Branches of Science Biology study of living things

Branches of Science Biology- study of living things. Ecology- study of all living things and their environment. Chemistry- study of elements and their reactions. Physics- Study of matter, energy, & how things move. Geology- study of the earth and how it changes. Astronomy- study of the stars and planets. Meteorology- study of weather.

Scientific Method Problem- Question you want to solve. Hypothesis- Possible solution to the problem. Materials- items you need to experiment. Experimental Procedures- step by step instructions to perform experiment. Observation- taking measurements and using your 5 senses. Conclusion- Answers your hypothesis and tells what happened in the experiment.

Scientific Method Control- standard used to compare to. Constant- things that stay the same. Independent Variable- what the experimenter changes, X-axis. Dependent Variable- what you measure and responds to I. V.

Terms: Science- invention or discovery. Technology- applied science. Inference- conclusion based on an observation or previous knowledge. Observation-taking measurements or using your 5 senses. Theory- experiment tested 1, 000’s of times, explanation as to why something happens. Law- rule in nature Model- representation of an idea or object.

Metric Prefixes kilo- means 1, 000 hecto- 100 deka-10 base units meter, gram, & liter – 1 deci- 1/10 or. 1 centi- 1/100 or. 01 milli- 1/1000 or. 001

Physical and Chemical Properties Color, odor, texture Density, size, phase Luster, hardness Physical Paper crumbled Glass breaking Boiling water Ice melting flammability, corrosion, rust and changes Chemical burning a candle digesting food metal rusting

Tool Length ruler Volume ruler graduated cylinder Unit cm 3 cm m. L Mass - balance scale Temperature – thermometer g °C K °F

Derived or calculated Quantities Formula Units Area = Lx. W Density = mass volume cm 2 g/m. L g/cm 3, or g/cc

States of Matter ex. Solid – def. shape & volume - atoms tightly packed - Move slow Liquid – no def. shape. -Def. volume - atoms loosely packed - move faster than solid Gas- No def. shape nor volume - atoms far apart - move faster than a liquid. Diagram
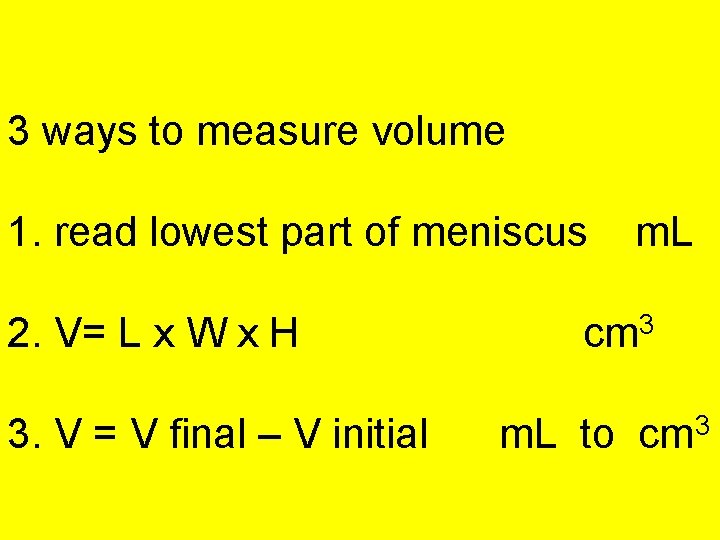
3 ways to measure volume 1. read lowest part of meniscus 2. V= L x W x H 3. V = V final – V initial m. L cm 3 m. L to cm 3

Mass- # of atoms an object has Weight- gravity pulling on mass. Inertia- resistance to movement which depends on mass. Gravity- attractive force between objects that keeps the solar system aligned.

Temperature- how hot or cold an object is measured with a thermometer. Scales 1. Celsius 2. Kelvin 3. Fahrenheit Converting from C to K °C + 273 = K Boiling 100 °C 212 °F Freezing 0 °C 32 °F Room 20°C 70 °F Body 37°C 98. 6 °F
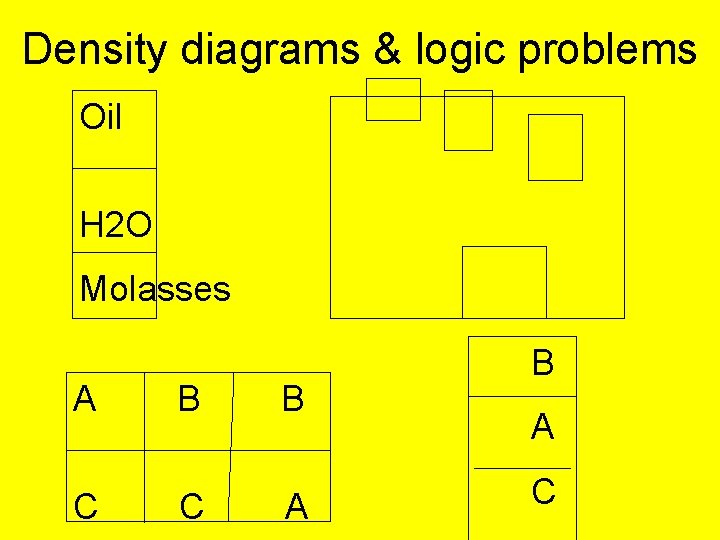
Density diagrams & logic problems Oil H 2 O Molasses A B B C C A B A C

Phase change diagram A = solid F= condensation B = melting C= liquid D = boiling E = gas G = freezing

Melting - solid to liquid Boiling - liquid to gas Condensation – gas to liquid Freezing – liquid to solid Sublimation - solid to gas directly Which absorb energy? Which release energy? Endothermic -absorbs Exothermic- releases

Rounding to the nearest tenth if more that 5 raise the score if less it stays the same 1. 56 1. 23 = 1. 6 cm = 1. 2 cm Include unit whenever possible!
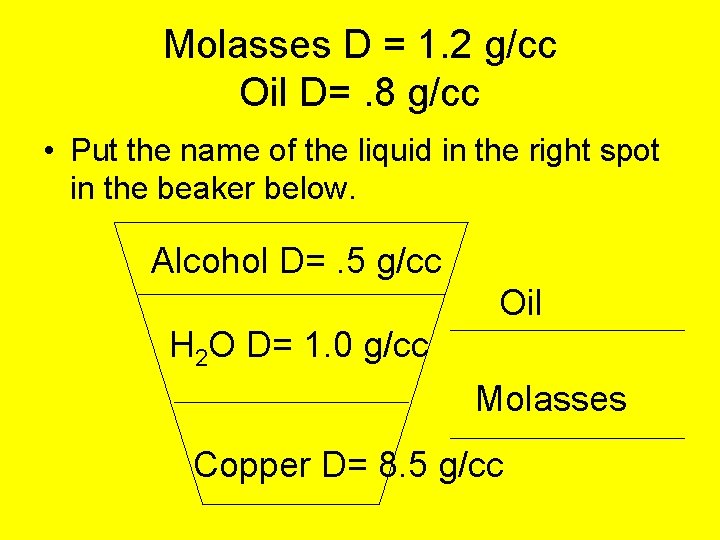
Molasses D = 1. 2 g/cc Oil D=. 8 g/cc • Put the name of the liquid in the right spot in the beaker below. Alcohol D=. 5 g/cc Oil H 2 O D= 1. 0 g/cc Molasses Copper D= 8. 5 g/cc
- Slides: 17