Break Ionic Nomenclature into two sections binary ionic

• Break Ionic Nomenclature into two sections – binary ionic (with different oxidation states and a HW) – polyatomic/oxyanion (with own HW) slide 1

Do Now Homework Check Write the correct formula(s) for the compounds formed between atoms listed 1) bromine and lead Pb. Br 2 & Pb. Br 4 2) chromium and sulfur Cr. S & Cr 2 S 3 3) gold and nitrogen Au 3 N & Au. N slide 2

Chemistry Worksheet - Oxidation Numbers and Chemical Formulas 1) lithium and fluorine Li. F 2) rubidium and sulfur Rb 2 S 3) magnesium and bromine Mg. Br 2 4) strontium and oxygen Sr. O 5) sodium and nitrogen Na 3 N 6) scandium and iodine Sc. I 3 7) phosphorus and barium Ba 3 P 2 8) boron and oxygen B 2 O 3 9) nitrogen and yttrium YN 10) carbon and chlorine CCl 4 slide 3

Chemistry Worksheet - Oxidation Numbers and Chemical Formulas 11) silver and iodine Ag. I 12) nickel and chlorine Ni. Cl 2 13) oxygen and gold Au 2 O and Au 2 O 3 Ga 3 P 2 and Ga. P 14) gallium and phosphorus 15) fluorine and chromium 16) lead and oxygen 17) indium and selenium Cr. F 2 and Cr. F 3 Pb. O and Pb. O 2 In 2 Se and In 2 Se 3 18) chlorine and copper Cu. Cl and Cu. Cl 2 19) mercury and bromine Hg 2 Br 2 and Hg. Br 2 20) titanium and arsenic Ti 3 As 2 and Ti. As slide 4

Nomenclature • Oxidation States and Writing Formulas • Ionic Nomenclature • Covalent Nomenclature • Acid Nomenclature slide 5

Ionic Nomenclature Write this in your notes SWBAT convert back and forth between formulas and names of: 1) binary ionic compounds 2) polyatomic ionic compounds 3) oxyanion ionic compounds slide 6
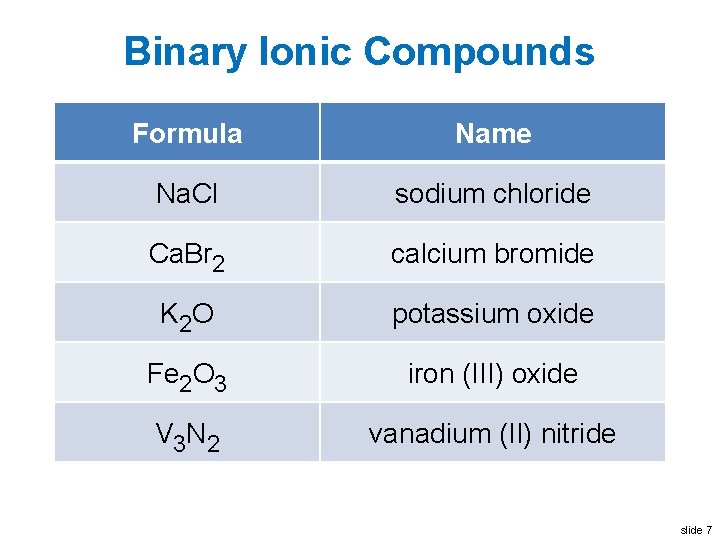
Binary Ionic Compounds Formula Name Na. Cl sodium chloride Ca. Br 2 calcium bromide K 2 O potassium oxide Fe 2 O 3 iron (III) oxide V 3 N 2 vanadium (II) nitride slide 7

Binary Ionic Compounds Write this in your notes • Ionic compounds made from just two elements • Formula Format: Cation. CAnion. A • where: Cation = element symbol for cation Anion = element symbol for anion C = number of cation atoms A = number of anion atoms slide 8

Ionic Compounds Nomenclature cation anion 1) Cation comes before anion slide 9

Ionic Compounds Nomenclature cation anion 1) Cation comes before anion 2) Cation uses its element name 3) Anion uses its element stem plus "-ide" slide 10

Ionic Compounds Nomenclature cation anion-ide 1) Cation comes before anion 2) Cation uses its element name 3) Anion uses its element stem plus "-ide" 4) If the cation has multiple oxidation numbers, the number in this compound is indicated by Roman numerals in parentheses slide 11

