Brain Death Organ Donor Management William Snyder RN

Brain Death & Organ Donor Management William Snyder, RN, BSN, CPTC Organ Procurement Coordinator University of Wisconsin Organ Procurement Organization 1 -866 -UWHC OPO (1 -866 -894 -2676)

Effective Donor Management • Stabilize the donor – Facilitate brain death exam or DCD Tool. • Manage the donor – To optimize the function and viability of all transplantable organs. “

Effective Donor Management • Requires clinical expertise, vigilance, flexibility, and the ability to address multiple complex clinical issues simultaneously and effectively. • Requires collaboration among the OPO, donor hospital critical care staff and consultants, and transplant program staff.

Effective Donor Management • Donor care is not usually assumed until after consent for donation has been obtained. • It is appropriate to collaborate prior to brain death, consent, etc, to prevent death and keep the option of organ donation open.

Effective Donor Management • Revision of existing orders or placement of new • • • medical orders is intended to: D/C medications no longer needed or appropriate (e. g. , anticonvulsants, mannitol, sedatives, antipyretics) Continue needed medications, or therapy (e. g. , vasoactive drug infusions, IV fluids and vent settings) Create “call orders” that inform bedside personnel of the goals for physiologic parameters and alert OPC of changes in donor status.

Diagnosing and Declaring Brain Death

Uniform Determination of Death Act An individual who has sustained either: (1) irreversible cessation of circulatory and respiratory function or (2) irreversible cessation of all functions of the entire brain, including the brain stem, is dead. A determination of death must be made in accordance with accepted medical standards. JAMA Nov 13, 1981 – Vol 246, No. 19

Diagnosis of Brain Death • Brain death is a clinical diagnosis. It can be made without confirmatory testing if you are able to establish the etiology, eliminate reversible causes of coma, complete fully the neurologic examination and apnea testing. • The diagnosis requires demonstration of the absence of both cortical and brain stem activity, and demonstration of the irreversibility of this state. R. Erff, D. O. , Walter Reed Army Medical Center

Etiology of Brain Death • Severe head trauma • Aneurismal subarachnoid hemorrhage • Cerebrovascular injury • Hypoxic-ischemic encephalopathy • Fulminant hepatic necrosis • Prolonged cardiac resuscitation or asphyxia • Tumors R. Erff, D. O. , Walter Reed Army Medical Center

Prerequisites to the Diagnosis Evidence of acute CNS catastrophe compatible with brain death: - Clinical or Neuroimaging Exclusion of reversible medical conditions that can confuse the clinical assessment: - Severe electrolyte, acid base and endocrine disturbance Absence of drug intoxication and poisoning Absence of sedation and neuromuscular blockade Hypotension (suppresses EEG activity and CBF) Absence of severe hypothermia (core temp < 35 C)

Brain Stem Reflexes • Cranial nerve examination: - No pupillary response to light. Pupils midline - and dilated 4 -6 mm. No oculocephalic reflex (Doll’s eyes) – contraindicated in C- spine injury. No oculovestibular reflex (tonic deviation of eyes toward cold stimulus) – contraindicated in ear trauma. Absence of corneal reflexes Absence of gag reflex and cough to tracheal suction.

Apnea Testing • Once coma and absence of brain stem reflexes • • • have been confirmed Apnea testing. Verifies loss of most rostral brain stem function Confirmed by Pa. CO 2 > 60 mm. Hg or Pa. CO 2 > 20 mm. Hg over baseline value. Testing can cause hypotension, severe cardiac arrhythmias and elevated ICP. Therefore, apnea testing is performed last in the clinical assessment of brain death. Consider confirmatory tests if apnea test inconclusive.

Apnea Testing • Following conditions must be met before apnea - test can be performed: Core temp > 35. 0 C Systolic blood pressure > 90 mm. Hg. Euvolemia Corrected diabetes insipitus Normal Pa. CO 2 ( Pa. CO 2 35 - 45 mm. Hg). Preoxygenation (Pa. O 2 > 200 mm. Hg).

