BPCO e carenza ereditaria di AAT Ilaria Ferrarotti

BPCO e carenza ereditaria di AAT Ilaria Ferrarotti Centro per la Diagnosi del Deficit di Alfa 1 -Antitripsina Fondazione IRCCS Policlinico S. Matteo Dipartimento di Medicina Molecolare Università di Pavia

Alpha-1 antitrypsin Deficiency ØCommon genetic disease among Caucasians (1/5000) ØAutosomal co-dominant heredity ØReduced or complete lack of alpha-1 -antitrypsin secretion from hepatocytes into the bloodstream ØSevere AAT deficiency is most often associated with homozygous Zmutation (Pi. ZZ); over 120 genetic variants of the Pi gene are currently known ØLung, liver, and skin (very rare) can be affected ØOften remains unrecognized or is wrongly diagnosed as COPD
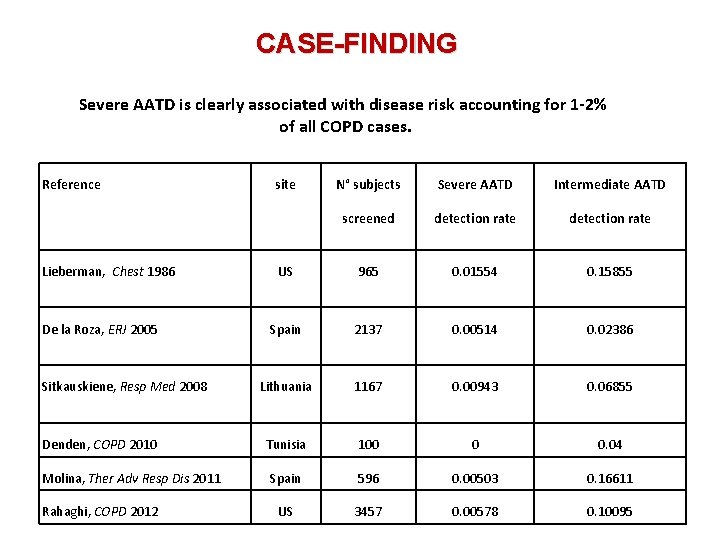
CASE-FINDING Severe AATD is clearly associated with disease risk accounting for 1 -2% of all COPD cases. Reference Lieberman, Chest 1986 De la Roza, ERJ 2005 Sitkauskiene, Resp Med 2008 Denden, COPD 2010 Molina, Ther Adv Resp Dis 2011 Rahaghi, COPD 2012 site N° subjects Severe AATD Intermediate AATD screened detection rate US 965 0. 01554 0. 15855 Spain 2137 0. 00514 0. 02386 Lithuania 1167 0. 00943 0. 06855 Tunisia 100 0 0. 04 Spain 596 0. 00503 0. 16611 US 3457 0. 00578 0. 10095
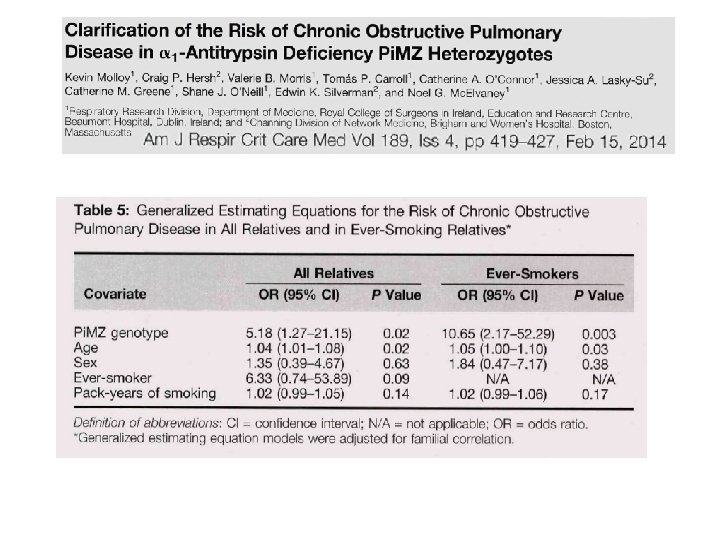

Nevertheless. . . . large genome-wide association studies (GWAS) on these pulmonary outcomes have so far failed to point to this chromosomal region.
