Boyles Law There is an inverse relationship between

Boyle’s Law • There is an inverse relationship between the volume and pressure of a gas (if one increases the other decreases) • If temperature is constant: • PV = k where k is a constant • P 1 V 1=P 2 V 2 KMT – decreasing the volume of a container will cause an increase in the number of collisions; thus an increase in pressure

As the pressure of a gas at 760 torr is changed to 380 torr at constant temperature, the volume of the gas A. ) decrease B. ) increase C. ) remain the same Decrease Pressure, Increase Volume

Example • 5. 00 L of a gas is at 1. 08 atm. What pressure is obtained when the volume is 10. 0 L? P 1 = V 1 = P 2 = V 2 = 1. 08 atm 5. 00 L ? 10. 0 L P 1 V 1 = P 2 V 2 (1. 08 atm)(5. 00 L) = P(10. 0 L) P = 0. 54 atm

Example • A sample of oxygen takes up 34 liters of space at standard pressure. When the pressure is changed to 340 k. Pa, find the new volume. P 1 = 101. 3 k. Pa V 1 = 34 L P 2 = 340 k. Pa V 2 = ? P 1 V 1 = P 2 V 2 (101. 3 k. Pa)(34 L) = (340 k. Pa)V V = 10. 13 L

A gas sample has a volume of 25. 0 millilters at a pressure of 1. 00 atmosphere. If the volume increases to 50. 0 milliliters and the temperature remains constant, the new pressure will be A. ) 1. 00 atm B. ) 2. 00 atm C. ) 0. 250 atm D. ) 0. 500 atm PV = PV (1. 00 atm)(25. 0 m. L) = P(50. 0 m. L)

Do This Now! • You are now wearing scuba gear and swimming under water at a depth of 66. 0 ft. You are breathing air at 3. 00 atm and your lung volume is 10. 0 L. Your scuba gauge indicates that your air supply is low so, to conserve air, you make a terrible and fatal mistake: you hold your breath while you surface. What happens to your lungs? Why? PV = PV The pressure at the surface is standard (1 atm) (3. 00 atm)(10. 0 L) = (1 atm)V V = 30 L Your lungs will explode
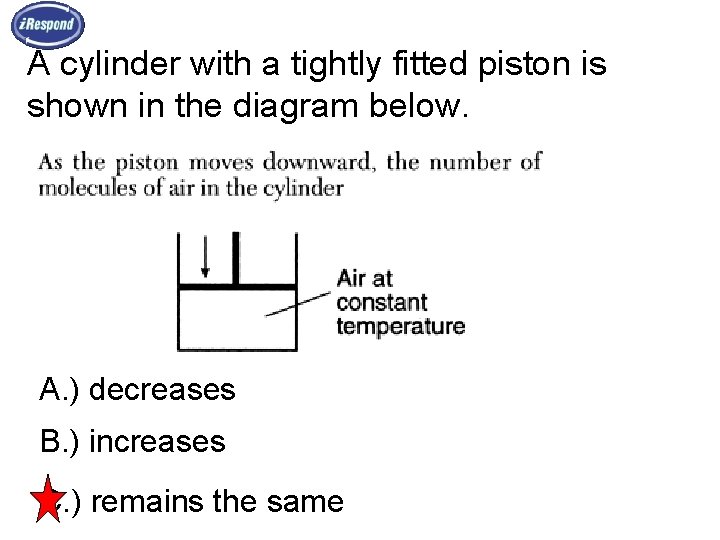
A cylinder with a tightly fitted piston is shown in the diagram below. A. ) decreases B. ) increases C. ) remains the same

A sample of gas is held at constant pressure. Increasing the kelvin temperature of this gas sample causes the average kinetic energy of its molecules to A. ) decrease and the volume of the gas sample to decrease B. ) decrease and the volume of the gas sample to increase C. ) increase and the volume of the gas sample to decrease D. ) increase and the volume of the gas sample to increase
- Slides: 8