Brain Death in Children • Clinical exam is same as in adults. • Testing criteria depends on age of child. - Neonate < 7 days Brain death testing is not valid. - 7 days – 2 months - Two clinical exams and two EEG 48 hrs apart. - 2 months – 1 year - Two clinical exams and two EEG 24 hrs apart. - or two clinical exams, EEG and blood flow study. - Age > 1 year to 18 years - Two clinical exams 12 hrs apart, confirmatory study - Optional

Confirmatory Testing • Purely optional when the clinical criteria are met • unambiguously. A confirmatory test is needed for patients in whom specific components of clinical testing cannot be reliably evaluated - Incomplete brain stem reflex testing Incomplete apnea testing Toxic drug levels Children younger than 1 year old. Required by institutional policy R. Erff, D. O. , Walter Reed Army Medical Center

Confirmatory Tests for Brain Death • Cerebral Blood Flow (CBF) Studies – Cerebral Angiography – Nuclear Flow Study • EEG (when brain scan is not utilized)
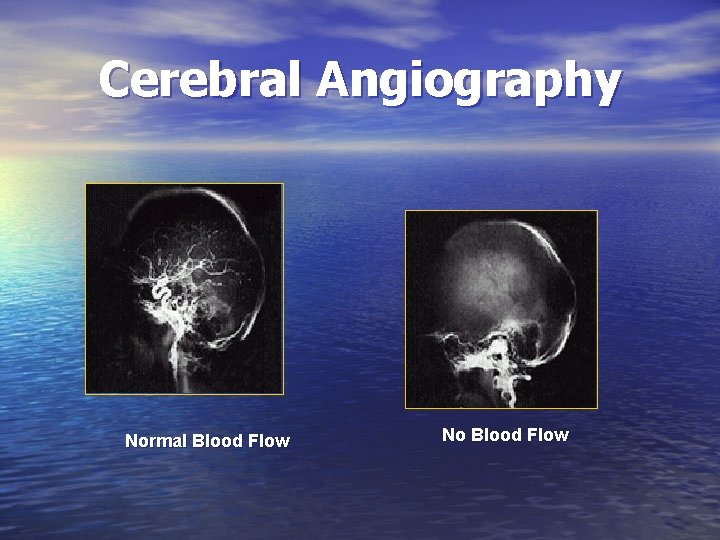
Cerebral Angiography Normal Blood Flow No Blood Flow
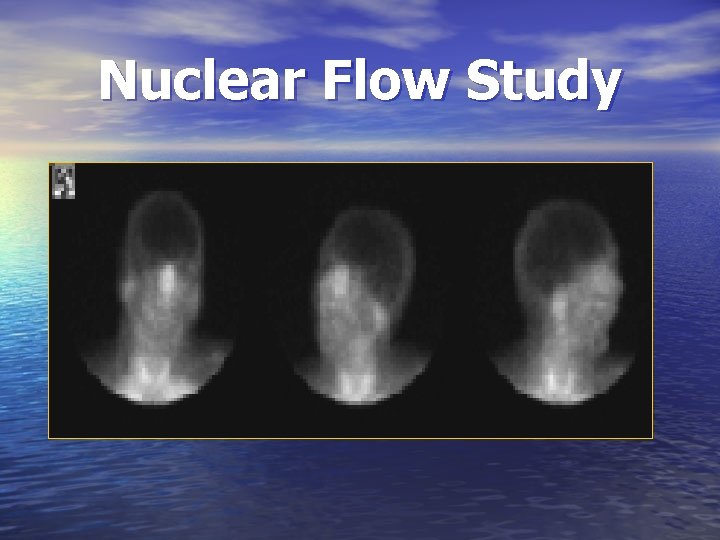
Nuclear Flow Study

Elements of brain death declaration • Date • Time • Detailed documentation of Clinical Exam including specifics of Apnea Testing • Physician signature

What to expect after brain death

Pathophysiology • Loss of brain stem function results in systemic - physiologic instability: Loss of vasomotor control leads to a hyperdynamic state. Cardiac arrhythmias Loss of respiratory function Loss of temperature regulation Hypothermia Hormonal imbalance DI, hypothyroidism

Perioperative Management • Following the diagnosis of Brain Death ü Therapy shifts in emphasis from cerebral resuscitation to optimizing organ fxn for subsequent transplantation. ü The normal sequelae of brain death results in cardiovascular instability & poor organ perfusion. ü Medical staff must focus on: Providing hemodynamic stabilization. Support of body homeostasis. Maintenance of adequate cellular oxygenation and donor organ perfusion. ü Without appropriate intervention brain death is followed by severe injury to most other organ systems. Circulatory collapse will usually occur within 48 hrs.