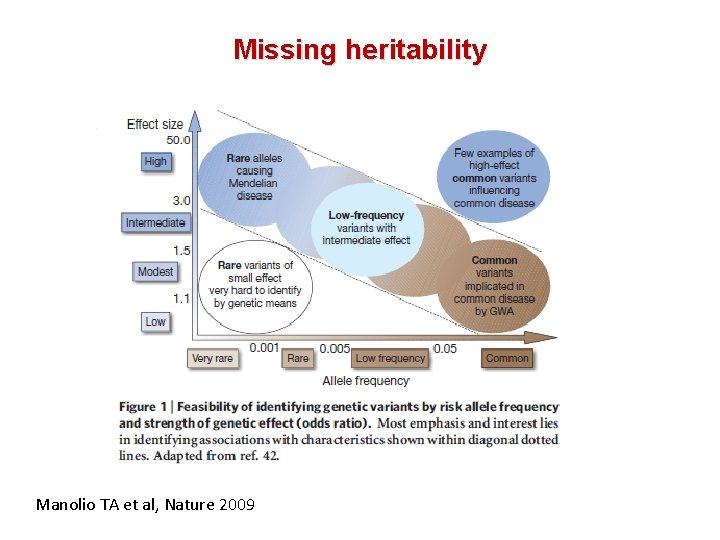
Missing heritability Manolio TA et al, Nature 2009
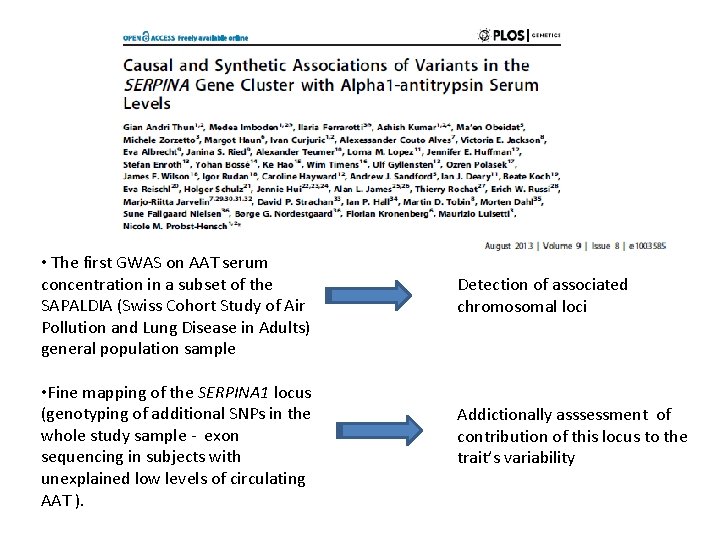
• The first GWAS on AAT serum concentration in a subset of the SAPALDIA (Swiss Cohort Study of Air Pollution and Lung Disease in Adults) general population sample • Fine mapping of the SERPINA 1 locus (genotyping of additional SNPs in the whole study sample - exon sequencing in subjects with unexplained low levels of circulating AAT ). Detection of associated chromosomal loci Addictionally asssessment of contribution of this locus to the trait’s variability
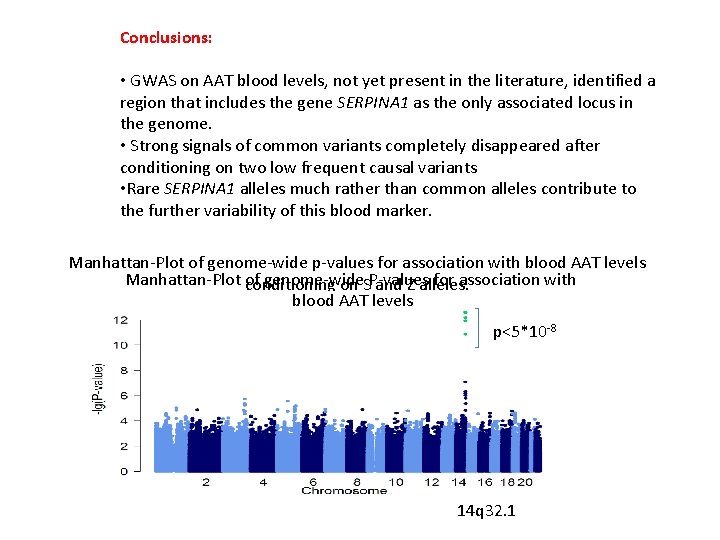
Conclusions: • GWAS on AAT blood levels, not yet present in the literature, identified a region that includes the gene SERPINA 1 as the only associated locus in the genome. • Strong signals of common variants completely disappeared after conditioning on two low frequent causal variants • Rare SERPINA 1 alleles much rather than common alleles contribute to the further variability of this blood marker. Manhattan-Plot of genome-wide p-values for association with blood AAT levels Manhattan-Plot conditioning of genome-wide for association with on SP-values and Z alleles. blood AAT levels p<5*10 -8 14 q 32. 1

Inference: • this locus is suggestive to contribute to the missing heritability of pulmonary outcomes. The absence of this locus among the statistically significantly associated signals in corresponding GWAS may reflect 1) small effect sizes of the common variants in combination with insufficient sample size and stringent correction for multiple testing leading to a high risk of type II errors 2) poor tagging of the underlying causal variants by nearby SNPs in the chip arrays would further reduce the chance to find this locus among the significantly associated results.
