BRAIN DUMP PERIODIC TABLE Metalsnonmetalsmetalloids Groupsperiods Valence electrons

BRAIN DUMP PERIODIC TABLE

/Metals/non-metals/metalloids /Groups/periods /Valence electrons /Most reactive-metals/nonmetals /Most Stable /APEMAN /Protons/electrons/atomic number /Physical Properties

Are you Smarter Than The Periodic Table?

You will need a Periodic Table!

Brain Dump on your Periodic Table - Record the # of valence electrons for groups 1, 2 and 13 – 18 Set up: APE MAN Write any other notes that will help you use the periodic table

What element is found in Group 2, Period 4? 20 Ca 40. 08 Calcium

What element has one valence electron and two energy levels? 3 Li 6. 941 Lithium

What element is found in Group 18, Period 1? 2 He 4. 0026 Helium

What element has one valence electron and 4 energy levels? 19 K 39. 098 Potassium
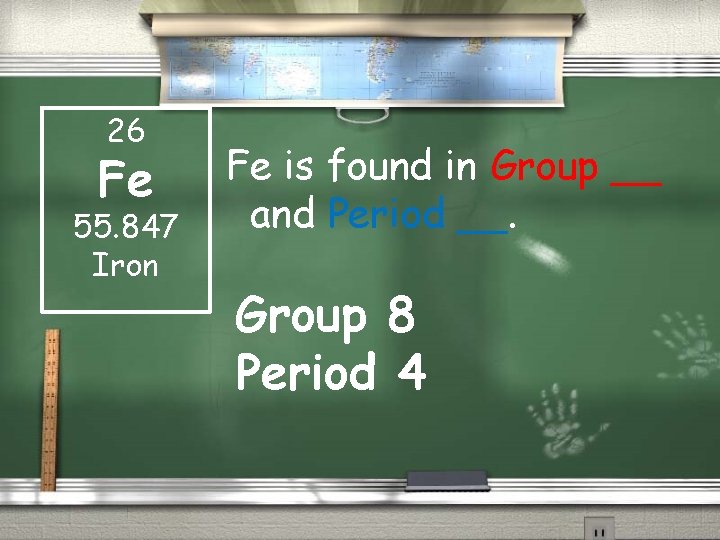
26 Fe 55. 847 Iron Fe is found in Group __ and Period __. Group 8 Period 4

The elements in Group 18 are what type of elements ______? Noble Gases
The elements in Group 6 are what type of elements ____? Metals

The elements that make up the periodic table are mostly in what state of matter? Solids

The elements that make up the periodic table are mostly _____. Metals
What group is more reactive: group 1 or group 2? Group 1 Reactivity lessens as you move to the right
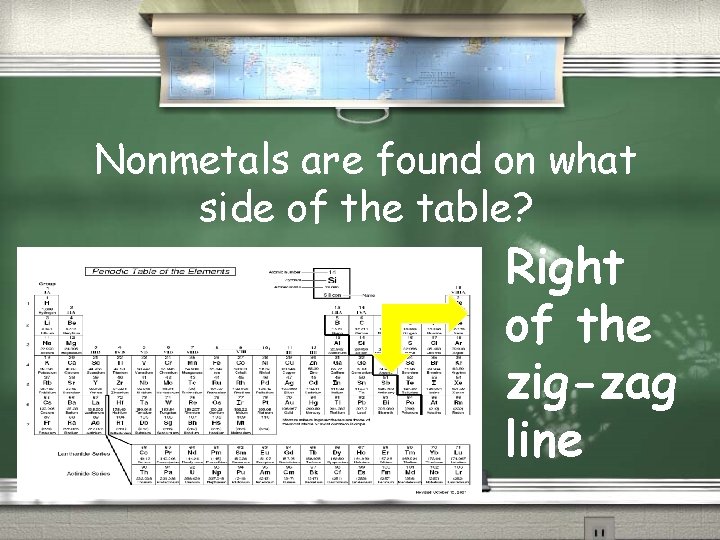
Nonmetals are found on what side of the table? Right of the zig-zag line
What group is more reactive: group 1 or group 18? Group 1