Autonomic/Sympathetic Storm • Release of catecholamines from adrenals (Epinephrine and Norepinephrine) results in a hyperdynamic state: – – Tachycardia Elevated C. O. Vasoconstriction Hypertension

Failure of the Hypothalamus results in: • Impaired temperature regulation hypothermia or hyperthermia • Leads to vasodilation without the ability to vasoconstrict or shiver (loss of vasomotor tone) • Leads to problems with the pituitary. . .

Normal Pituitary Gland • Controlled by the hypothalamus • Releases ADH to conserve water • Stimulates the release of thyroid hormone

Pituitary Failure results in: • ADH ceases to be produced = Diabetes Insipidus • Can lead to hypovolemia and electrolyte imbalances • Leads to problems with the thyroid gland
Normal Thyroid Gland • Produces hormones that increase the metabolic rate and sensitivity of the cardiovascular system ü Levothyroxine (T 4) ü Triiodothyronine (T 3)
Thyroid Failure Leads to: ü Cardiac instability ü Labile blood pressure ü Potential coagulation problems
Cardiovascular System Intensive care management • “Rules of 100’s” - Maintain SBP > 100 mm. HG HR < 100 BPM UOP > 100 ml/hr Pa. O 2 > 100 mm. Hg • Aggressive fluid resuscitative therapy directed at restoring and maintaining intravascular volume. SBP > 90 mm. Hg (MAP > 60 mm. Hg) or CVP ~ 10 mm. Hg.
Neurogenic Pulmonary Edema ü Brain death is associated with numerous pulmonary problems ü The lungs are highly susceptible to injury resulting from the rapid changes that occur during the catecholamine storm ü Left-sided heart pressures exceed pulmonary pressure, temporarily halting pulmonary blood flow ü The exposed lung tissue is severely injured, resulting in interstitial edema and alveolar hemorrhage, a state commonly referred to as neurogenic pulmonary edema

Release of Plasminogen Activator DIC ü Results from the passage of necrotic brain tissue into the circulation ü Leads to coagulopathy and sometimes progresses further to DIC ü DIC may persist despite factor replacement requiring early organ recovery (Also affected by hypothermia, release of catecholamines & hemodilution as a result of fluid resuscitation)

Organ Donor Management (in a nutshell) • Hypertension Hypotension • Excessive Urinary Output • Impaired Gas Exchange • Electrolyte Imbalances • Hypothermia
Hypotension Management üFluid Bolus – NS or LR (Followed by MIVF NS or. 45 NS) üConsider colloids üDopamine üNeosynephrine üVasopressin üThyroxine (T 4 protocol) (seriously)

T 4 Protocol Background • Brain death leads to sudden reduction in circulating pituitary hormones • May be responsible for impairment in myocardial cell metabolism and contractility which leads to myocardial dysfunction • Severe dysfunction may lead to extreme hypotension and loss of organs for transplant
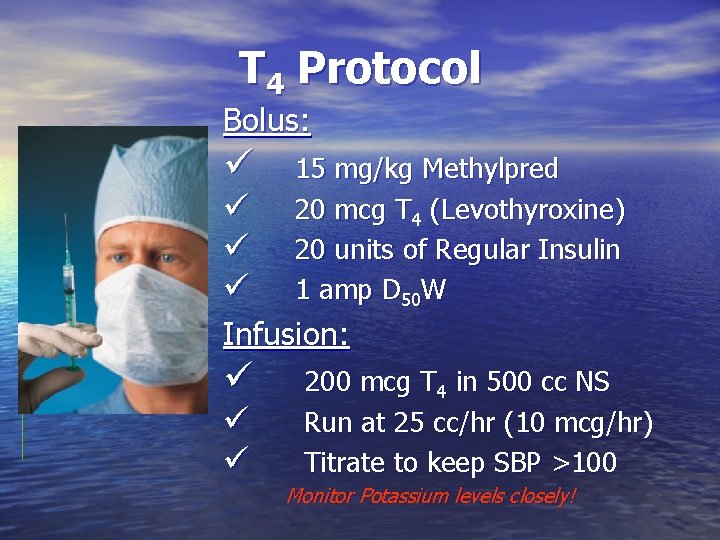
T 4 Protocol Bolus: ü ü 15 mg/kg Methylpred 20 mcg T 4 (Levothyroxine) 20 units of Regular Insulin 1 amp D 50 W Infusion: ü ü ü 200 mcg T 4 in 500 cc NS Run at 25 cc/hr (10 mcg/hr) Titrate to keep SBP >100 Monitor Potassium levels closely!