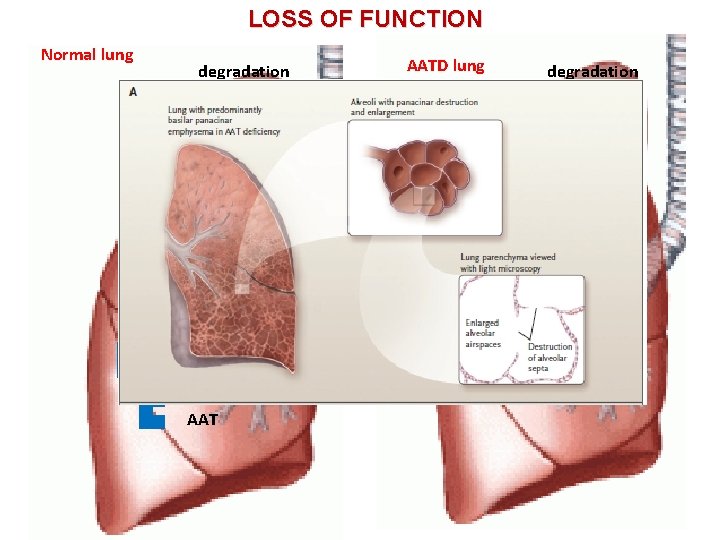
LOSS OF FUNCTION Normal lung degradation AATD lung degradation HNE AAT
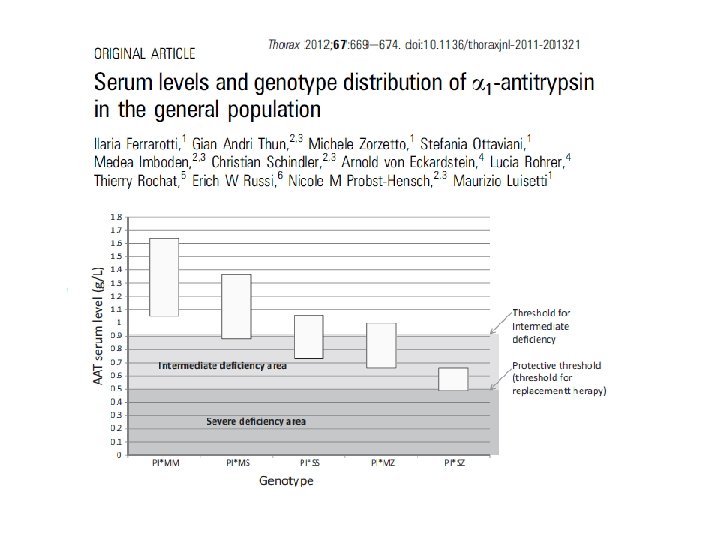
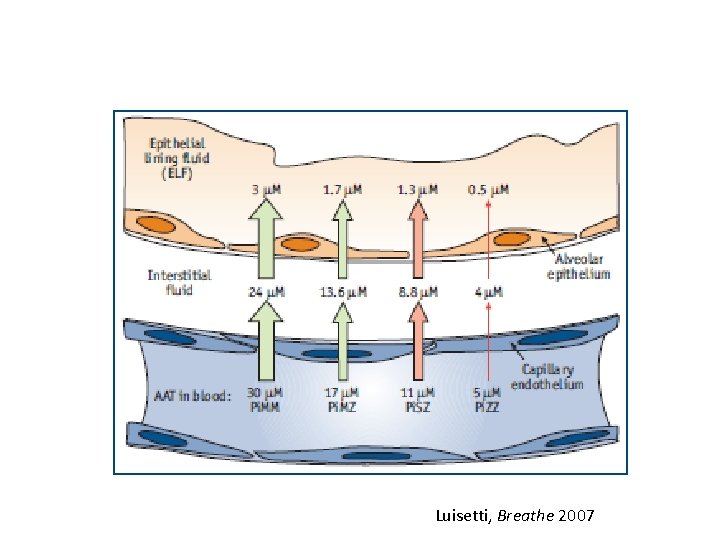
Luisetti, Breathe 2007
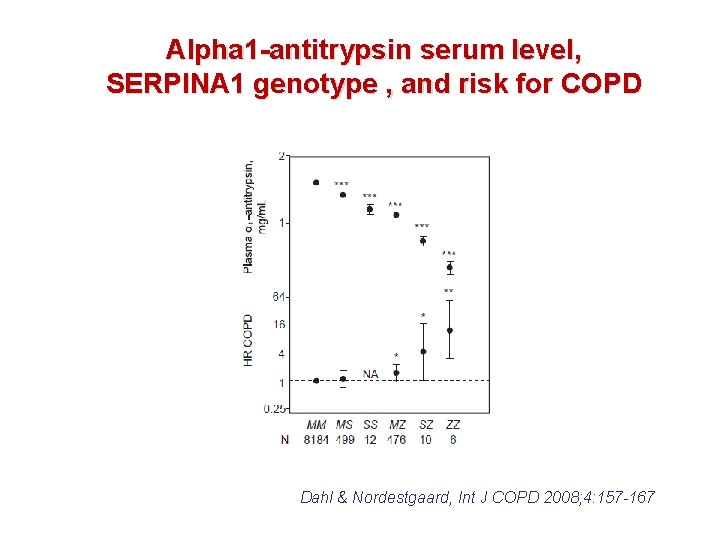
Alpha 1 -antitrypsin serum level, SERPINA 1 genotype , and risk for COPD Dahl & Nordestgaard, Int J COPD 2008; 4: 157 -167
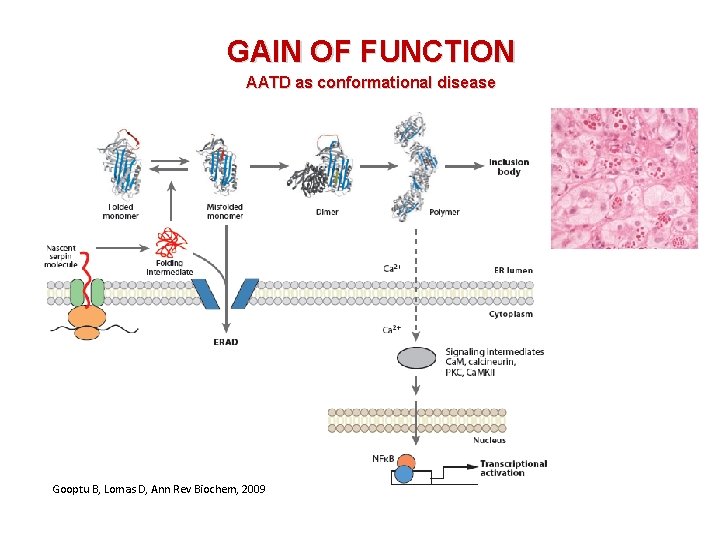
GAIN OF FUNCTION AATD as conformational disease Gooptu B, Lomas D, Ann Rev Biochem, 2009
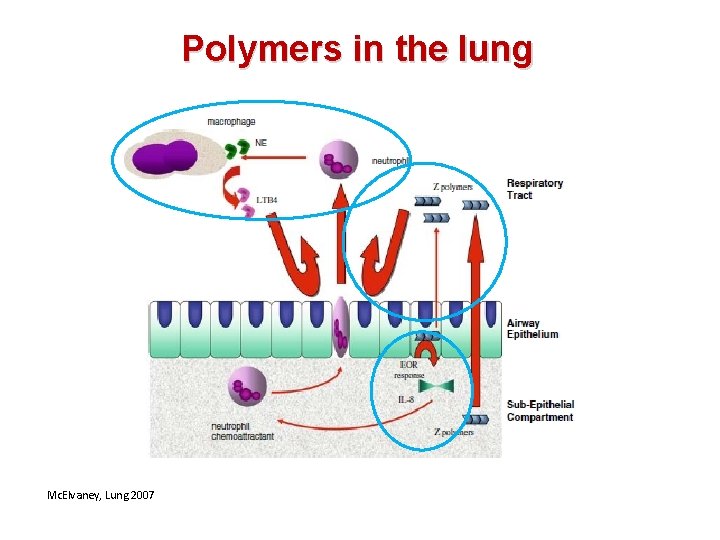
Polymers in the lung Mc. Elvaney, Lung 2007
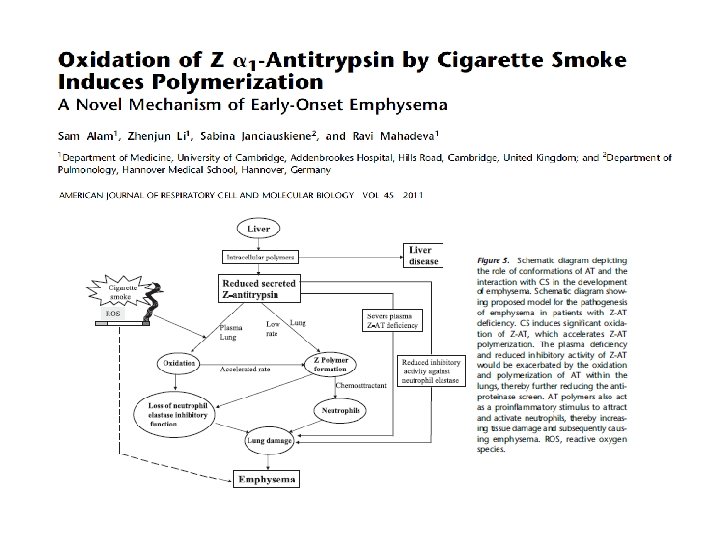