What is another word for ability to change shape? Malleable or ductile

What is another word for how shiny a metal is? Luster

Metals are good _____ of electricity and heat. Conductors

If nonmetals are not good conductors, then they are good ________? Insulators

More that ½ of the nonmetals at room temperature are in what state of matter? Gas
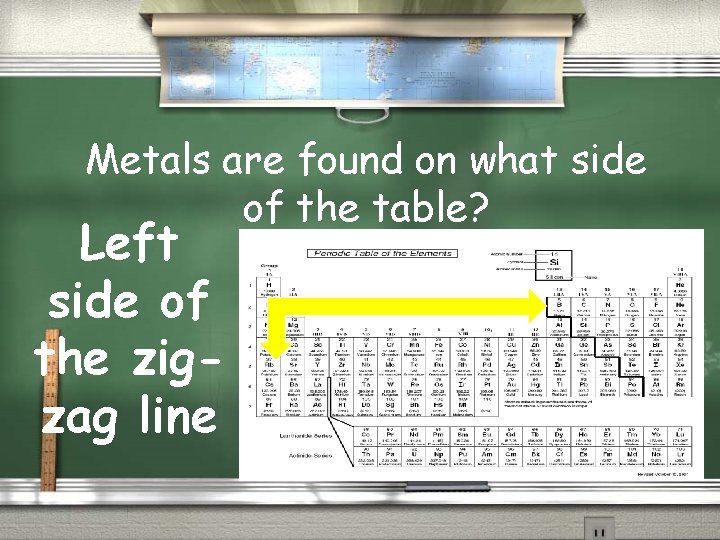
Metals are found on what side of the table? Left side of the zigzag line

If a metal reacts with a nonmetal, will a compound form? How do you know? Yes, the word reacts means the elements have combined to produce a new substance, this is a chemical change.

Name an element that has similar properties (chemical and physical) to Fluorine. Chlorine

What element has an atomic number of 15? Phosphorus

What element has an atomic mass of 47. 88? 22 Ti 47. 88 Titanium
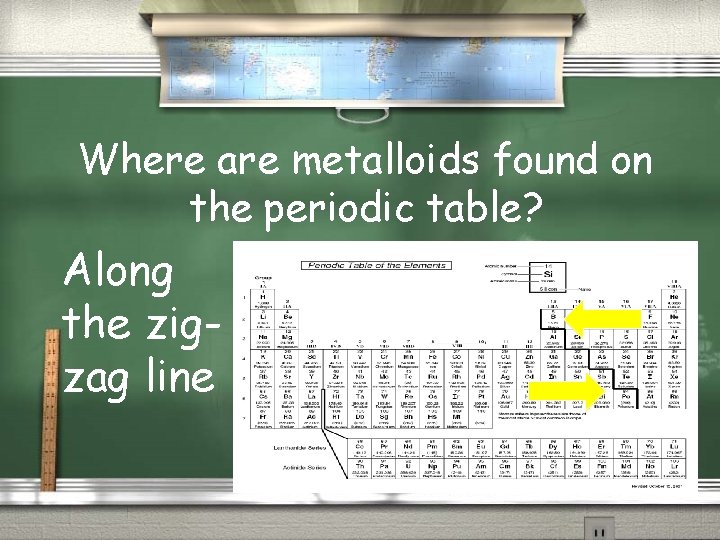
Where are metalloids found on the periodic table? Along the zigzag line

What element has 13 protons? 13 Al 26. 982 Aluminum
Is Sodium similar in properties to Magnesium? Why? No, because Sodium is a member of Group 1 and Magnesium is a member of Group 2.

The modern periodic table is arranged in order of increasing ________ Atomic Number

30 Zn Zinc 65. 39 How many neutrons are found in the element shown? 35

How many atoms are found in the compound, H₂SO₄? 7

How many elements are found in the compound, Na. OH 3
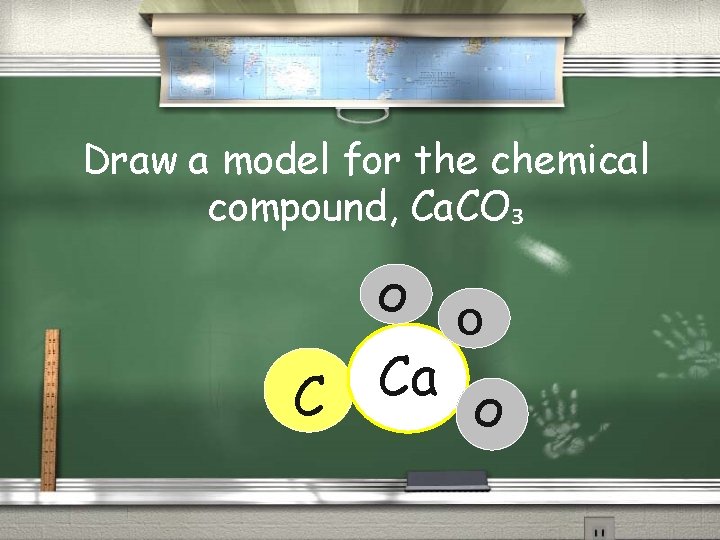
Draw a model for the chemical compound, Ca. CO₃ O Ca C O O

