Brain Pop State Changes http www brainpop comsciencemat

Brain Pop State Changes § http: //www. brainpop. com/science/mat terandchemistry/matterchangingstates /

Essential Question § How do I read a phase change diagram?
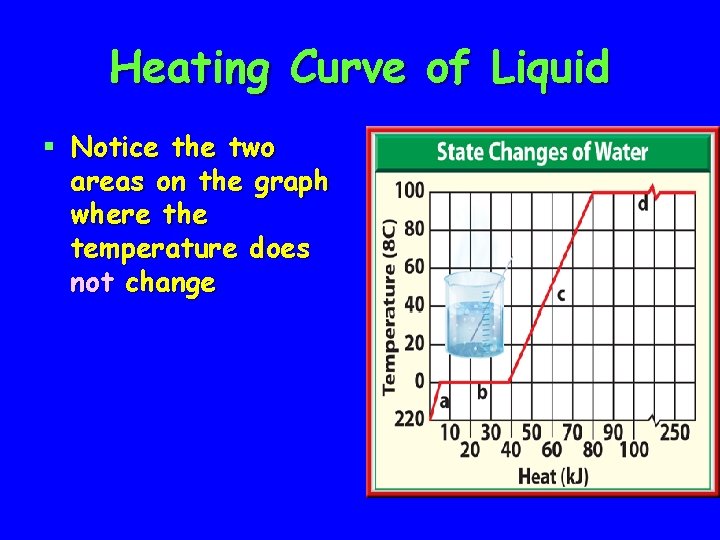
Heating Curve of Liquid § Notice the two areas on the graph where the temperature does not change
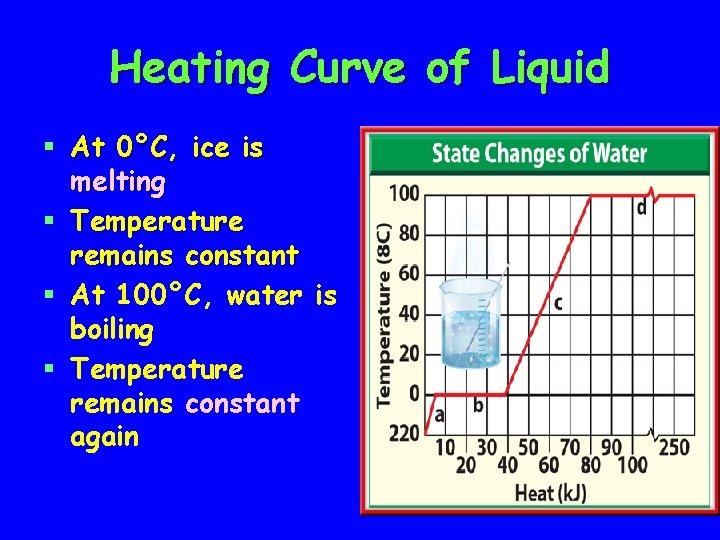
Heating Curve of Liquid § At 0°C, ice is melting § Temperature remains constant § At 100°C, water is boiling § Temperature remains constant again