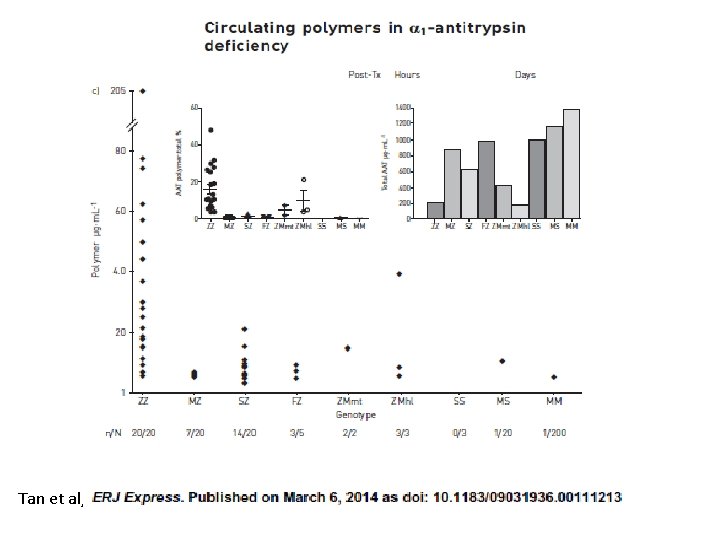
Tan et al,

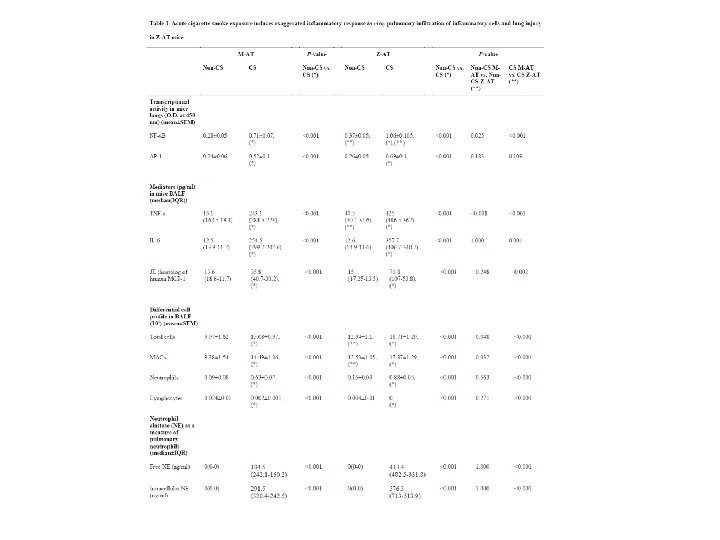
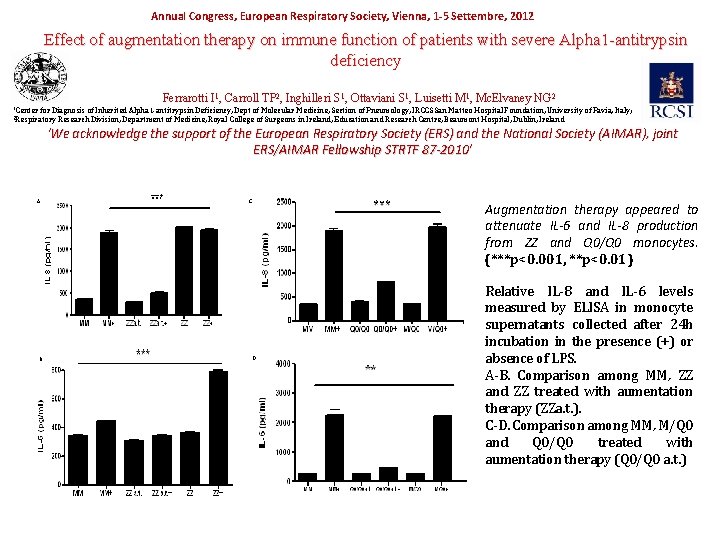
Annual Congress, European Respiratory Society, Vienna, 1 -5 Settembre, 2012 Effect of augmentation therapy on immune function of patients with severe Alpha 1 -antitrypsin deficiency Ferrarotti I 1, Carroll TP 2, Inghilleri S 1, Ottaviani S 1, Luisetti M 1, Mc. Elvaney NG 2 1 Center for Diagnosis of Inherited Alpha 1 -antitrypsin Deficiency, Dept of Molecular Medicine, Section of Pneumology, IRCCS San Matteo Hospital Foundation, University of Pavia, Italy; Research Division, Department of Medicine, Royal College of Surgeons in Ireland, Education and Research Centre, Beaumont Hospital, Dublin, Ireland. 