In the chemical equation: CO₂ + H₂O + sunlight energy C₆H₁₂O₆ + O₂ What are the reactants (in words)? Carbon dioxide and water

In the chemical equation: CO₂ + H₂O + sunlight energy C₆H₁₂O₆ + O₂ What are the products (in words)? glucose and oxygen

This element is soft, brittle, and a poor conductor of electricity. Which side of the periodic table would it be found on? Right side of the zig-zag line

If I am Nitrogen, who is most like me in my “family”? Phosphorus
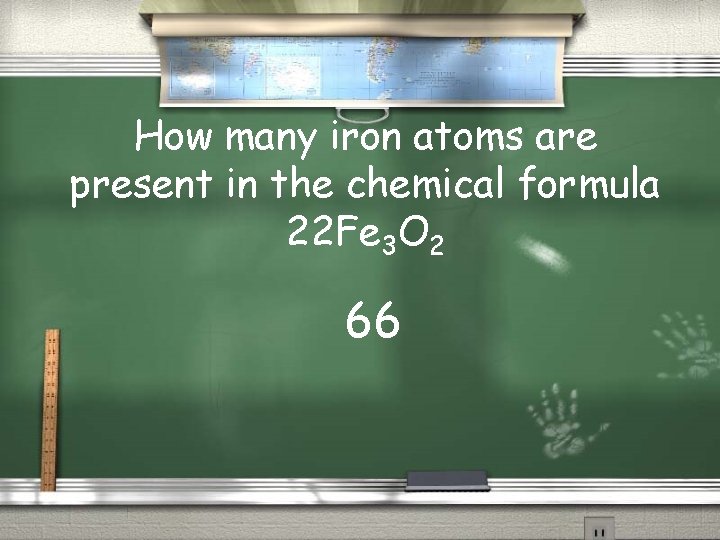
How many iron atoms are present in the chemical formula 22 Fe 3 O 2 66
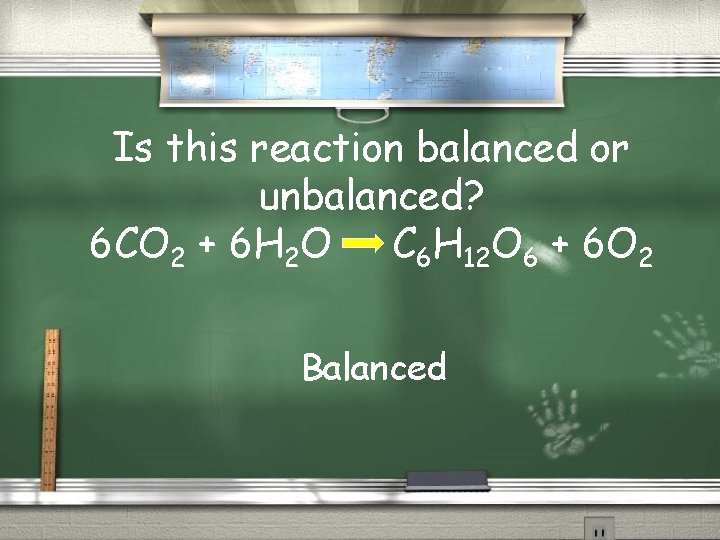
Is this reaction balanced or unbalanced? 6 CO 2 + 6 H 2 O C 6 H 12 O 6 + 6 O 2 Balanced