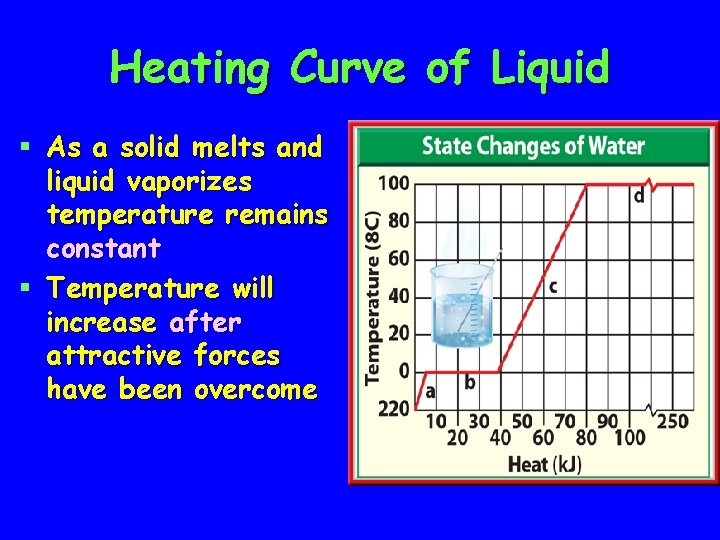
Heating Curve of Liquid § As a solid melts and liquid vaporizes temperature remains constant § Temperature will increase after attractive forces have been overcome
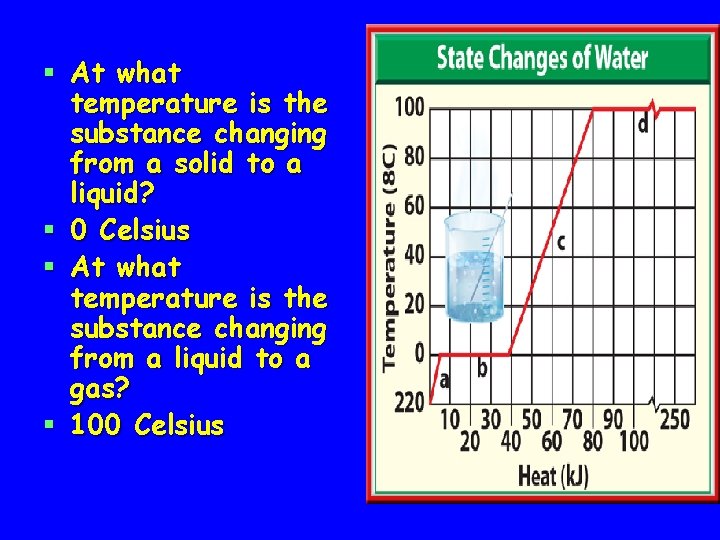
§ At what temperature is the substance changing from a solid to a liquid? § 0 Celsius § At what temperature is the substance changing from a liquid to a gas? § 100 Celsius
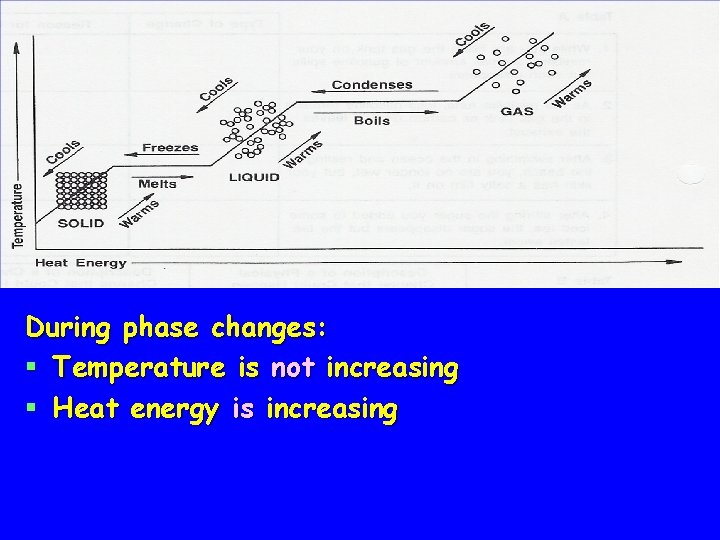
During phase changes: § Temperature is not increasing § Heat energy is increasing
