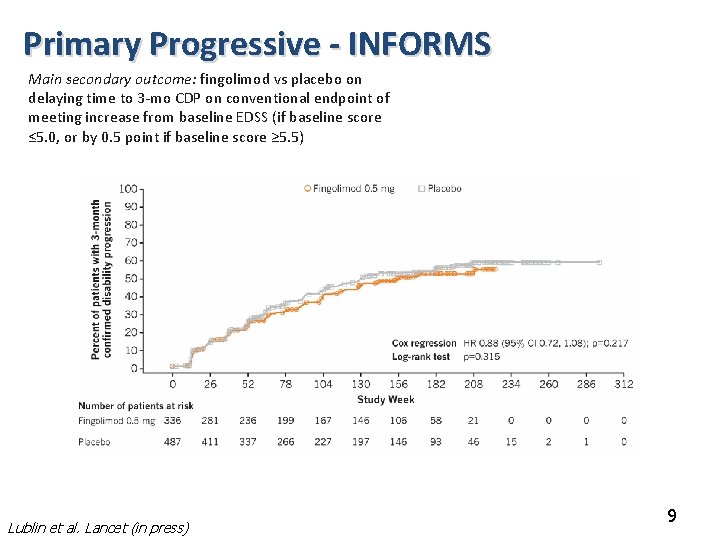
Primary Progressive - INFORMS Main secondary outcome: fingolimod vs placebo on delaying time to 3 -mo CDP on conventional endpoint of meeting increase from baseline EDSS (if baseline score ≤ 5. 0, or by 0. 5 point if baseline score ≥ 5. 5) Lublin et al. Lancet (in press) 9
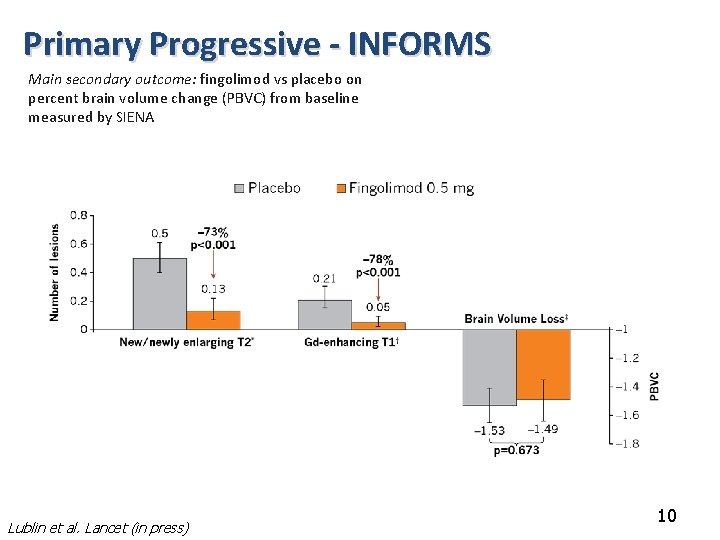
Primary Progressive - INFORMS Main secondary outcome: fingolimod vs placebo on percent brain volume change (PBVC) from baseline measured by SIENA Lublin et al. Lancet (in press) 10

A Clinical Study of the Efficacy of Natalizumab on Reducing Disability Progression in Participants With SPMS (ASCEND in SPMS) (NCT 01416181) • Primary Objective (Part 1): To investigate whether treatment with natalizumab slows the accumulation of disability not related to relapses in participants with SPMS. Primary endpoint Percentage of patients with CDP (confirmed at a second visit ≥ 6 months later and at week 96 using one or more of the assessments) – Primary composite endpoint was not met • • • EDSS score: increased ≥ 1 point if baseline EDSS ≤ 5. 5, or ≥ 0. 5 point if baseline EDSS ≥ 6. 0 T 25 FW: ≥ 20% increase from baseline in the time taken for the 25 -foot walk 9 HPT: ≥ 20% increase from baseline in the time taken for the 9 HPT (increase must be confirmed in the same hand) – Significant improvement in 9 HPT (only) demonstrated with natalizumab vs placebo Secondary endpoints (2 -year endpoints) Percentage of participants with a T 25 -FW response (improvement from the best pre-dose T 25 -FW in at least 75% of the scheduled on-treatment visits Change in the 12 -Item MS Walking Scale (MSWS-12) Change in manual ability score based on the ABILHAND Questionnaire Change in in the Multiple Sclerosis Impact Scale-29 Physical (MSIS-29 Physical) score) Change from week 24 in whole brain volume Percentage of participants with confirmed worsening in individual EDSS Physical Functional System scores (≥ 1 point increase if baseline EDSS ≤ 5. 5 or ≥ 0. 5 point increase if baseline EDSS > 5. 5). https: //clinicaltrials. gov/ct 2/show/NCT 01416181. US Medical Affairs / Genentech Confidential — Do not copy, distribute or use without prior written consent
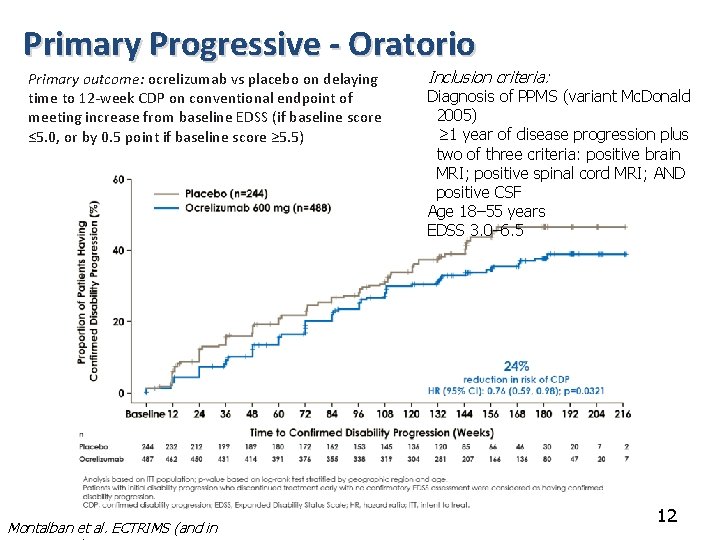
Primary Progressive - Oratorio Primary outcome: ocrelizumab vs placebo on delaying time to 12 -week CDP on conventional endpoint of meeting increase from baseline EDSS (if baseline score ≤ 5. 0, or by 0. 5 point if baseline score ≥ 5. 5) Montalban et al. ECTRIMS (and in Inclusion criteria: Diagnosis of PPMS (variant Mc. Donald 2005) ≥ 1 year of disease progression plus two of three criteria: positive brain MRI; positive spinal cord MRI; AND positive CSF Age 18– 55 years EDSS 3. 0– 6. 5 12