Define the Law of Conservation of Mass cannot be created or destroyed.
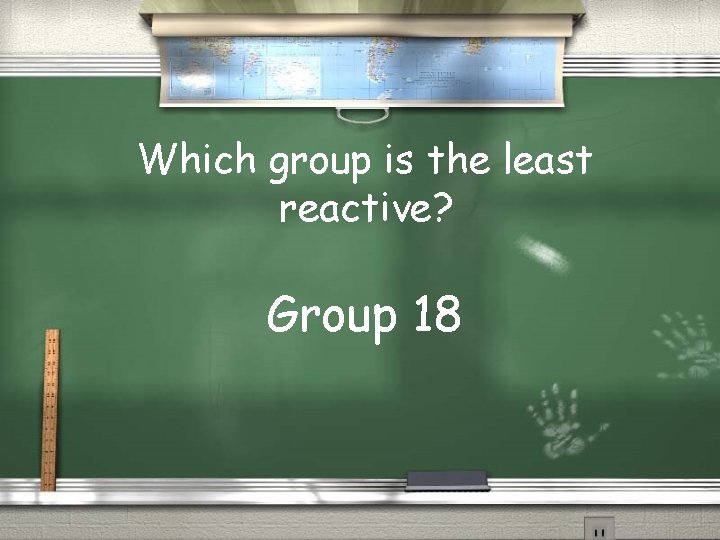
Which group is the least reactive? Group 18
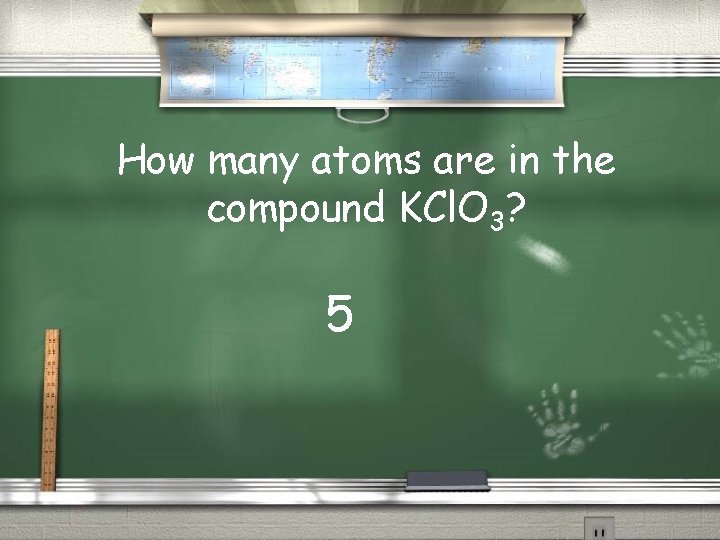
How many atoms are in the compound KCl. O 3? 5
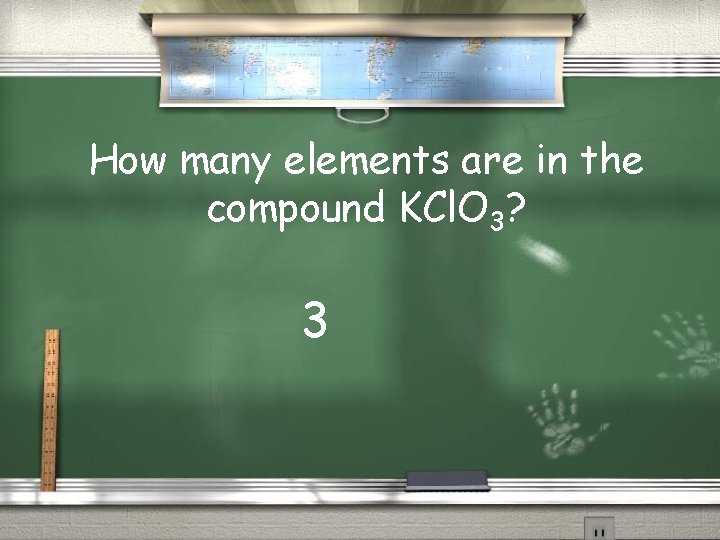
How many elements are in the compound KCl. O 3? 3