2 Respiratory ‘We acknowledge the support of the European Respiratory Society (ERS) and the National Society (AIMAR), joint ERS/AIMAR Fellowship STRTF 87 -2010’ A B C D Augmentation therapy appeared to attenuate IL-6 and IL-8 production from ZZ and Q 0/Q 0 monocytes. (***p<0. 001, **p<0. 01) Relative IL-8 and IL-6 levels measured by ELISA in monocyte supernatants collected after 24 h incubation in the presence (+) or absence of LPS. A-B. Comparison among MM, ZZ and ZZ treated with aumentation therapy (ZZa. t. ). C-D. Comparison among MM, M/Q 0 and Q 0/Q 0 treated with aumentation therapy (Q 0/Q 0 a. t. )
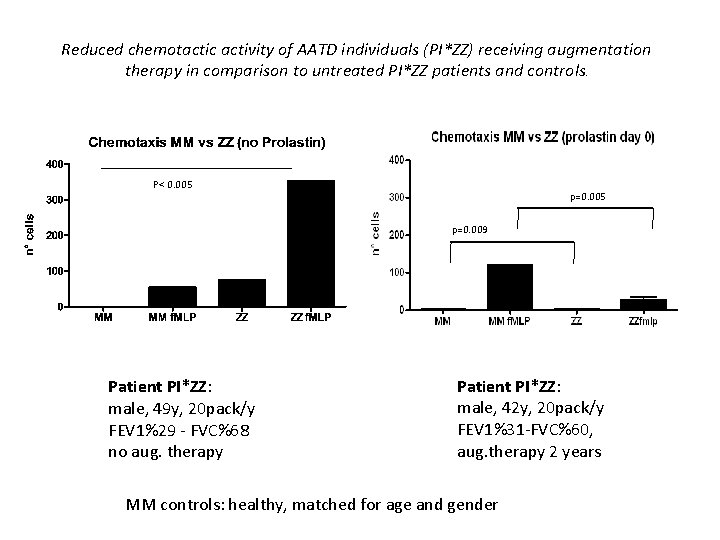
Reduced chemotactic activity of AATD individuals (PI*ZZ) receiving augmentation therapy in comparison to untreated PI*ZZ patients and controls. P< 0. 005 p=0. 009 Patient PI*ZZ: male, 49 y, 20 pack/y FEV 1%29 - FVC%68 no aug. therapy Patient PI*ZZ: male, 42 y, 20 pack/y FEV 1%31 -FVC%60, aug. therapy 2 years MM controls: healthy, matched for age and gender
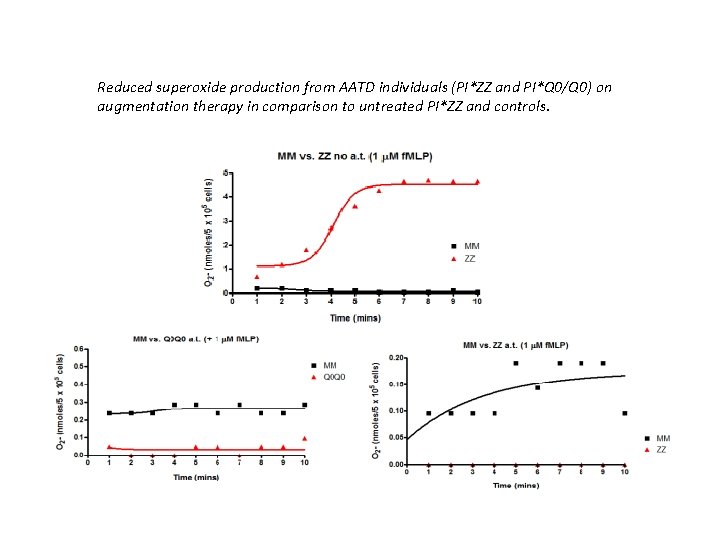
Reduced superoxide production from AATD individuals (PI*ZZ and PI*Q 0/Q 0) on augmentation therapy in comparison to untreated PI*ZZ and controls.