Ionic Compounds Nomenclature cation (oxidation state) anion-ide 1) Cation comes before anion 2) Cation uses its element name Write this in your notes 3) Anion uses its element stem plus "-ide" 4) If the cation has multiple oxidation numbers, the number in this compound is indicated by Roman numerals in parentheses 5) Polyatomic ions and oxyanions are used with no modification slide 12

Example 1 - Na. Cl 1) Cation comes before anion 2) Cation uses its element name sodium chlorine 3) Anion uses its element stem plus "-ide" sodium chlor– sodium chloride 4) If the cation has multiple oxidation numbers, the number in this compound is indicated by Roman numerals in parentheses 5) Polyatomic ions and oxyanions are used with no modification slide 13

Example 2 - Ca. Br 2 1) Cation comes before anion 2) Cation uses its element name calcium bromine 3) Anion uses its element stem plus "-ide" calcium brom– calcium bromide Notice there is nothing in the name to indicate two bromines. The reader is expected to be able to determine the ratio by themselves 4) If the cation has multiple oxidation numbers, the number in this compound is indicated by Roman numerals in parentheses 5) Polyatomic ions and oxyanions are used with no modification slide 14

Example 3 - Fe 2 O 3 1) Cation comes before anion 2) Cation uses its element name iron oxygen 3) Anion uses its element stem plus "-ide" iron ox– iron oxide 4) If the cation has multiple oxidation numbers, the number in this compound is indicated by Roman numerals in parentheses You have to determine iron (III) oxide 5) Polyatomic ions and oxyanions are used with no modification iron's oxidation number from the formula slide 15

Check for Understanding 1 Write the correct names for the formula: 1) Mg. O magnesium oxide 2) Ca 3 N 2 calcium nitride 3) VF 3 vanadium (III) fluoride slide 16

Check for Understanding 2 Write the correct formulas for the names 1) lithium sulfide Li 2 S 2) gallium (II) phosphide Ga 3 P 2 3) gold (III) oxide Au 2 O 3 slide 17

Do Now Fill in the missing formula or name Formula 1 2 Name calcium oxide Rb. Br 3 gold (I) chloride 4 gold (III) chloride 5 Ag. Cl 6 manganese (III) oxide 7 Ti 3 N 2 8 Hg 2 F 2 slide 18

Ionic Compounds Nomenclature cation (oxidation state) anion-ide 1) Cation comes before anion 2) Cation uses its element name Review from last class 3) Anion uses its element stem plus "-ide" 4) If the cation has multiple oxidation numbers, the number in this compound is indicated by Roman numerals in parentheses 5) Polyatomic ions and oxyanions are used with no modification slide 19

Do Now Fill in the missing formula or name Formula 1 2 Name calcium oxide Rb. Br 3 gold (I) chloride 4 gold (III) chloride 5 Ag. Cl 6 manganese (III) oxide 7 Ti 3 N 2 8 Hg 2 F 2 slide 20

Do Now Fill in the missing formula or name Formula Name 1 Ca. O calcium oxide 2 Rb. Br 3 gold (I) chloride 4 gold (III) chloride 5 Ag. Cl 6 manganese (III) oxide 7 Ti 3 N 2 8 Hg 2 F 2 slide 21

Do Now Fill in the missing formula or name Formula Name 1 Ca. O calcium oxide 2 Rb. Br rubidium bromide 3 gold (I) chloride 4 gold (III) chloride 5 Ag. Cl 6 manganese (III) oxide 7 Ti 3 N 2 8 Hg 2 F 2 slide 22

Do Now Fill in the missing formula or name Formula Name 1 Ca. O calcium oxide 2 Rb. Br rubidium bromide 3 Au. Cl gold (I) chloride 4 5 gold (III) chloride Ag. Cl 6 manganese (III) oxide 7 Ti 3 N 2 8 Hg 2 F 2 slide 23

Do Now Fill in the missing formula or name Formula Name 1 Ca. O calcium oxide 2 Rb. Br rubidium bromide 3 Au. Cl gold (I) chloride 4 Au. Cl 3 gold (III) chloride 5 Ag. Cl 6 manganese (III) oxide 7 Ti 3 N 2 8 Hg 2 F 2 slide 24

Do Now Fill in the missing formula or name Formula Name 1 Ca. O calcium oxide 2 Rb. Br rubidium bromide 3 Au. Cl gold (I) chloride 4 Au. Cl 3 gold (III) chloride 5 Ag. Cl silver chloride or silver (I) chloride 6 manganese (III) oxide 7 Ti 3 N 2 8 Hg 2 F 2 slide 25