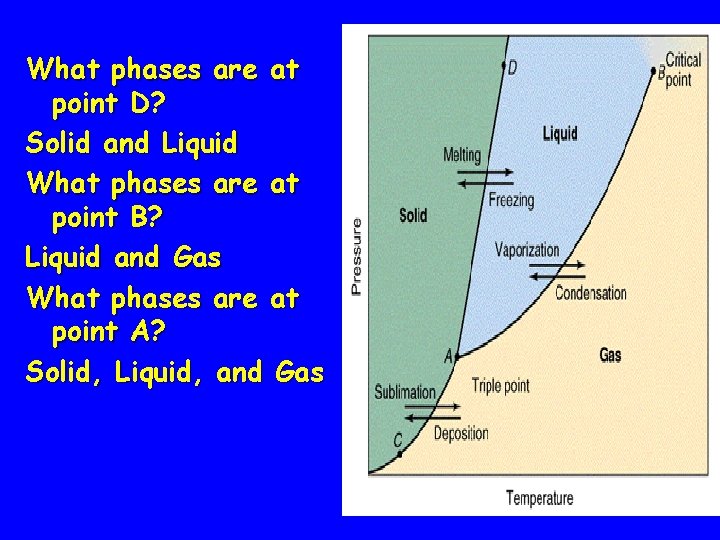
What phases are at point D? Solid and Liquid What phases are at point B? Liquid and Gas What phases are at point A? Solid, Liquid, and Gas
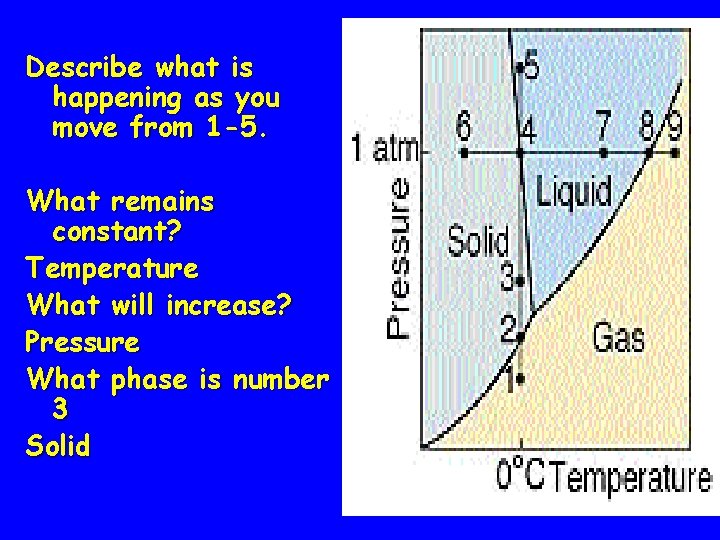
Describe what is happening as you move from 1 -5. What remains constant? Temperature What will increase? Pressure What phase is number 3 Solid
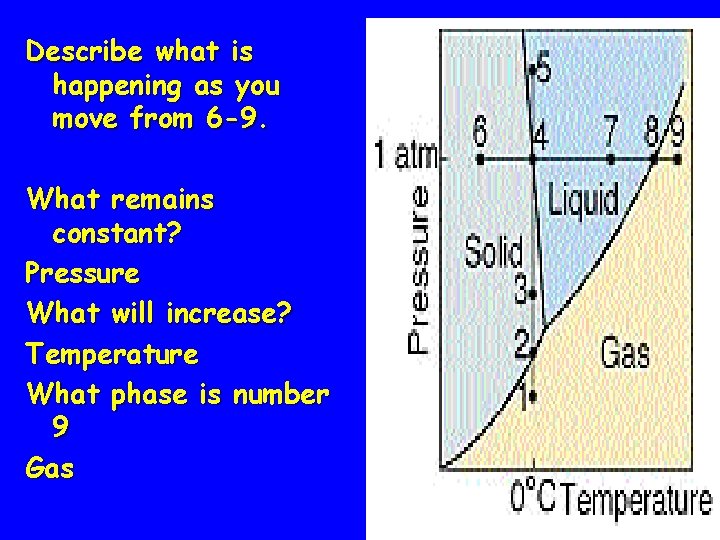
Describe what is happening as you move from 6 -9. What remains constant? Pressure What will increase? Temperature What phase is number 9 Gas