What do reactants always yield? Products

What are the products of photosynthesis? Glucose & Oxygen
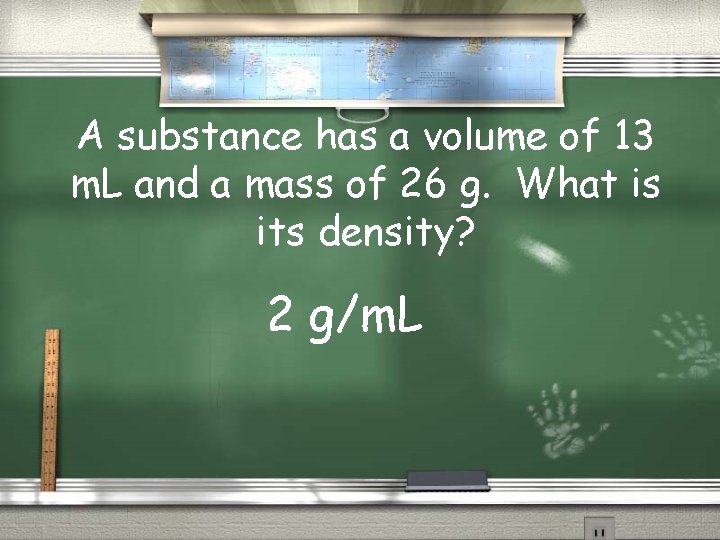
A substance has a volume of 13 m. L and a mass of 26 g. What is its density? 2 g/m. L

Why is group 18 the least reactive? Because it’s outer shell is full

What element is most similar to Ne? Flourine or Argon

What type of reaction has taken place when a precipitate is formed? Chemical reaction

What type of reaction occurs when a tooth rots? Chemical Reaction

Is the following reaction balanced or unbalanced? Si 2 H 3 + O 2 Si. O 2 + H 2 O Unbalanced
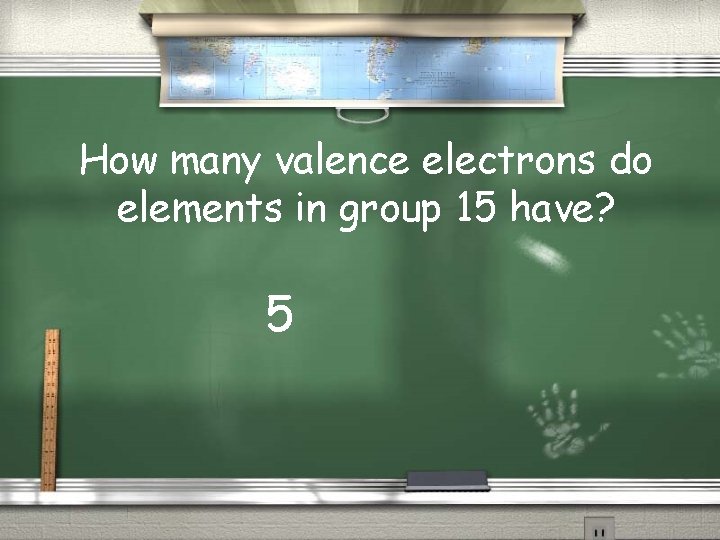
How many valence electrons do elements in group 15 have? 5

What elements are present in this chemical formula? 2 Na. OH Sodium, Oxygen, & Hydrogen

Hydrogen would easily react with elements in what group? 17
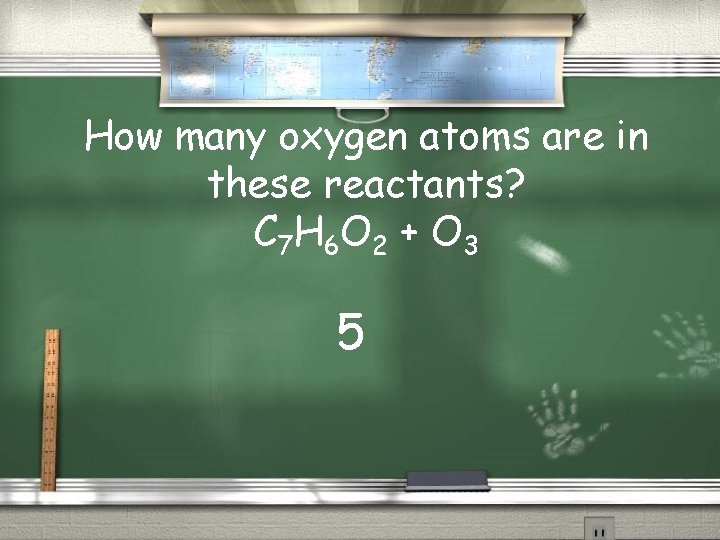
How many oxygen atoms are in these reactants? C 7 H 6 O 2 + O 3 5

What group do elements in group 2 like to react with? Group 16
- Slides: 58