Do Now Fill in the missing formula or name Formula Name 1 Ca. O calcium oxide 2 Rb. Br rubidium bromide 3 Au. Cl gold (I) chloride 4 Au. Cl 3 gold (III) chloride 5 Ag. Cl silver chloride or silver (I) chloride 6 Mn 2 O 3 manganese (III) oxide 7 Ti 3 N 2 8 Hg 2 F 2 slide 26

Do Now Fill in the missing formula or name Formula Name 1 Ca. O calcium oxide 2 Rb. Br rubidium bromide 3 Au. Cl gold (I) chloride 4 Au. Cl 3 gold (III) chloride 5 Ag. Cl silver chloride or silver (I) chloride 6 Mn 2 O 3 manganese (III) oxide 7 Ti 3 N 2 titanium (II) nitride 8 Hg 2 F 2 slide 27

Do Now Fill in the missing formula or name Formula Name 1 Ca. O calcium oxide 2 Rb. Br rubidium bromide 3 Au. Cl gold (I) chloride 4 Au. Cl 3 gold (III) chloride 5 Ag. Cl silver chloride or silver (I) chloride 6 Mn 2 O 3 manganese (III) oxide 7 Ti 3 N 2 titanium (II) nitride 8 Hg 2 F 2 mercury (I) fluoride slide 28

Polyatomic Ions Write this in your notes • Ions made up of more than one atom. • The atoms in a polyatomic ion are usually covalently bonded together • Examples: OH– 1 NH 4+1 CN– 1 slide 29

Oxyanions Write this in your notes • Polyatomic ions composed of an element bonded to one or more oxygen atoms • Examples PO 4– 3 SO 4– 2 Cr 2 O 7– 2 slide 30
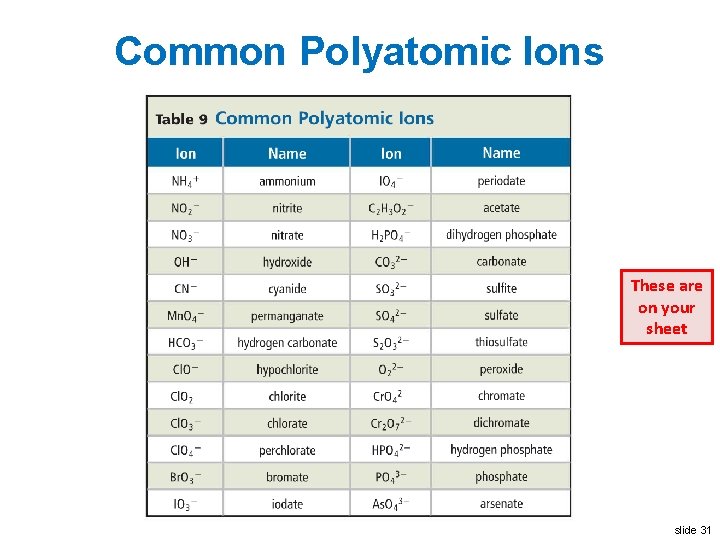
Common Polyatomic Ions These are on your sheet slide 31

Oxyanion Naming Conventions These are on your sheet slide 32

Formulas with Polyatomic Ions • If just one ion present, no parentheses are used NH 4 Cl Na 2 Cr 2 O 7 Write this in your notes • If more than one ion present, the ion is put in parentheses and a subscript indicates the number Pb(CO 3)2 (NH 4)3 PO 4 slide 33

Ionic Compounds Nomenclature cation (oxidation state) anion-ide 1) Cation comes before anion 2) Cation uses its element name 3) Anion uses its element stem plus "-ide" 4) If the cation has multiple oxidation numbers, the number in this compound is indicated by Roman numerals in parentheses 5) Polyatomic ions and oxyanions are used with no modification slide 34

Example 4 - NH 4 F 1) Cation comes before anion 2) Cation uses its element name ammonium fluorine 3) Anion uses its element stem plus "-ide" ammonium fluor– ammonium fluoride 4) If the cation has multiple oxidation numbers, the number in this compound is indicated by Roman numerals in parentheses 5) Polyatomic ions and oxyanions are used with no modification slide 35

