Boyles Law The relationship between pressure and volume

Boyle’s Law The relationship between pressure and volume • Robert Boyle, an English scientist, first measured gases quantitatively in 1662. • He found that the value of PV remained the same if the temperature was held constant. • Boyle’s Law states PV = constant • As the pressure increases, the volume decreases • P 1 V 1 = P 2 V 2

Try this problem using Boyle’s Law Helium gas at constant temperature is contained in a 0. 80 L cylinder at a pressure of 1. 5 atm. If the pressure is increased to 2. 1 atm, what is the new volume?


Basic Boyle’s Law Problems 1) The pressure on 3. 0 L of CO at 0 o. C and 800 torr is reduced to 500 torr. What is the new volume? 2) 9. 5 L of NO 2 and 30 mm. Hg is moved to a 7. 5 L container. What is the new pressure? 3) 10. 75 L of CO 2 has its pressure increased to 2. 0 atm. If the starting conditions were, 92 o. C, 0. 80 atm, what is the volume of this gas?

Boyle’s Law Examples A 6. 0 liter container of nitrogen gas at 742 mm Hg has what volume when the pressure changes to 879 mm Hg?
What is the volume of 30. 0 grams of sulfur dioxide gas at 0 o. C and 400 torr?
What is the volume of 82 grams of acetylene gas (C 2 H 2) at 0 OC and 1. 9 atm pressure?

73. 9 L of benzene, C 6 H 6, at 0 o. C and 3. 25 atm, has a mass of how many grams?

52 liter of methane(CH 4) at 1800 mm Hg and 0 OC contains how many grams of methane?

One Element in the Molecule 12. 6 L of ammonia gas at 0 o. C and 2. 0 atm will contain how many grams of the element hydrogen?

100 L of sulfur pentoxide gas at 0 o. C and 430 mm Hg will contain how many grams of the element oxygen?
330 grams of sulfur dioxide at 0 OC and a pressure of 550 torr contains how many liters of oxygen gas?

Example with a Chemical Reaction Oxygen gas is formed from the decomposition of mercury(II) oxide. If 145 liters of oxygen gas at a pressure of 2 atm are formed, how many grams of mercury(II) oxide are required?

Review for Boyle’s Law Quiz 1) 16. 2 liters of chlorine gas, at a pressure of 700 mm Hg and a temperature of 20 o. C, has what volume when the pressure is increased to 850 mm Hg?

2) Lithium metal reacts with nitric acid to form hydrogen gas and lithium nitrate. What volume of hydrogen gas, at 273 K and 0. 90 atm, is produced using 46. 3 grams of nitric acid?
3) 2. 0 liters of dichlorine monoxide, at 32 o. F and 1000 torr, contains how many grams of chlorine gas?

4) Ammonia gas is oxidized to produce water and nitric oxide. NH 3 + O 2 H 2 O + NO If 7. 4 liters of ammonia, measured at 0ºC and 950 torr, is completely reacted, how many grams of nitric oxide gas, NO, will be produced?

5) 5200 g of mercury are produced via the decomposition of mercury(II) oxide. How many liters of oxygen gas are produced at 32 o. F and 2. 0 atm of pressure?

6) 10. 4 liters of nitrous oxide at 178 mm Hg, 45 o. F, is at what pressure when its volume is decreased to 2. 4 liters?
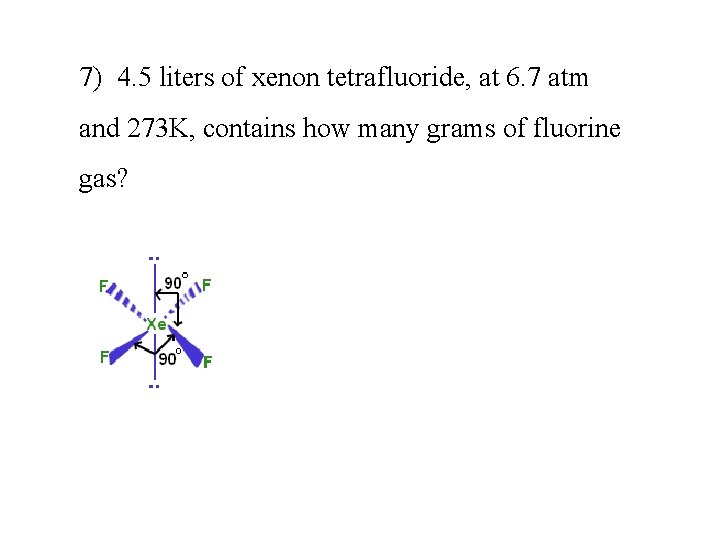
7) 4. 5 liters of xenon tetrafluoride, at 6. 7 atm and 273 K, contains how many grams of fluorine gas?

8) Air bags inflate via this reaction, Na. N 3 Na + N 2 An air bag is inflated with 36 liters of nitrogen gas at 1. 75 atm and 0 o. C. How many grams of Na. N 3 decomposed when the bag was inflated?
- Slides: 21