MS-SPI: Study Design ■ Randomized, multicenter, double-blind, placebo-controlled study to evaluate the efficacy of oral biotin 300 mg/day in patients with SPMS or PPMS over 1 year ■ Key eligibility criteria • Age 18– 75 years • PPMS or SPMS • Evidence of disease progression in the preceding 2 years • EDSS 4. 5– 7. 0 Tourbah, et al. AAN 2015 Washington, DC.

MS-SPI: Endpoints ■ Primary endpoint: Proportion of patients who improved at month 9 and confirmed at month 12, defined as decreased EDSS (by at least 1 point for EDSS ≤ 5. 5 and. 5 point for EDSS ≥ 6) or improved TW 25 of ≥ 20% ■ Secondary endpoints: • MS Walking Scale • Clinical Global Impressions Scale (CGI) • Proportion of patients with stable or worsened EDSS • SF-36 • Fatigue Impact Scale • 9 HPT Tourbah, et al. AAN 2015 Washington, DC.
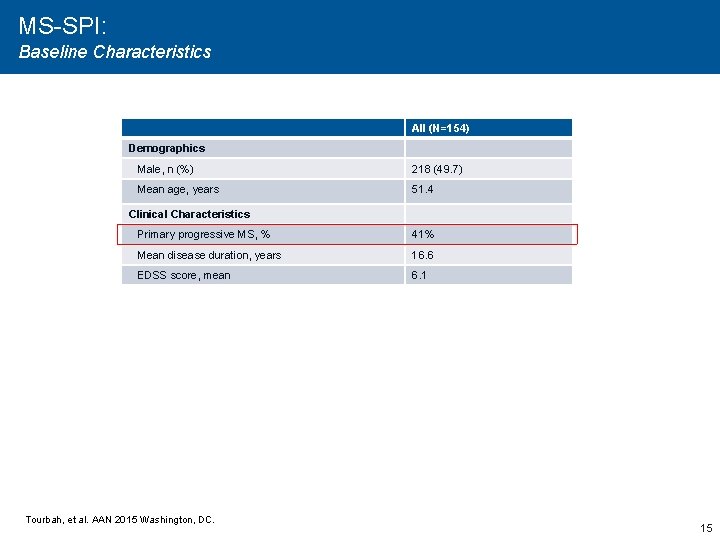
MS-SPI: Baseline Characteristics All (N=154) Demographics Male, n (%) 218 (49. 7) Mean age, years 51. 4 Clinical Characteristics Primary progressive MS, % 41% Mean disease duration, years 16. 6 EDSS score, mean 6. 1 Tourbah, et al. AAN 2015 Washington, DC. 15

MS-SPI: Efficacy ■ 13% of patients receiving MD 1003 met the primary endpoint vs 0% in placebo arm; p=0. 0051 ■ Patients receiving MD 1003 had -0. 3 mean change in EDSS at 1 year vs 0. 13 for placebo; p<0. 014 ■ 4% of patients receiving MD 1003 progressed at 1 year vs 13. 6% for placebo; p<0. 0727 ■ No difference in TW 25 ■ The proportion of responders was higher in patients with • Baseline EDSS score of 4. 5– 5 vs 6– 7; (21. 4% vs 9. 3%) • No concomitant fampridine administration vs concomitant fampridine administration (20. 7% vs 2. 2%) and • SPMS, compared to PPMS (14. 8% vs 9. 5%). Tourbah, et al. AAN 2015 Washington, DC; Tourbah et al. ECTRIMS 2015 Barcelona, Spain.

LAQ in PPMS Study underway but high dose arm (1. 5 mg/day) dropped for safety

Conclusions We are making progress with progressive disease We learn from each study I wish I had more breaking news to report We are developing a clearer understanding of how to more effectively approach progression
- Slides: 18