Example 5 - Fe 2(CO 3)3 1) Cation comes before anion 2) Cation uses its element name iron carbonate 3) Anion uses its element stem plus "-ide" The polyatomic ions and oxyanions are used with no modification iron carbonate 4) If the cation has multiple oxidation numbers, the number in this compound is indicated by Roman numerals in parentheses iron (III) carbonate 5) Polyatomic ions and oxyanions are used with no modification slide 36

Check for Understanding 3 Write the correct formulas for the names 1) Mg(C 2 H 3 O 2)2 magnesium acetate 2) Na. H 2 PO 4 sodium dihydrogen phosphate 3) Pb(Cl. O 4)4 lead (IV) perchlorate slide 37

Check for Understanding 4 Write the correct formulas for the names 1) copper (I) thiosulfate Cu 2 S 2 O 3 2) ammonium dichromate (NH 4)2 Cr 2 O 7 3) titanium (III) peroxide Ti 2(O 2)3 slide 38

Homework • Worksheet - Ionic Nomenclature slide 39

Latin Names Write this in your notes • Some compounds were given Latin names before systematic nomenclature was created • These are still in use even today • On cations with two primary oxidation states, a suffix indicates the oxidation state • suffix "–ic" means higher oxidation state • suffix "–ous" means lower oxidation state • Most often used on elements with Latin names slide 40

Example of Latin Name • In Latin, iron is called ferrum • This is why its atomic symbol is Fe • Iron has two primary oxidation states, +3 and +2 Ø Fe+3 is called ferric Ø Fe+2 is called ferrous slide 41

What is this called? A ferrous wheel!!! slide 42

Example of Latin Name • In each oxidation state, Fe can be combined with O Ø Fe 2 O 3 is ferric oxide Ø Fe. O is ferrous oxide slide 43
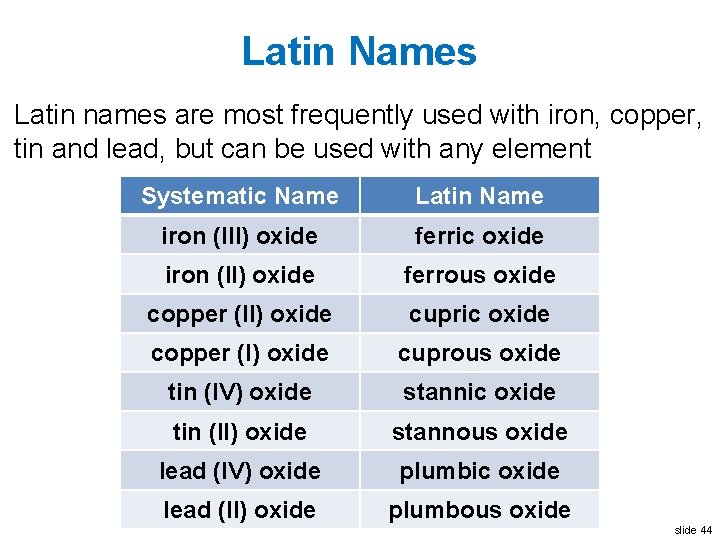
Latin Names Latin names are most frequently used with iron, copper, tin and lead, but can be used with any element Systematic Name Latin Name iron (III) oxide ferric oxide iron (II) oxide ferrous oxide copper (II) oxide cupric oxide copper (I) oxide cuprous oxide tin (IV) oxide stannic oxide tin (II) oxide stannous oxide lead (IV) oxide plumbic oxide lead (II) oxide plumbous oxide slide 44

Do Now Fill in the missing formula or name Formula 1 2 Name calcium oxide Rb. Br 3 gold (I) chloride 4 gold (III) chloride 5 Ag. Cl 6 manganese (III) oxide 7 Ti 3 N 2 8 Hg 2 F 2 slide 45

Do Now Fill in the missing formula or name Formula Name 1 Ca. O calcium oxide 2 Rb. Br rubidium bromide 3 Au. Cl gold (I) chloride 4 Au. Cl 3 gold (III) chloride 5 Ag. Cl 6 manganese (III) oxide 7 Ti 3 N 2 8 Hg 2 F 2 slide 46

Do Now Fill in the missing formula or name Formula Name 1 Ca. O calcium oxide 2 Rb. Br rubidium bromide 3 Au. Cl gold (I) chloride 4 Au. Cl 3 gold (III) chloride 5 Ag. Cl silver chloride or silver (I) chloride 6 Mn 2 O 3 manganese (III) oxide 7 Ti 3 N 2 titanium (II) nitride 8 Hg 2 F 2 mercury (I) fluoride slide 47
- Slides: 47