Vasopressin (AVP, Pitressin) • Low dose shown to reduce inotrope use • Plays a critical role in restoring vasomotor tone Vasopressin Protocol ü 4 unit bolus ü 1 - 4 u/hour – titrate to keep SBP >100 or MAP >60

Diabetes Insipidus Management ü Treatment is aimed at correcting hypovolemia ü Desmopressin (DDAVP) 1 mcg IV, may repeat x 1 after 1 hour. ü Replace hourly U. O. on a volume per volume basis with MIVF to avoid volume depletion ü Leads to electrolyte depletion/instability monitor closely to avoid hypernatremia and hypokalemia

Diabetes Insipidus • Goal is UOP 1 -3 • • ml/kg/hr Rule of thumb – 500 ml UOP per hour x 2 hours is DI Severe cases – Notify OPC. Assess clinical situation.

Impaired Gas Exchange Management • Maintain Pa. O 2 of >100 and a • • saturation >95% Monitor ABG’s q 2 h or as requested by OPO PEEP 5 cm, HOB up 30 o Increase ET cuff pressure immediately after BD declaration Aggressive pulmonary toilet (Keep suctioning & turning q 2 h) CXR (Radiologist to provide measurements & interpretation) OPO may request bronchoscopy CT of chest requested in some cases

Correct Impaired Gas Exchange and Maximize Oxygenation! Most organ donors are referred with: üChest trauma üAspiration üLong Hospitalization with bed rest resulting in atelectasis or pneumonia üImpending Neurogenic Pulmonary Edema Brain Death contributes to and complicates all of these conditions

Impaired Gas Exchange Goals… • Goals are to maintain health of lungs for transplant while optimizing oxygen delivery to other transplantable organs • Avoid over-hydration • Ventilatory strategies aimed to protect the lung • Avoid oxygen toxicity by limiting Fi 02 to achieve a Pa 02 100 mm. Hg & PIP < 30 mm. Hg.

Electrolyte Imbalance Management Hypokalemia If K+ < 3. 4 – Add KCL to MIVF (anticipate low K+ with DI & T 4 administration) Hypernatremia If NA+ >155 – Change MIVF to include more free H 20, Free H 20 boluses down NG tube (this is often the result of dehydration, NA + administration, and free H 20 loss 2 o to diuretics or DI) Calcium, Magnesium, and Phosphorus Deficiencies here common…often related to polyuria associated with osmotic diuresis, diuretics & DI.

Hypothermia Management • Monitor temperature • • • continuously NO tympanic, axillary or oral temperatures. Central only. Place patient on hypothermia blanket to maintain normal body temperature In severe cases (<95 degrees F) consider: – – warming lights covering patient’s head with blankets – hot packs in the axilla – warmed IV fluids – warm inspired gas

Anemia ü Hematocrit < 30% must be treated ü Transfuse 2 units PRBC’s immediately ü Reassess 1 o after completion of 2 nd unit and repeat infusion of 2 units if HCT remains below 30% ü Assess for source of blood loss and treat accordingly

Incidence of pathophysiologic changes following Brain Death: - Hypotension Diabetes Insipidus DIC Cardiac arrhythmias Pulmonary edema Metabolic acidosis 81% 65% 28% 25% 18% 11% Physiologic changes During Brain Stem Death – Lessons for Management of the Organ Donor. The Journal of Heart & Lung Transplantation Sept 2004 (suppl)
Organ Donation Process • Evaluate organ function • Labs (& UA) within 6 • • hours of surgery Type and Screen Consent signed Serology testing Medical Social History Locate potential recipients Manage hemodynamics Arrange operating room

The Teams. . . Your Hospital - Anesthesia - Primary Care Physician or Intensivist (For DCD) - Surgical Technician/Scrub Nurse - Circulating Nurse Abdominal Transplant Team - Surgeon - Physician Assistant - Surgical Recovery Coordinator Cardiothoracic Team - Surgeon - Surgical Fellow - Surgical Recovery Coordinator
Organ Preservation Time • • • Heart: 4 -6 hours Lungs: 4 -6 hours Liver: 12 hours Pancreas: 12 -18 hours Kidneys: 72 hours Small Intestines: 4 -6 hours

- Slides: 49