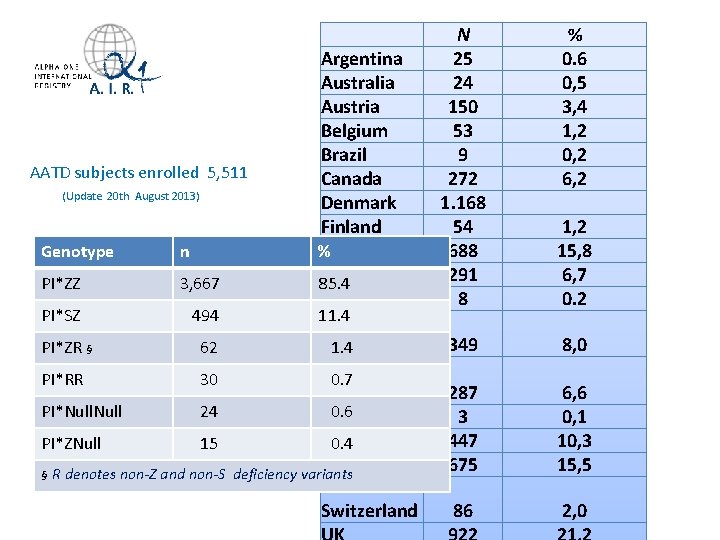
N 25 24 150 53 9 272 1. 168 54 688 291 8 1, 2 15, 8 6, 7 0. 2 Netherlands 1. 4 New PI*RR 30 0. 7 Zealand PI*Null 24 0. 6 Poland Spain PI*ZNull 15 0. 4 Sweden § R denotes non-Z and non-S deficiency variants 349 8, 0 287 3 447 675 6, 6 0, 1 10, 3 15, 5 Switzerland 86 2, 0 AATD subjects enrolled 5, 511 (Update 20 th August 2013) Genotype n PI*ZZ 3, 667 PI*SZ 494 PI*ZR § Argentina Australia Austria Belgium Brazil Canada Denmark Finland % Germany Italy 85. 4 Israel 11. 4 62 % 0. 6 0, 5 3, 4 1, 2 0, 2 6, 2
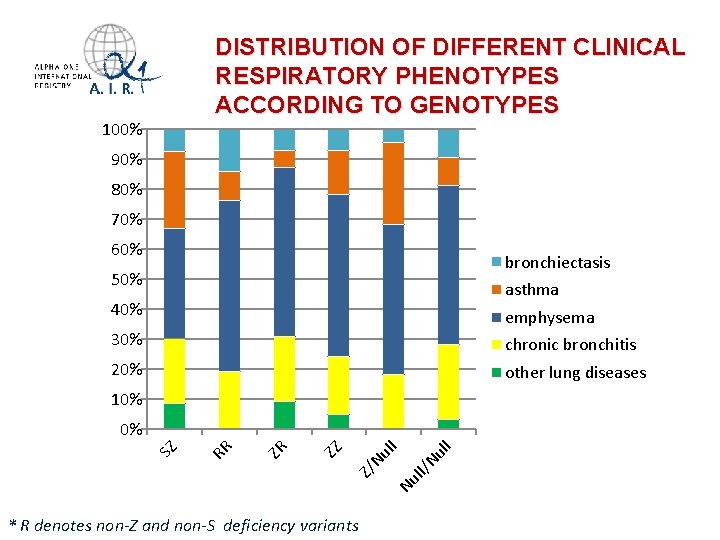
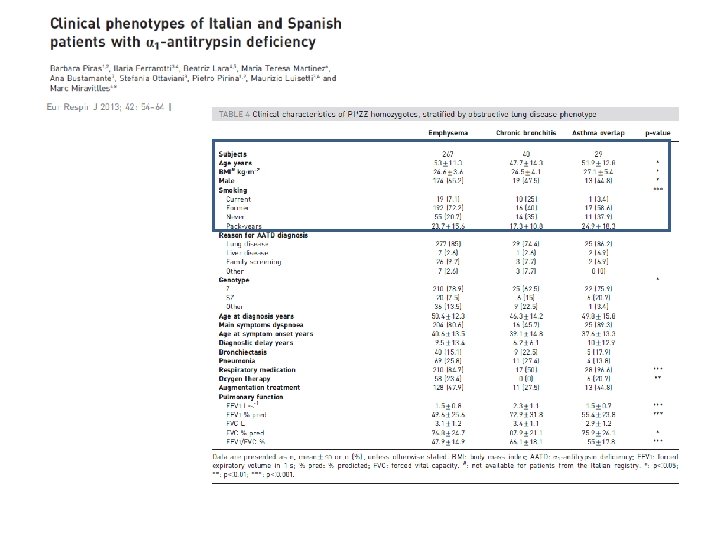
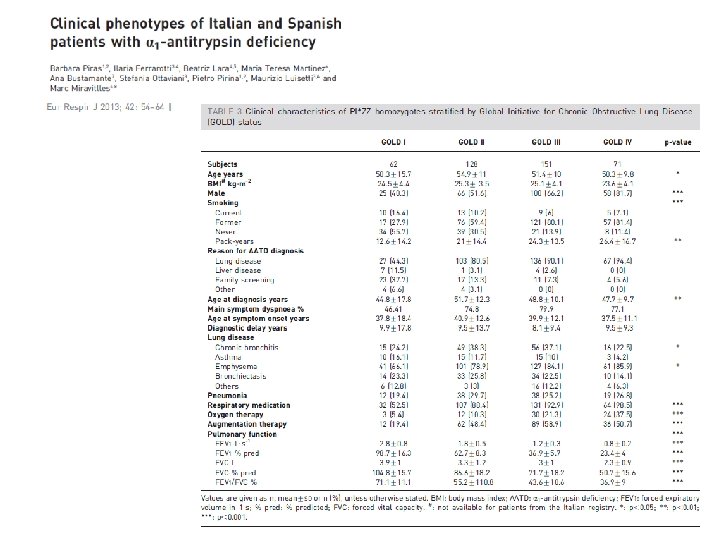
DISTRIBUTION OF DIFFERENT CLINICAL RESPIRATORY PHENOTYPES ACCORDING TO GENOTYPES 100% 90% 80% 70% 60% bronchiectasis 50% asthma 40% emphysema 30% chronic bronchitis 20% other lung diseases 10% * R denotes non-Z and non-S deficiency variants ll Nu ll/ Nu Z/ Nu l l ZZ ZR RR SZ 0%