SPS 7 d Daphne placed a glass of water with a thermometer in it inside a freezer. She took temperature readings every 2 minutes for 30 minutes and found that the temperature remained constant during the time interval between the 12 - and 22 minute marks. What is the most likely explanation for this? A The ice was melting during that time. B The water was freezing during that time. C The water’s kinetic energy was increasing. D The water’s kinetic energy was decreasing.

Essential Question § How do pressure, temperature, volume relate to the behavior of gases?

Pressure § Kinetic theory state particles in constantly moving and colliding § Colliding creates pressure § Pressure is measured in a unit called pascal (Pa)

Pressure § Balloon remains inflated because of collisions the air particles have with the walls of their container
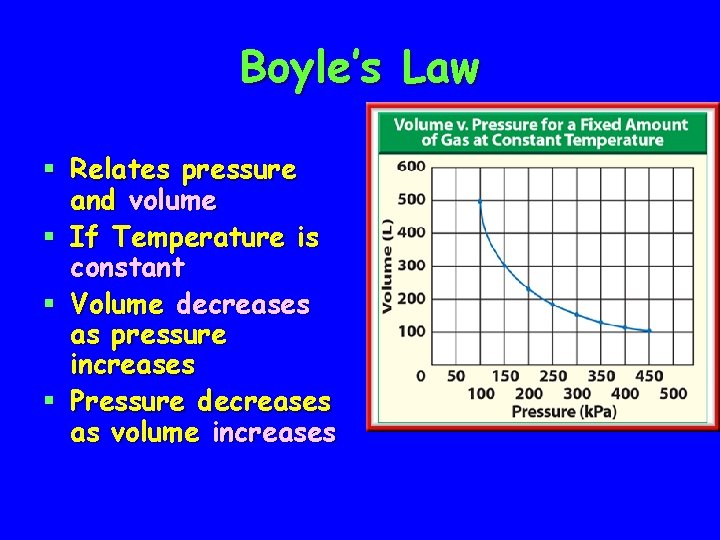
Boyle’s Law § Relates pressure and volume § If Temperature is constant § Volume decreases as pressure increases § Pressure decreases as volume increases
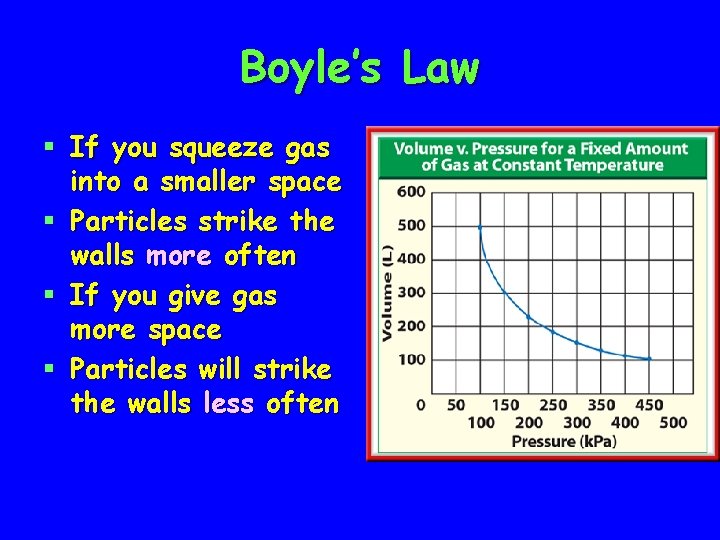
Boyle’s Law § If you squeeze gas into a smaller space § Particles strike the walls more often § If you give gas more space § Particles will strike the walls less often
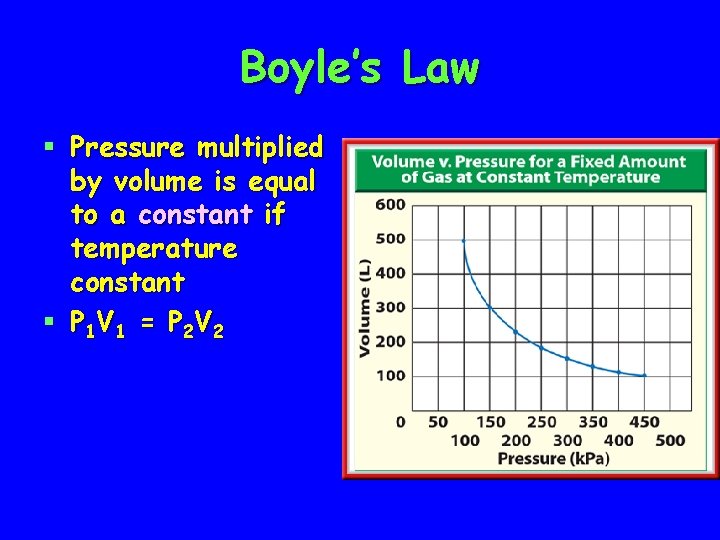
Boyle’s Law § Pressure multiplied by volume is equal to a constant if temperature constant § P 1 V 1 = P 2 V 2
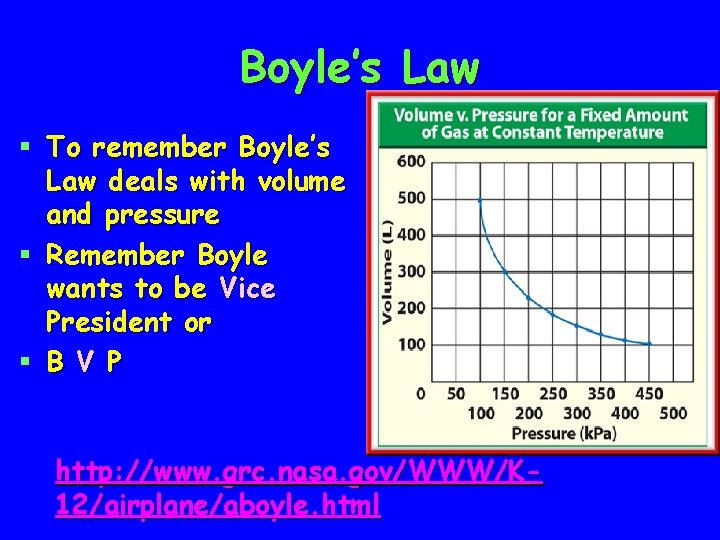
Boyle’s Law § To remember Boyle’s Law deals with volume and pressure § Remember Boyle wants to be Vice President or § B V P http: //www. grc. nasa. gov/WWW/K 12/airplane/aboyle. html
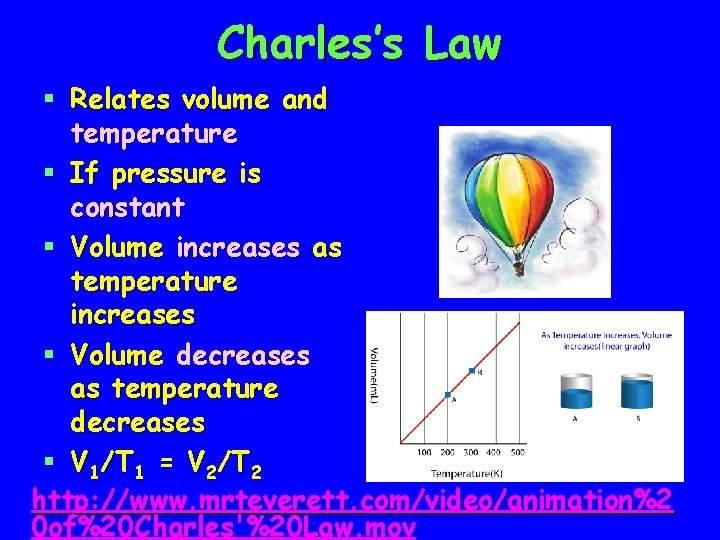
Charles’s Law § Relates volume and temperature § If pressure is constant § Volume increases as temperature increases § Volume decreases as temperature decreases § V 1/T 1 = V 2/T 2 http: //www. mrteverett. com/video/animation%2 0 of%20 Charles'%20 Law. mov

Charles's Law § To remember Charles's law deals with volume and temperature § Remember Charle’s like to watch TV § Or CTV § To remember the direct relationship think Direct TV http: //www. grc. nasa. gov/WW W/K-12/airplane/aglussac. html

SPS 5 b Use the information below to answer question 2. Temperature and Volume of Air at Constant Pressure Temperature Volume 298 K 100 L 358 K 120 L 447 K 150 L 536 K ? 2. Assuming conditions remain the same, what is the final volume of the air will be A Larger than 150 L B Smaller than 150 L C Equal to 150 L D None of the above

SPS 5 b 3. According to Charles’s Law, if the pressure is held constant what will result from an increase in temperature? A. Nothing B. Decrease in volume C. Increase in volume D. None of the above
- Slides: 22