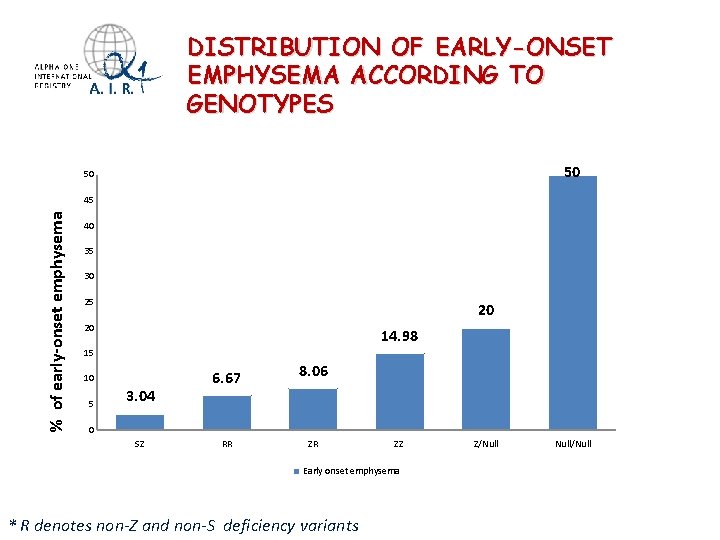
DISTRIBUTION OF EARLY-ONSET EMPHYSEMA ACCORDING TO GENOTYPES 50 50 % of early-onset emphysema 45 40 35 30 25 20 20 14. 98 15 10 5 3. 04 6. 67 8. 06 0 SZ RR ZR ZZ Early onset emphysema * R denotes non-Z and non-S deficiency variants Z/Null/Null
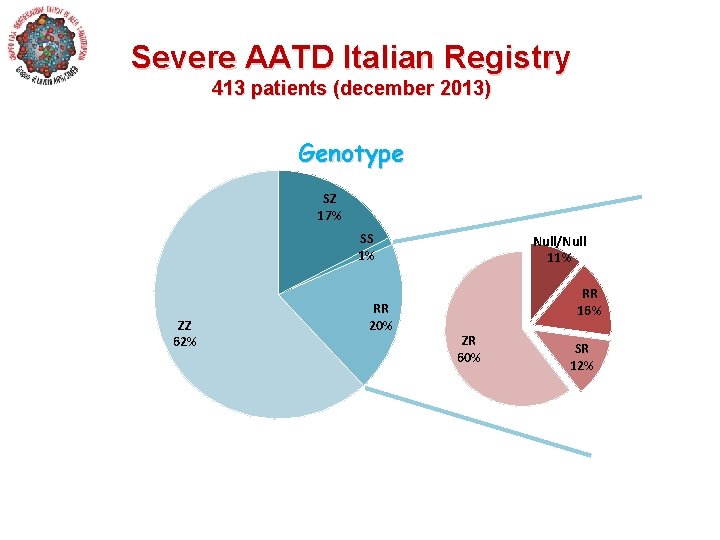
Severe AATD Italian Registry 413 patients (december 2013) Genotype SZ 17% SS 1% ZZ 62% RR 20% Null/Null 11% RR 16% ZR 60% SR 12%

IRCCS San Matteo Hospital Foundation, University of Pavia: Pulmonology Unit Head of Department: Maurizio Luisetti Laboratory of Biochemistry and Genetics of Lung Disease Stefania Ottaviani Marina Gorrini Michele Zorzetto Ilaria Campo Simona Inghilleri Simona Ferrari Unit of Pulmonology Francesca Mariani Zamir Kadija Elena Paracchini
- Slides: 29